Research Article - Onkologia i Radioterapia ( 2020) Volume 14, Issue 6
Trace elements and electrolytes disturbances in cancerous patients: A prospective comparison study
Enas Khudhair Al-Bdaer* and Ahmed Salih AlsheweredEnas Khudhair Al-Bdaer, Department of Medicine, College of Medicine, Basra, 60061,, Iraq, Email: Rafidalkhalidy79@gmail.com, edicalresearch68@yahoo.com
Received: 04-Nov-2020 Accepted: 26-Nov-2020 Published: 30-Nov-2020
Abstract
Cancer is still the leading cause of most morbidity and mortality in many countries worldwide. The abnormalities in electrolyte concentrations including hypokalaemia or hyperkalaemia, hypomagnesemia or hypomagnesemia, hypocalcaemia or hypercalcemia, and trace elements deficient can occur due to cancer itself or due to its management like the use of chemotherapy. A prospective comparison study was carried out on newly diagnosed cancer patients before and after receives cytotoxic agents for a period of four months from March 2020 to July 2020. A total of 100 newly diagnosed cancerous patients enrolling and recruited. Demographic characters of patients like age, gender, address, jobs, co-morbid diseases, BSA, cancer types, and chemotherapy regimens were collected from the medical records of the participants. The Calcium Colorimetric Assay Kit, Magnesium Assay Kit, Zinc Assay Kit, and Potassium (K) turbid metric Assay Kit were used. In this study, 80(80%) were females and 20(20%) were males enrolled, with a mean age was 46.35±13.8 years. 79/100 (79%) of participants lived in Basra city. The mean body surface area (BSA) was 1.7±0.23 m2. The majority were females with breast cancers about 56(56%). Regarding chemotherapy regimens, the AC+Taxen protocol was mostly used as 54%. There was no difference between pre and post-chemotherapy concentrations of calcium. The same for unchanged potassium concentrations. There was a significant decline in magnesium concentration in pre- and post-chemotherapy (2.23±0.34 vs 1.88±0.37 mg/dL), but this deviation still within the normal range with a strong significant association (P<0.000). Besides, the mean zinc concentration dropping from (90.61±13.05 μg/dL) to (78.51±13.56 μg/dL), which was highly statistically differences (P<0.000). These findings could be explained by the fact that most if not all those patients took supplement either described by physicians or by themselves before and during cancer management. Differences in the results between the pre and post-chemotherapy may be unchanged or there was a large decrement in electrolytes level, but this is still within normal. This may be partially due to the replacement supplement of elements before and after treatment.
Keywords
trace elements, electrolyte, cancer, Mg2+, Ca2+, K1+, Zinc
Introduction
The Ministry of Health/Environment and Iraqi Cancer Board released an annual report of the Iraqi cancer Registry in 2015 about the incidence and mortality of cancer in all Iraqi provinces. They found that the total new cases of cancer were 25,269 with male to female ratio of 0.8:1. The highest incidence was recorded in Karbala province (98.7/100,000 population) and the lowest was in Anbar province (26.1/100,000 ppulations). The top ten cancers were breast (19.1%), lung (8.1%), leukemia (6.3%), Central Nervous System (CNS) (6.1%), colorectal (5.7%), bladder (5.1%), Non-Hodgkin lymphomas (4.3%), thyroid (3.8%), skin (3.2%), and stomach (3.2%). The highest incidence of cancer in men was lung cancer (6.7/100,000 males population), while in women was breast cancer (25.8/100,000 females population). The cancer mortality during the year 2015 was 8,825 deaths with an equal ratio between both sexes. The highest incidence of cancer mortality has belonged to lung (7.3/100,000 populations) followed by breast (2.7/100,000 populations) [1].
Chemotherapy is a type of cancer treatment that utilizes one or more anti-cancer or chemotherapeutic agents as part of a treatment regimen. Chemotherapy may be given with curative intent as combinations of drugs, or to prolong life or to reduce symptoms in palliative chemotherapy [2]. Chemotherapy side effects are traced to destroy normal cells that divide rapidly and thus include cells in the bone marrow, digestive tract, and hair follicles [2, 3]. Sometimes received of cytotoxic agents maybe lead to severe electrolyte abnormalities, as hypokalemia and hypomagnesemia, which can result in QTinterval prolongation and fatal ventricular arrhythmia, if levels are not carefully followed and maintained in the high-normal range. The laboratory abnormalities including hypokalemia, hypomagnesemia, hyperglycemia, and edema are seen in about one-half of cancerous patients. Different abnormalities as hyperkalemia, hypocalcemia, hypoglycemia, acidosis, hypophosphatemia, hypocalcemia, and hyperglycemia and increased liver function tests are reported [4]. Here, we try to determine whether electrolytes deficient occur in cancerous patients due to the administration of cytotoxic drugs altering cancer management.
Methods
Study design and setting
A prospective comparison study was carried out on newly diagnosed cancer patients before and after receives cytotoxic agents for a period of four months from March 2020 to July 2020. Assessments of the studied samples will be conducted as a baseline before receiving drugs, while the period after administration may be at the first, or second, or third cycles of chemotherapy.
Participants
A total of 100 newly diagnosed cancerous patients enrolling and recruited at their first visit to the center. Each patient attending our center meeting the inclusion criteria were invited to be included in our study after which written informed consent was obtained. Demographic characters of patients like age, gender, address, jobs, co-morbid diseases, BSA, cancer types, and chemotherapy regimens were collected from the medical records of the participants. Follow up will be recorded after the first, third, and sixth cycles of chemotherapy.
Inclusion criteria
• Patients do not receive chemotherapy
• Cooperative people to respond to the data collection
Exclusion criteria
• Anticancer treatment that did not include chemotherapy
• Patients in the second cycle of chemotherapy
• Low-performance status
The kits
The panel used for evaluation of minerals level included the following kits: Calcium Colorimetric Assay Kit (SIGMAALDRICH, Catalogue No.: MAK022), Magnesium Assay Kit (SIGMA-ALDRICH, Catalogue No.: MAK026), Zinc Assay Kit (SIGMA-ALDRICH, Catalogue No.: MAK032), Potassium (K) turbidimetric Assay Kit (SIGMA-ALDRICH, Catalogue No.: MAK054) [5].
Data sources
In non-anticoagulated tubes (ATACO/China, Catalogue No.: 753134), the blood samples were collected (2-3 ml). We centrifuged it for 10min at 1000-3000rpm and then take a supernatant tested immediately. We diluted the concentrated washing solution with double distilled water (1:25). Each kit has its directions and steps as mention in the leaflets [5].
Procedures
In vitro tests for the quantitative determination of minerals in serum, in collection tubes that were have not contained chelating anticoagulants such as EDTA, fluoride, and oxalate. In an alkaline solution, a complex added. The magnesium concentration is measured photometrically via the decrease in the xylidyl blue absorbance. In calcium assay, a chromogenic complex is used to detections. We added up to 2.5 μL sample into each well of a 96 well black plate. Serum may be diluted with buffer before addition to wells if higher than 10 mM level is expected [5].
Ethical approval and patients consent
Written informed consent was obtained from the patients or the parents/guardians of minors for those below the age of 18 years, for participating in this study, and was conducted according to the ethical standards established by the 1964 Declaration of Helsinki. The Medical Ethical Committee of Basra University and Basra Oncology Center approved this study (code:xxxxxxxx).
Statistical
We use mean and standard deviation to represent the data while describing variables presented using their numbers and parentage. Two-sided paired t-test for variables was used. SPSS version 20 was used for data entry and analysis. p-value was considered significant if <0.05.
Results
Demographic finding analysis
We enrolled 100 patients, 80 (80%) were females and 20 (20%) were males, with a mean age was 46.35 ± 13.8 years. The most distributed age group belonged to fifth decades 30(30%) patients. 79/100 (79%) of participants lived in Basra city. 60 (60%) of women were housewives. The mean Body Surface Area (BSA) was 1.7 ± 0.23 m2. The majority were females with breast cancers about 56 (56%). Regarding chemotherapy regimens, the AC+Taxen protocol was mostly used as 54% (Table 1).
| Variables | No. | % | |
| Gender | Male | 20 | 20 |
| Female | 80 | 80 | |
| Age (years) 46.35 ± 13.8 | <20 | 1 | 1 |
| 21-30 | 7 | 7 | |
| 31-40 | 14 | 14 | |
| 41-50 | 30 | 30 | |
| 51-60 | 25 | 25 | |
| 61-70 | 15 | 15 | |
| >70 | 8 | 8 | |
| Address | Basra | 79 | 79 |
| Others | 21 | 21 | |
| Occupation | Employer | 21 | 21 |
| Housewife | 60 | 60 | |
| Non- employer | 19 | 19 | |
| BSA (m2) 1.7 ± 0.23 | <1.7 | 41 | 41 |
| 1.7 | 20 | 20 | |
| >1.7 | 39 | 39 | |
| Comorbidity | Yes | 38 | 38 |
| No | 62 | 62 | |
| Cancer types | Bladder | 10 | 10 |
| Breast | 56 | 56 | |
| Cervix | 8 | 8 | |
| Colorectal | 7 | 7 | |
| Lung | 14 | 14 | |
| Lymphoma | 2 | 2 | |
| Ovary | 2 | 2 | |
| Prostate | 1 | 1 | |
| Chemotherapy protocols | 5FU+Cisplatin | 5 | 5 |
| AC+Taxen* | 54 | 54 | |
| Carboplatin+Taxen* | 16 | 16 | |
| FOLFIRINOX** | 2 | 2 | |
| Xelox | 20 | 20 | |
| Taxen | 3 | 3 | |
*Taxen: Paclitaxel or Docetaxel; AC: Adriamycin and Cyclophosphamide; **FOLFIRINOX: Folinic acid, 5FU, Irinotecan, Oxaliplatin; Xelox: Oxaliplatin and Capecitabine
Tab. 1. General characters of the study (n=50)
Electrolytes finding analysis
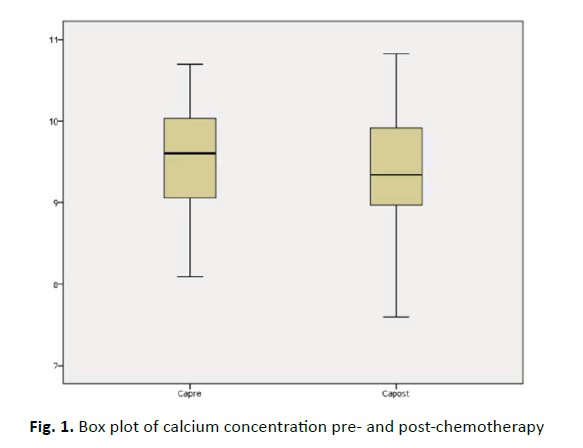
In (Table 2), we figured all mean value of electrolytes level in the study. Regarding calcium, there was no difference between pre and post-chemotherapy and could be explained by the fact that most if not all those patients took calcium supplement either described by physicians or by themselves (P=0.08), (Figure 1).
| Electrolytes | Pre | Post | Paired t-test | P-value |
| (Mean ± SD) | ||||
| Ca2+ (8.7-10.2 mg/dL) | 9.54 ± 0.63 | 9.4 ± 0.61 | 1.771 | 0.08 |
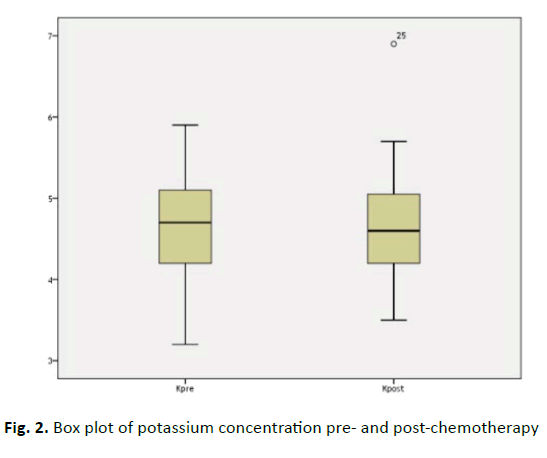
| K+ (3.5-5 meq/L) | 4.65 ± 0.61 | 4.66 ± 0.57 | 0.239 | 0.812 |
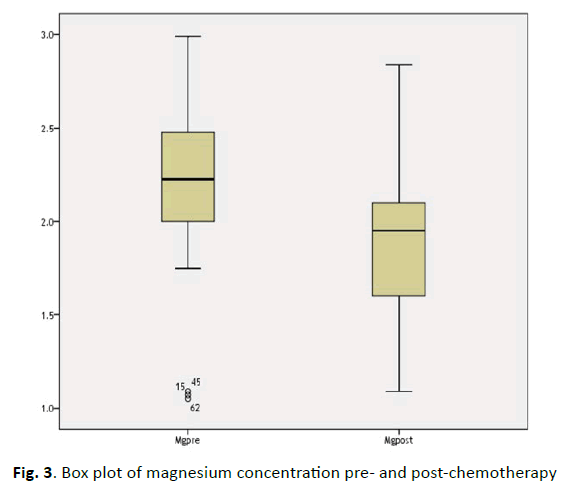
| Mg2+ (1.5-2.3 mg/dL) | 2.23 ± 0.34 | 1.88 ± 0.37 | 6.747 | 0.000 |
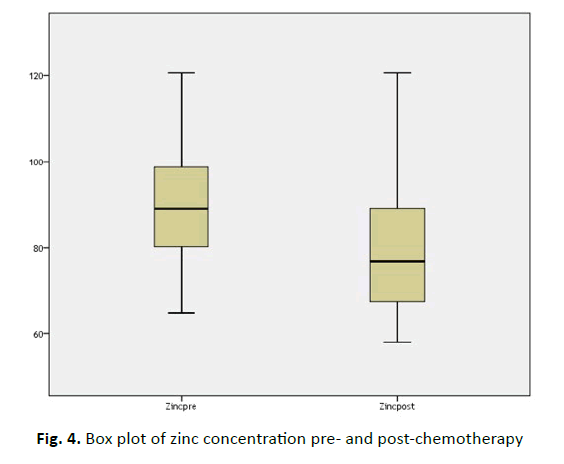
| Z (75-120 μg/dL) | 90.61 ± 13.05 | 78.51 ± 13.56 | 6.761 | 0.000 |
Tab. 2. Trace elements concentration pre- and post-chemotherapy
Figure 1: Box plot of calcium concentration pre- and post-chemotherapy
Approximately, potassium concentrations always almost unchanged in both arms (4.65 ± 0.61 vs 4.66 ± 0.57 meq/L). This is with no significant difference (P=0.812), (Figure 2)
Figure 2: Box plot of potassium concentration pre- and post-chemotherapy
There was a significant decline in magnesium concentration in pre and post-chemotherapy (2.23 ± 0.34 vs 1.88 ± 0.37 mg/dL), but this deviation still within the normal range. The variation in concentration between pre- and post-chemotherapy strong significant association (P<0.000), (Figure 3).
Figure 3: Box plot of magnesium concentration pre- and post-chemotherapy
Lastly, the mean zinc concentration initially was (90.61 ± 13.05 μg/dL), whereas it dropped to (78.51 ± 13.56 μg/dL), despite this alteration it was still within the normal range. This finding has high statistically significant differences (P<0.000), (Figure 4).
Figure 4: Box plot of zinc concentration pre- and post-chemotherapy
Discussion
Diarrhea, nausea, and vomiting are common side effects after doses of Cyclophosphamide, Dacarbazine, Ifosfamide, Nitrogen mustard, BCNU, Procarbazine, Streptozocin, Temozolomide, Cisplatin, Carboplatin, Oxaliplatin, 5-FU, Leucovorin, Capecitabine, Gemcitabine, 6-MT, MTX, Hydroxyurea, Pemetrexed, anti-tumor antibiotics, Paclitaxel, Docetaxel, Vincristine, Vinorelbine, Etoposide, Irinotecan, and Topotecan [2, 6-14]. Hypotension, hypokalemia, and renal tubular acidosis are common in administering of ifosfamide, and streptozocin [6]. Cisplatin can cause hypokalemia and hypomagnesemia. Carboplatin causes a cation electrolyte imbalance. Actinomycin D lead to elevation of LFTs, and hypocalcemia [6-14].
Cardiopulmonary arrest and/or sudden death have been reported in patients receiving cetuximab with radiation therapy for squamous cell cancer of the head and neck [4]. These events occurred within 1 to 43 days of the last treatment. ECGs should be performed at baseline and 7 days after initiation, and periodically thereafter, as well as following any dose adjustments [4]. Assess fluid status and serum electrolytes at baseline, and follow closely during therapy, especially if diuretics are also being administered, because fatal hypokalemia has been reported in patients receiving ifosfamide and diuretic therapy [4].
Bisphosphonate is the drug indicated for the treatment of hypercalcemia of malignancy and the treatment of patients with multiple myeloma and patients with documented bone metastases from solid tumors, in conjunction with standard antineoplastic therapy [4, 6]. It has many side effects as electrolyte dysfunction including hypocalcemia, hypokalemia, hypomagnesemia, and hypophosphatemia [4, 6].
During the initial management of the patient, it is important to monitor serum electrolyte, calcium, phosphorus, blood urea nitrogen, and creatinine levels every 6 to 8 hours for 48 hours [4, 15].
Hypomagnesemia occurs in approximately half the patients receiving cisplatin and may worsen renal impairment [15]. The main site of injury is the corticomedullary S3 segment of the proximal tubule where cisplatin is preferentially absorbed, leading to dysfunction of the local transport mechanisms involving sodium, water, glucose, amino acids, and magnesium transport [4]. The pattern of injury seen includes low GFR, Fanconi-like syndrome with glucosurea and aminoacidurea, hypomagnesemia, and salt-wasting [16]. Hypomagnesemia may be involved in the development of vasospastic disorders [16]. Hypomagnesemia, common toxicity of cisplatin, occurs in 75% to 87% of patients [4].
Magnesium is an important enzyme cofactor and is essential to several metabolic processes [17]. The mineral help to regulate blood pressure, insulin metabolism, muscular contraction, vasomotor tone, cardiac excitability, nerve transmission, and neuromuscular conduction system, and is necessary for RNA, DNA, and protein synthesis among several other functions [18]. Disruptions in homeostatic levels of magnesium (oftentimes hypomagnesemia) can impact the nervous system, muscles, or can lead to cardiac abnormalities [18, 19]. Hypermagnesemia symptoms include diarrhoea and other gastrointestinal effects, thirst, muscle weakness, drowsiness, severe back, and pelvic pain, hypotension, dizziness, confusion, difficulty breathing, lethargy, and deterioration of kidney function. Other more severe symptoms associated with magnesium overdose include loss of consciousness, respiratory arrest, cardiac arrhythmias, and cardiac arrest [18-20]. Both Ca2+ and K+ concentration unchanged in our study. Those two important elements in all body systems function.
Calcium plays an important vital role in the anatomy, physiology, and biochemistry of organisms and the cell, particularly in signal transduction pathways [4]. The bones act as a major mineral storage site for the element and release Ca2+ ions into the bloodstream under controlled conditions [4, 6].
Potassium is an essential nutrient identified as a shortfall nutrient by the 2015-2020 Advisory Committee of Dietary Guidelines for Americans [21]. Some examples of potassiumrelated complications include life-threatening arrhythmia, neuromuscular dysfunction, diarrhoea, nausea, and vomiting [22, 23]. It maintains an electrolyte gradient on cell surfaces, keeping at specific concentrations inside and outside of the cell; this impacts fluid and electrolyte balance, nerve transmission, muscle contraction, as well as cardiac and kidney function [22, 23]. Vomiting, diarrhoea, renal disease, medications, and other conditions are altering potassium excretion or shift it inside or outside of cells [24]. Hypokalemia reduces intravascular volume, by reducing sodium reabsorption through an increase in urinary sodium excretion [24]. Reduced serum potassium (or imbalance) increases the risk of ventricular arrhythmia, heart failure, and Left Ventricular Hypertrophy (LVH) [25]. Hyperkalemia may result in death due to various causes of cardiovascular, neurological, and musculoskeletal manifestations in nature [26].
In this study, zinc level different in both phases of the study, but it still with normal range. Zinc is a necessary trace element in the diet, forming an essential part of many enzymes, and playing an important role in protein synthesis and cell division [27-29]. Zinc deficiency is associated with anaemia, short stature, hypogonadism, impaired wound healing, and geophagia [27]. Newer studies suggest implies that an imbalance of zinc is associated with the neuronal damage associated with traumatic brain injury, stroke, and seizures [28]. It is utilized for boosting the immune system, treating the common cold and recurrent ear infections, as well as preventing lower respiratory tract infections [29]. In HL-60 cells (promyelocytic leukaemia cell line), zinc enhances the up-regulation of A20 mRNA, which, via the TRAF pathway, decreases NF-kappa B activation, leading to decreased gene expression and generation of tumour necrosis factor-alpha (TNF-alpha), IL-1beta, and IL-8.
Conclusion
Differences in the results between the pre and post-chemotherapy may be unchanged or there was a large decrement in electrolytes level, but this is still within normal. This may be partially due to the replacement supplement of elements before and after treatment. Always almost, must be recommended those elements for such patients due to the possibility of deficient as a result of cancer itself or/and its management.
Competing Interests
No conflict of interest.
References
- Iraqi Cancer Registry. Ministry Of Health, Iraqi Cancer Board, Baghdad. 2015.
- Corrie PG, Pippa G. Cytotoxic chemotherapy: clinical aspects. Med. 2008;36:24-28.
- Wagner AD, Syn NL, Moehler M, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8:CD004064.
- Perry MC, Doll DC, Freter CE. The chemotherapy sourcebook. 5th ed. LIPPINCOTT WILLIAMS & WILKINS, a WOLTERS KLUWER business, Philadelphia, PA 19103, USA. 2012;1:181-191.
- https//www.sigmaaldrich.com/catalog
- Makin G, Hickman JA. Apoptosis and cancer chemotherapy. Cell Tissue Res. 2000;301:143-152.
- Gill P, Grothey A, Loprinzi C. Nausea and Vomiting in the Cancer Patient. Oncol. 2006;8:1482-1496.
- Cohen L, De Moor CA, Eisenberg P. Chemotherapy-induced nausea and vomiting: incidence and impact on patient quality of life at community oncology settings. Support Care Cancer. 2007;15:497-503.
- Lind MJ. Principles of cytotoxic chemotherapy. Med. 2008;36:19-23.
- Berger AM, Abernethy AP, Atkinson A. NCCN clinical practice guidelines cancer-related adverse effects. J Natl Compr Canc Netw. 2010;8:904-931.
- Ian O. The MASCC textbook of cancer supportive care and survivorship.Springer sci & business media. 2010;1:351.
- Weeks JC, Catalano PJ, Cronin A. Patients' expectations about effects of chemotherapy for advanced cancer. New Eng J Med. 2012;367:1616-1625.
- NCCN. Clinical practice guidelines in oncology. NCCN GUIDELINES FOR SUPPORTIVE CARE. Adult cancer pain, anti-emesis, cancer- and chemotherapy-induced anemia, cancer-associated venous thromboembolic disease, cancer-related fatigue, distress management, palliative care, prevention and treatment of cancer-related toxicities. 2019.
- Lajer H, Kristensen M, Hansen HH. Magnesium depletion enhances cisplatin-induced nephrotoxicity. Cancer Chemother Pharmacol. 2005;56:535-542.
- Kim SW, Lee JU, Nah MY. Cisplatin decreases the abundance of aquaporin water channels in rat kidney. J Am Soc Nephrol. 2001;12:875-882.
- Swaminathan R. Magnesium metabolism and its disorders. Clin Biochem Rev. 2003;24:47-66.
- Grober U, Schmidt J, Kisters K. Magnesium in prevention and therapy. Nutrients. 2015;7:8199-8226.
- Schwalfenberg GK, Genuis SJ. The importance of magnesium in clinical healthcare. Scientifica (Cairo). 2017;2017;4179326.
- Bokhari SR, Siriki R, Teran FJ, Batuman V. Fatal hypermagnesemia due to laxative use. Am J Med Sci. 2018;355:390-395.
- Papanikolaou Y, Fulgoni VL. Grains contribute shortfall nutrients and nutrient density to older us adults: data from the national health and nutrition examination survey, 2011-2014. Nutrients. 2018;10:534.
- Stone MS, Martyn L, Weaver CM. Potassium intake, bioavailability, hypertension, and glucose control. Nutrients. 2016;8:444.
- Viera AJ, Wouk N. Potassium disorders: hypokalemia and hyperkalemia. Am Fam Physician. 2015;92:487-495.
- Hinderling PH. The pharmacokinetics of potassium in humans is unusual. J Clin Pharmacol. 2016;56:1212-1220.
- He FJ, MacGregor GA. Beneficial effects of potassium on human health. Physiol Plant. 2008;133:725-735.
- Bosse GM, Platt MA, Anderson SD, Presley MW. Acute oral potassium overdose: the role of hemodialysis. J Med Toxicol. 2011;7:52-56.
- Berni Canani R, Buccigrossi V, Passariello A: Mechanisms of action of zinc in acute diarrhea. Curr Opin Gastroenterol. 2011;27:8-12.
- Prakash A, Bharti K, Majeed AB. Zinc: indications in brain disorders. Fundam Clin Pharmacol. 2015;29:131-149.
- Prasad AS. Zinc in human health: effect of zinc on immune cells. Mol Med. 2008;14:353-357.
- Dardenne M. Zinc and immune function. Eur J Clin Nutr. 2002;56:S20-S23.