Research Article - Onkologia i Radioterapia ( 2021) Volume 15, Issue 10
The neurotransmitters imbalance in participants receive cytotoxic agents
Emad Hazim Mhmood*Emad Hazim Mhmood, Ibn Sina Hospital, DOH of Ninwa, Ministry of health/ Environment, Mosul, Iraq, Email: Ayam.mohammad@yahoo.com
Received: 01-Sep-2021 Accepted: 01-Oct-2021 Published: 08-Oct-2021
Abstract
Neurotransmitters are endogenous chemical compounds that have the ability for neurotransmission and play a major role in shaping everyday life and functions. Cytotoxic drugs are a type of cancer and other diseases like rheumatoid arthritis, SLE, and different autoimmune syndromes treatment that utilize one or more chemotherapeutic agents as part of a regimen. The study aims are to calculate levels of neurotransmitters in patients receiving cytotoxic drugs and determined the effect of these agents on NTs concentration before and after. A prospective comparison study carried out before and after receives cytotoxic treatment for a period of three months from June 2020 to September 2020. Assessments of the studied samples will be conducted as a baseline before and after receiving drugs. A total of 50 participants were involved in the study. The blood samples were collected about 5 ml and diluted in the concentrated washing solution. The neurotransmitters evaluated are Adrenaline, Acetylcholine, Dopamine, Histamine, Glutamate, GABA, Serotonin, and Somatostatin. The mean age of the sample of the study was 49.28 ± 12.44. Patients with comorbidity conditions were 60%. The mean acetylcholine concentration pre and post receiving cytotoxic drugs was relatively not altering. Cytotoxic agents affected the mean epinephrine concentration, which was directly dropping to half. There was a decline of concentration of somatostatin post treatment compared with pre (from 33.83 ± 9.78 ng/L to 8.4 ± 2.66 ng/L), with a significant association (t-test=2.3, p=0.028). The mean concentration of dopamine pre was relatively unchanged from post-treatment. There was no differences association between pre and post mean level of histamine in this study. In this study, we found a high level of glutamine in the pre-phase (2025.4 ± 753.7 μmol/L), but it converted to normal level post treatment, with statistically significant differences (t-test=9.051, p<0.000). GABA concentration, the pre was above the normal range (1.31 ± 0.18 mumol/l), while the post was within normal, this dropping curve was statistically significant differences (t-test=9.442, p<0.000). There was a slight decrease in serotonin level among pre to post (from 366.6 ± 237.72 ng/mL to 298.77 ± 231.8 ng/mL), but these changes not significant. In conclusion, the NTs concentration may be altered by the administration of cytotoxic drugs in chronic diseases in a different pattern either decrease or return to normal.
Keywords
neurotransmitters, cytotoxic agents, CNS tumours, adrenaline, acetylcholine, dopamine, histamine, glutamate, GABA, serotonin, somatostatin
Introduction
Neurotransmitters are endogenous chemical compounds that have the ability for neurotransmission by carrying messages among neurons via influence on the postsynaptic membranes, changing the structures of the synapse, and communicate by sending reverse-direction messages that affect the release or reuptake of transmitters [1]. It transmits signals across a chemical synapse, such as a neuromuscular junction, from one neuron to another neuron, muscle cell, or gland cell, and is released from synaptic vesicles in synapses into the synaptic cleft, where they are received by receptors. Many neurotransmitters are synthesized from simple and plentiful precursors such as amino acids, which are readily available from the diet. Neurotransmitters play a major role in shaping everyday life and functions [1].
Cytotoxic drugs are a type of cancer and other diseases like rheumatoid arthritis, SLE, and different autoimmune syndromes treatment that utilize one or more chemotherapeutic agents as part of a regimen [2]. The traditional chemotherapeutic agents are almost always cytotoxic, are interfering with cell division (mitosis), besides that the chemotherapy destroys cells whether cancerous or healthy, which may then lead to cell death [2]. Chemotherapy side effects are traced to destroy normal cells that divide rapidly and thus include cells in the bone marrow, digestive tract, and hair follicles [2,3].
There are several modalities in the administration of chemotherapeutic drugs [2,4-7], including: Induction, combined, consolidation, intensification, combination, neoadjuvant, adjuvant and salvage or palliative chemotherapy.
In this research, we try to compare NTs concentrations at pre and post-treatment with cytotoxic drugs using different morbid and chronic diseases like CNS tumours, RA, SLE, and others.
Methods
Study design and setting
A prospective comparison study carried out before and after receives cytotoxic treatment for a period of three months from June 2020 to September 2020. Assessments of the studied samples will be conducted as a baseline before and after receiving drugs. A total of 50 participants were involved in the study, who accepted patient inform consent.
Inclusion criteria
➢ Patients before undergo the first cycle of cytotoxic.
➢ Those who have to agree to involvement in the study.
Exclusion criteria
➢ Patients who changing drugs protocols.
➢ Those refused study entrance from the beginning.
➢ Those whom referral from another centre.
➢ Patients receiving chemotherapy previously.
➢ Uncomfortable.
Data measurement
The blood samples were collected about 5 ml put in a refrigerator at 4°C. Taken test when it balances to room temperature and diluted the concentrated washing solution with double distilled water (1:25). Each neurotransmitter kit has its directions and steps of preparation procedure as mention in the special leaflets.
Neurotransmitters
Adrenaline (epinephrine), Acetylcholine, Dopamine, Histamine, Glutamate, GABA, Serotonin, and Somatostatin. Foods or liquids such as pineapple, eggplant, avocados, bananas, currants, kiwis, melon, mirabelles, plums, peaches chocolate, gooseberries, tomatoes, or walnuts, should be avoided 2 days before and including the day of the sample collection (Figure 1).
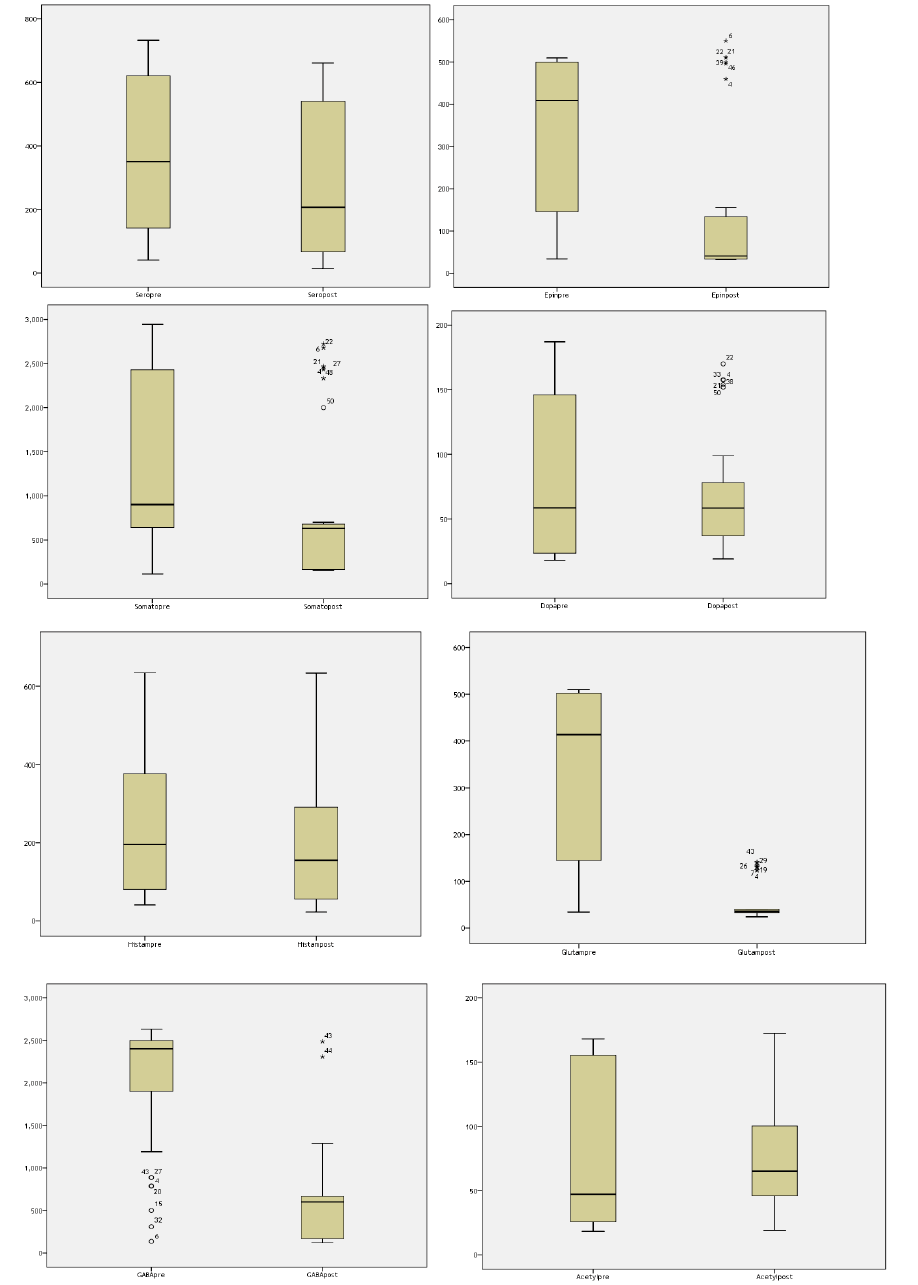
Figure 1: Box plot of neurotransmitters concentration before and after cytotoxic
Statistical
We used mean and SD to represent the data while describing variables presented using their frequencies and parentage. One way paired T-test for variables was used. SPSS version 24 was used for data entry and analysis. P-value was considered significant if <0.05.
Result
Patient’s demographic variables
25(50%) of patients were females and 25(50%) were males. The mean age of the sample of the study was 49.28 ± 12.44 years with a median of 50 years. Most patients were from urban areas 37(74%), and the rest from rural 13(26%). Most female patients were housewives. Patients with comorbidity conditions were 60%.
Regarding Body Surface Area (BSA), we recorded 50% of patients with BSA equal or less than 1.7 m2 for each. The mean Body Mass Index (BMI) of patients was 35.77 ± 3.63 m2/Kg. Most patients figured to overweight 60%.
The majority of patients suffered from RA 40%. The most regimens of drugs used were RCHOP (cyclophosphamide and rituximab) in 76% of patients (Table 1).
| Variables | No. (%) | Mean±SD | |
|---|---|---|---|
| Gender | Male | 25 (50) | |
| Female | 25 (50) | ||
| Total | 50 | ||
| Age (years) | 21-30 | 3 (6) | 49.28±12.44 |
| 31-40 | 6 (12) | ||
| 41-50 | 20 (40) | ||
| 51-60 | 10 (20) | ||
| 61-70 | 11 (22) | ||
| Total | 50 | ||
| Address | Urban | 37 (74) | |
| Rural | 13 (26) | ||
| Total | 50 | ||
| Occupation | Employer | 20 (40) | |
| Housewife | 15 (30) | ||
| Non- employer | 10 (20) | ||
| Student | 5 (10) | ||
| Total | 50 | ||
| Comorbidity | Present | 30 (60) | |
| Not present | 20 (40) | ||
| Total | 50 | ||
| BSA (m2) | <1.7 | 25 (50) | 1.66±0.13 |
| = 1.7 | 25 (50) | ||
| Total | 50 | ||
| BMI (m2/Kg) | Normal (18.6-24.9) | 15 (30) | 35.77±3.63 |
| Overweight (25-29.9) | 30 (60) | ||
| Moderate obesity (30-34.9) | 3 (6) | ||
| Severe obesity (35-39.9) | 2 (4) | ||
| Total | 50 | ||
| Diseases | Cancers (CNS tumors) | 12 (24) | |
| Rheumatoid arthritis | 20 (40) | ||
| SLE | 8 (16) | ||
| Autoimmune syndromes | 6 (12) | ||
| Others | 4 (8) | ||
| Total | 50 | ||
| Cytotoxic | R-CHOP | 38 (76) | |
| Temodalizamide | 8 (16) | ||
| Vincristine | 4 (8) | ||
| Total | 50 | ||
Tab. 1. All study variables (n=50)
Our findings showed that the mean acetylcholine concentration pre and post receiving cytotoxic drugs was relatively not altering (from 7.68 ± 5.85 U/mL to 7.86 ± 4.75 U/mL), with no significant differences (t-test=0.145, p=0.885). Cytotoxic agents affected the mean epinephrine concentration, which was directly dropping to the half (from 301.1 ± 193.68 pmol/L to 141 ± 168.6 pmol/L), with strong statistical significant differences (t-test=4.6, p=0.000).
There was a massive decline of concentration of somatostatin post treatment compared with pre (from 33.83 ± 9.78 ng/L to 8.4 ± 2.66 ng/L), with a significant association (t-test=2.3, p=0.028).
The mean concentration of dopamine pre was 82.1 ± 59.5 pg/ mL, whereas it was 71.4 ± 44.4 pg/mL post-treatment. These findings were not significant (t-test=0.789, p=0.436).
There was no differences association between pre (25.26 ± 19.72 nm/mL) and post (20.23 ± 18.11 nm/mL) mean level of histamine in this study (t-test=1.705, p=0.096).
In this study, we found a high level of glutamine in the samples in pre-phase (2025.4 ± 753.7 μmol/L), but it converted to normal level post-treatment (563.8 ± 531.2 μmol/L), with statistically significant differences (t-test=9.051, p<0.000).
Regarding GABA concentration, the pre was above the normal range (1.31 ± 0.18 mumol/l), while the post was within normal (0.53 ± 0.39 mumol/l). The dropping curve was statistically significant differences (t-test=9.442, p<0.000).
There was a slight decrease in serotonin levels among pre to post (from 366.6 ± 237.72 ng/mL to 298.77 ± 231.8 ng/mL), but these changes not significant (t-test=1.565, p=0.126) (Table 2).
| NTs (Normal range) | Paired Differences | t | df | p-value (2-tailed) | |||
|---|---|---|---|---|---|---|---|
| Pre | Post | 95% Confidence Interval | |||||
| Lower | Upper | ||||||
| Acetyl choline (8-18 U/mL) | 7.68 ± 5.85 | 7.86 ± 4.75 | -2.735 | 8.369 | -0.145 | 37 | 0.885 |
| Epinephrine (<273 pmol/L) | 301.1 ± 193.68 | 141 ± 168.6 | 89.66 | 230.6 | 4.6 | 36 | 0 |
| Somatostatin (<25 ng/L) | 33.83 ± 9.78 | 8.4 ± 2.66 | 6.68 | 10.92 | 2.3 | 34 | 0.028 |
| Dopamine (<87pg/mL) | 82.1 ± 59.5 | 71.4 ± 44.4 | -16.78 | 38.06 | 0.789 | 33 | 0.436 |
| Histamine (=10 nm/mL) | 25.26 ± 19.72 | 20.23 ± 18.11 | -9.33 | 10.98 | 1.705 | 40 | 0.096 |
| Glutamine (550-750 µmol/L) | 2025.4 ± 753.7 | 563.8 ± 531.2 | 1134.39 | 1788.79 | 9.051 | 37 | 0 |
| GABA (0.2-0.8 mumol/l) | 1.31 ± 0.18 | 0.53 ± 0.39 | 0.2 | 1.31 | 9.442 | 40 | 0 |
| Serotonin(50-200 ng/mL) | 366.6 ± 237.72 | 298.77 ± 231.8 | -19.97 | 155.65 | 1.565 | 37 | 0.126 |
Tab. 2. Comparison between NTs concentration pre-and postcytotoxic administrationoff
Discussion
Nausea and vomiting are common side effects of chemotherapy and molecular-targeted therapy. With the advent of newer agents used to treat Chemotherapy-Induced Nausea and Vomiting (CINV), the incidence has greatly decreased; however, this continues to be a significant cause of concern to patients. The incidence of acute CINV (within 24 hours of drug administration) is estimated to be 37% to 59% and delayed CINV (>24 hours after administration) occurs in 47% to 75% of patients treated with chemotherapy [8,9]. Although emesis appears to be better controlled with the newer antiemetic agents, nausea is still a problem and can adversely affect the patient’s quality of life [9-11]. CINV can cause a decrease in quality of life [10], disruption in treatment schedules, decrease in compliance, medical complications such as dehydration, poor nutrition, electrolyte imbalance, frequently resulting in hospitalization, and elevation in the costs of health care and loss of workdays [12].
There has been a lot of work done in the past couple of decades toward elucidating the pathophysiology of CINV, which is complex and involves several pathways. The concept of a single “vomiting centre” in the brain proposed by Wang and Morison in 1950 is now replaced by a complex set of neurons called the “central pattern generator,” believed to coordinate the series of events that lead to emesis, as proposed by Koga and Fukuga in 1992 [6].
It is now believed that the principal actors are the neurotransmitter receptors for serotonin 5-hydroxy tryptamine 3 (5HT3), dopamine, substance P, and cholecystokinin, which are located in the entero-chromaffin cells in the small intestinal epithelium and are the terminal ends of vagal afferents. Antineoplastic agents stimulate these vagal afferents either by direct toxicity or by breaking of the mucosal barrier and causing the release of neurotransmitters from the entero-chromaffin cells, which then bind to the receptors; the stimulus is carried to the dorsal brain stem (NTS and area postrema) and activates the central pattern generator [13].
Area postrema contains the chemotherapy trigger zone and may have different mechanisms of activation, which are incompletely understood. The amygdala is another less-understood area of the brain that is associated with CINV. 5HT receptors are found in all three key areas: the vagal afferent chain, NTS, and area postrema. 5HT3 is the most important of all of the receptors in mediating CINV. It is has been shown that the abdominal vagal afferents are the principal mediators of CINV with 5HT3 agonists [14]. Although dopamine, 5HT3, and substance P (a neurokinin) all exert pro-emetic effects, endocannabinoids have antagonistic action [14, 15].
The size of neurotransmitters is about the size of one amino acid; but, some neurotransmitters may be larger than the size of proteins or peptides. The short-term exposure of the receptor to a neurotransmitter is typically sufficient for causing a postsynaptic response by way of synaptic transmission, this leads to a threshold action potential, and then a neurotransmitter is released at the presynaptic terminal. The released neurotransmitter may then move across the synapse to be detected by and bind with receptors in the postsynaptic neuron. The binding of neurotransmitters may influence the postsynaptic neuron in either an inhibitory or excitatory pathway [16,17].
The different functions of acetylcholine, glutamate, GABA, glycine, dopamine, norepinephrine, and serotonin play important and different roles in body physiology. Glutamate is the excitatory neurotransmitter used in the brain and acts as the primary mediator of nervous system plasticity by modifying synapses, which researchers suspect are the memory-storage elements of the brain. Whereas, Gamma-Aminobutyric Acid (GABA) and glycine, serve as the main inhibitory neurotransmitters [16, 17].
Dopamine, plays an essential role in several brain functions, like learning, motor control, reward, emotion, and executive functions, besides implicated in psychiatric and neurological disorders [18]. Serotonin is modulating multiple neuropsychological processes and neural activity, which has implications that affect gastrointestinal processes like bowel motility, bladder control, and cardiovascular function [19].
Epinephrine is a monoamine that is synthesized in the central nervous system, and when the release of norepinephrine in the brain exert effects on a variety of processes, involving stress, sleep, attention, focus, and inflammation. In addition, play important roles in modulating the responses of the autonomic nervous system [20].
Specifically, calcium-evoked exocytosis of the presynaptic vesicles is enabling the release of neurotransmitters into the synapse. The active zones on the presynaptic plasma membranes, bond the neurotransmitter-containing vesicles to the plasma membrane. Once an action potential triggers calcium influx into the presynaptic cleft, the zones undergo fusion with the vesicles, allowing neurotransmitter releasing [21]. Many proteins are involved in the process of fusion between neurotransmittercontaining vesicles and the active zone. The main one is the soluble N-ethyl maleimide Sensitive Factor Attachment Protein Receptors (SNAREs) syntaxin-1, SNAP-25, and others like synaptobrevin-2, which together form a SNARE complex, act as a key component in membrane fusion [21, 22].
Glutamate is including in many neurodegenerative published studies. Glutamate has been excitotoxicity undoubtedly which plays a role in the pathogenesis of Alzheimer's disease and Parkinson's syndrome, and it accelerates the progression of these diseases [23]. Gamma-Aminobutyric Acid (GABA) is a targeted factor in the treatment of anxiety disorder, insomnia, epilepsy [21].
Dopamine plays an essential role in many physiological processes, also it has a role in the pathology of psychiatric and neurodegenerative diseases. Disturbances in the neurotransmission of dopamine are implicated in schizophrenia, psychosis, depression, and attention deficit hyperactivity disorder. There is a degeneration of dopaminergic neurons in the substantia nigra pars compacta is involved in the pathogenesis of these diseases [24].
Serotonin control several neuropsychiatric processes have been implicated in the pathogenesis of depression, when patients exposure to endogenous depression, they have low plasma levels of tryptophan, which is a precursor of serotonin [19]. Norepinephrine is involved in the pathogenesis of neuropsychiatric disorders such as anxiety disorders, mood disorders, attention-deficit hyperactivity disorder, Alzheimer’s disease, and posttraumatic stress disorder [25].
Conclusion
Most of NTs dropping level occurs after receiving cytotoxic drugs, but this stay within the normal range of concentration in the body.
Conflict of Interest
The authors declare that there is no conflict of interest.
References
- http://psychology.about.com/od/nindex/g/neurotransmitter.html
- Corrie PG, Pippa G. Cytotoxic chemotherapy: clinical aspects. Med. 2008;36:24-28.
- Wagner AD, Syn NL, Moehler M, Grothe W, Yong WP, et al. Chemotherapy for advanced gastric cancer. Cochrane Database Syst Rev. 2017;8:CD004064.
- Epstein RJ. Maintenance therapy to suppress micrometastasis: the new challenge for adjuvant cancer treatment. Clinical Cancer Research. 2005;11:5337-5341.
- Wood M, David B. The Royal Marsden Hospital Handbook of cancer chemotherapy: a guide for the multidisciplinary team. 2005.
- Perry MC, Doll DC, Freter CE. Perry's The Chemotherapy Source Book. Lippincott Williams & Wilkins. 2012.
- Rachel A. Cancer chemotherapy. Wiley-Blackwell. 2009.
- Cohen L, de Moor CA, Eisenberg P. Chemotherapy-induced nausea and vomiting: incidence and impact on patient quality of life at community oncology settings. Support Care Cancer. 2007;15:497-503.
- Molassiotis A, Saunders MP, Valle J, Wilson G, Lorigan P, et al. A prospective observational study of chemotherapy-related nausea and vomiting in routine practice in a UK cancer center. Support Care Cancer. 2008;16:201-208.
- Bloechl-Daum B, Deuson RR, Mavros P, Hansen M, Herrstedt. Delayed nausea and vomiting continue to reduce patients’ quality of life after highly and moderately emetogenic chemotherapy despite antiemetic treatment. J Clin Oncol. 2006;24:4472-4478.
- Grunberg SM, Deuson RR, Mavros P, Geling O, Hansen M, et al. Incidence of chemotherapy-induced nausea and emesis after modern antiemetics. Cancer. 2004;100:2261-2268.
- Tina Shih YC, Xu Y, Elting LS. Costs of uncontrolled chemotherapy-induced nausea and vomiting among working-age cancer patients receiving highly or moderately emetogenic chemotherapy. Cancer. 2007;110:678-685.
- Hesketh PJ. Chemotherapy-induced nausea and vomiting. N Engl J Med. 2008;358:2482-2494.
- Endo T, Minami M, Hirafuji M, Ogawa T, Akita K, et al. Neurochemistry and neuropharmacology of emesis the role of serotonin. Toxicology. 2000;153:189-201.
- Tramèr MR, Carroll D, Campbell FA, Reynolds DJ, Moore RA, et al. Cannabinoids for control of chemotherapy-induced nausea and vomiting: a quantitative systematic review. BMJ. 2001;323:16-21.
- NCCN. Clinical Practice Guidelines in Oncology. NCCN GUIDELINES FOR SUPPORTIVE CARE. Adult cancer pain, anti-emesis, cancer- and chemotherapy-induced anemia, cancer-associated venous thromboembolic disease, cancer-related fatigue, distress management, palliative care, prevention, and treatment of cancer-related toxicities. 2019.
- Sapolsky R. Biology and Human Behavior: The neurological origins of individuality. The Teaching Company. 2005.
- Ko JH, Strafella AP. Dopaminergic neurotransmission in the human brain: new lessons from perturbation and imaging. Neuroscientist. 2012;18:149-168.
- Berger M, Gray JA, Roth BL. The expanded biology of serotonin. Annu Rev Med. 2009;60:355-366.
- O'Donnell J, Zeppenfeld D, McConnell E, Pena S, Nedergaard M. Norepinephrine: a neuromodulator that boosts the function of multiple cell types to optimize CNS performance. Neurochem Res. 2012;37:2496-2512.
- Rizo J. Mechanism of neurotransmitter release coming into focus. Protein Sci. 2008;27:1364-1391.
- Südhof TC. Neurotransmitter release: the last millisecond in the life of a synaptic vesicle. Neuron. 2013;80:675-690.
- Dong X, Wang Y, Qin ZH. Molecular mechanisms of excitotoxicity and their relevance to the pathogenesis of neurodegenerative diseases. Acta Pharmacol Sin. 2009;30:379-387.
- Rangel-Barajas C, Coronel I, Florán B. Dopamine Receptors and Neurodegeneration. Aging Dis. 2015;6:349-368.
- Ressler KJ, Nemeroff CB. Role of norepinephrine in the pathophysiology of neuropsychiatric disorders. CNS Spectr. 2001;6:663-666.



