Research Article - Onkologia i Radioterapia ( 2021) Volume 15, Issue 12
2Colleges of Medicine, Baghdad University, Baghdad, Iraq
Noaman Abdul Razzaq Almallah, Baghdad Teaching Hospital, Baghdad Medical City Complex, Ministry of Health/Environment, Baghdad, Iraq, Email: ahmedsalihdr2008@yahoo.com
Received: 26-Oct-2021 Accepted: 02-Dec-2021 Published: 24-Dec-2021
Abstract
Triple-Negative Breast Cancer (TNBC) is a type of breast cancer in which Estrogen Receptor (ER), Progesterone Receptor (PR) and human epidermal growth factor receptor 2 are all negative. Account for 10% to 15%, and it is more common among young and/or black women, and is classically associated with high-grade cancers. The study aimed to describe different clinicopathological parameters and analyse their effect on disease free survival in stage (I, II and III) triple negative breast cancer Iraqi women. A hospital based retrospective analytic study was conducted on 97 triple negative breast cancer Iraqi women. A total 1270 female patients with breast cancer diagnosed at oncology teaching hospital from January the 1st 2014 to December the 31st 2017. About 109 women we identified tumours lacking immunohistochemical expression of ER, PR and HER2 (Triple-Negative Status) only were included in our study. The data collected are demographic data (age, family history of breast cancer, body mass index), and pathological data (tumour histopathological type, lymphovascular invasion, tumour grade, and size, lymph node involvement, distant metastasis (M stage) at time of diagnosis). The median age of 50 years, the body mass index (BMI) mean was 29.8 ± 5.14 kg/m2. The predominant histological type was infiltrative ductal carcinoma (89.9%), grade II was the most common one (52.9%). Lymphovascular invasion was found in 61% of the pathological reports. Adjuvant chemotherapy was given in (89.7%). The median follows up period of our patients (stage I, II and III) was 12 months ranging from (2-46) months. The overall disease free survival mean was (33.2) months and the median was (36) months. Approximately, 20.6% of stage (I, II and III) developed recurrence after diagnosis; with visceral metastasis was the most common one. Tumour size as a risk factor can significantly affect the disease free survival, while patient age, body mass index, lymphovascular invasion, tumour grade and lymph nodes involvement have no statistical significance. Triple negative breast cancer is an aggressive disease with the most of the events (local recurrence, distance metastasis) occur in the first 3 years of follow up, and the most common type was visceral metastasis. Tumour size as a poor prognostic factor significantly shortens the disease free survival, while all the other clinicopathological data have no statistical significance.
Keywords
breast cancer, triple negative, disease free survival, invasive ductal carcinoma
Introduction
Breast cancer is a major public health problem for women throughout the word. In 2017, it was estimated there were 255,180 new cases of breast cancer, with 41,070 deaths [1]. Worldwide, breast cancer is the most frequently diagnosed cancer and the leading cause of cancer death among females, accounting for 25% of cancer cases and 15% of the cancer deaths, although there is a 4-fold variation in mortality rates and over 10- fold variation in incidence rates between highincidence areas such as the United States and Western Europe and low-incidence areas such as Africa and Asia [2]. Breast cancer mortality had been declining, suggesting a benefit from the combination of early detection and more effective treatment [3].
In Iraq, breast cancer is the commonest type of female malignancy, accounting for approximately one-third of the registered female cancers according to the latest Iraqi Cancer Registry/Ministry of Health [4]. A total of 23,792 incident breast cancer cases were registered among females aged ≥15 years, representing 33.8 % of all cancers in females registered during 2000-2009. It ranked first in all the years. The median age at diagnosis was 49 and the mean age was 52 years. The incidence rate of all female breast cancer in Iraq increased from 26.6 per 100,000 in 2000 to 31.5 per 100,000 in 2009. Except for the age group (6069) did not decline since 2003, while the incidence rates in the age group (15-39) which started to decline in 2004 [5]. The total number of new cases of all cancers during the year 2016 was 25,556 with male to female ratio 0.7:1. The top ten cancers in female were breast (34.27%). the highest incidence of cancer in females was breast cancer (26.26/100,000 female’s population) with highest incidence of cancer mortality in female (4.79/100,000 female populations) [6].
Triple-Negative Breast Cancer (TNBC) is a type of breast cancer in which Estrogen Receptor (ER), Progesterone Receptor (PR) and Human Epidermal Growth Factor Receptor 2 (HER2) are all negative. Due to lack of receptor expression, the cancer growth is not fueled by estrogen, progesterone or by HER2, so it is unresponsive to therapies that target these receptors [7]. Unique in its tumour aggression and high rate of recurrence, Triple- Negative Breast Cancer (TNBC) is less likely to be detected by mammogram and has a poorer prognosis than other breast cancer subtypes (non-triple negative breast cancers '' non-TNBC). TNBC accounts for only approximately 10% to 15% of all breast cancers, is more common among young and/ or black women, and is classically associated with high-grade cancers. Unlike other subtypes of breast cancer, prognosis of TNBC does not correlate with tumour size or nodal involvement as closely as other subtype do [7, 8].
Immunohistochemistry (IHC) has rapidly become the predominant method for measuring ER and PR in clinical practice. This method relies upon the recognition of the receptor protein through the use of specific antibodies and it replaces largely the old method, Ligand Binding Assay (LBA, an assay that involves the competitive binding of a radiolabeled steroid ligand to the receptor) in the early 1990's [9]. It can be performed on a variety of samples including Fine Needle Aspirates (FNA), core biopsies, small tumours, and cell blocks from body fluids such as pleural effusions, frozen tissue, or fixed, paraffin-embedded archival tissue. As well as, total receptor protein is measured, (in contrast to the LBA) and it is not affected by endogenous or exogenous steroids or tamoxifen. It is simple to perform and does not require specialized equipment. The Labs followed the ASCO/CAP guideline recommendation (American Society of Clinical Oncology/ College of American Pathologists) in diagnosis [9, 10].
Methods
Study design
A hospital based retrospective analytic study was conducted to evaluate the clinical and pathological criteria and their outcome on disease free survival in stage (I, II and III) triple negative breast cancer Iraqi patients.
Study population
Female patients with breast cancer diagnosed at Oncology Teaching Hospital/Medical city by fine needle aspiration cytology, core needle biopsy, lumpectomy or mastectomy specimen, and then the formalin-fixed paraffin embedded tissue blocks were sent to the hospital laboratory for H&E and IHC. The study included (1270) patients who diagnosed with breast cancer from January the 1st 2014 to December the 31st 2017. Only 109 women based on pathology reports, identified tumours lacking immunohistochemical expression of ER, PR and HER2 (Triple-Negative Status) were enrolled. Patients whose immunohistochemistry reports for ER, PR, or HER2neu were positive or undetermined have been excluded.
Data collection
Primary patient's data were collected during their scheduled visits for their treatment or follow up. Demographic data at time of diagnosis: age, family history of breast cancer, and Body Mass Index (BMI) was calculated by an equation developed by Adolph Quetelet, and categorized according to WHO [11]. Tumour histopathological study was performed in the hospital lab of histopathology, and the data collected: histopathological cellular type, lymphovascular invasion, tumour grade was categorized according to the Scarff-Bloom-Richardson grading system [12], tumour size, and lymph node involvement at time of diagnosis and staged according to American Joint Cancer Committee (AJCC) cancer staging manual, seventh edition (2010). Disease Free Survival (DFS) which is defined as the time passed after diagnosis and until an event happened (locoregional recurrence, lymph node recurrence or distant metastasis) was detected. The discontinuation of follow-up prior to a defining event is called “censoring". We consider the date of surgery is the started time of follow up and Patients with stage IV disease were excluded from the follow up patients. The median follows up period of our patients (stage I, II and III) was 12 months ranging from (246) months.
Statistical analysis
The continuous data are expressed as mean ± SD (standard deviation) while the discrete data are expressed as numbers and percentages. The data are entered on Microsoft Excel sheet version 2013 and was analyzed by Statistical Package for Social Sciences (SPSS) V.22. The descriptive data were presented through a frequency distribution tables. The p value of less 0.05 was considered as statistically significant. The disease free survival was estimated by Kaplan-Meier survival analysis.
Ethical consideration
The thesis proposal was fully discussed and accepted by ethical and scientific committee in Iraqi board of medical specialties. The agreement of health authority in oncology teaching hospital was obtained before the data collection. A verbal consent was taken from all included patients after a full explanation of the aim of the study and insurance of confidentiality of the collected data which will not be used but for research purposes.
Results
In this study, 109 female patients with TNBC, the age distribution were ranged from (27-73) years with a mean and standard deviation of (50.2 ± 9.8 years). 18 out of 109 women had a family history of breast cancer that demonstrate (16.5%). Regarding the Body Mass Index (BMI), the mean was (29.8) ± (5.14) and 50 patients out of 109 women had BMI ≥ 30 (obese) which demonstrate (45.9%) (Table 1).
| Age group (years) | No. (%) |
|---|---|
| = 30 | 3 (2.7) |
| 31-40 | 15 (13.7) |
| 41-50 | 41 (37.6) |
| 51-60 | 32 (29.3) |
| 61-70 | 16 (14.6) |
| 70 and above | 2 (1.8) |
| Family history | No. (%) |
| Positive | 18 (16.5) |
| Negative | 91 (83.5) |
| Body mass index (kg/m2) | No. (%) |
| Under weight (<18.5) | 0 (0) |
| Normal weight (18.5- ?25) | 22 (20.2) |
| Overweight (25 - ?30) | 37 (33.9) |
| Obese (= 30) | 50 (45.9) |
| Total | 109 (100) |
Tab.1. Clinical parameters of triple negative breast cancer
The pathological study showed that the predominant histological type was infiltrative ductal carcinoma (89.9%). According to Scarff Bloom and Richardson histological grading system, grade II (intermediate prognosis) was the most common one (52.9%) followed by grade III (poor prognosis) (38.2%) and grade I (good prognosis) (8.8%). Lymphovascular invasion was found in 61% of the pathological reports (Table 2).
| Tumour pathological subtype | No. (%) |
|---|---|
| Infiltrative ductal carcinoma | 98 (89.9) |
| Infiltrative lobular carcinoma | 8 (7.4) |
| Infiltrative papillary carcinoma | 2 (1.8) |
| Pure type mucinous carcinoma | 1 (0.9) |
| Tumour grade | No. (%) |
| Grade 1 | 9 (8.8) |
| Grade 2 | 54 (52.9) |
| Grade 3 | 39 (38.3) |
| Total | 102 (93.5) |
| Missing | 7 (6.5) |
| Lymphovascular invasion | No. (%) |
| Present | 58 (61) |
| Absent | 37 (39) |
| Total | 95 (87.2) |
| Missing | 14 (12.8) |
Tab. 2. The pathological parameters in TNBC
The tumour size ranged from 1 cm to 9.7 cm (the mean=3.6 cm) and according to the TNM classification, T2 was the most frequent (67.8%). Considering the lymph node involvement status, 61.5% had positive lymph nodes at time of diagnosis; N1 was in 21%, N2 in 26.7% and N3 in 13.7%. Considering the TNM stage, stage 2 was the most common in 43% followed by stage 3 in 40%, stage 4 in 11%, while the lowest was in stage 1 in 5.5%. Considering the status of metastasis, metastases was detected in 12 (11.0%) of the patients, 6 (5.5%) had visceral metastasis (2 patient’s lung metastasis, 2 patient’s liver metastasis and 2 patients with brain metastasis), 5(4.5%) had visceral and bone metastasis, and only 1 case (0.9%) had a pure bone metastasis (Table 3).
| T stage | No. (%) | N stage | No. (%) |
|---|---|---|---|
| T1 | 13 (11.9) | N0 | 42 (38.5) |
| T2 | 74 (67.9) | N1 | 23 (21.1) |
| T3 | 15 (13.7) | N2 | 29 (26.7) |
| T4 | 7 (6.4) | N3 | 15 (13.7) |
| Total | 109 (100) | Total | 109 (100) |
| TNM Stage | No. (%) | Metastasis | No. (%) |
| 1 | 6 (5.5) | Visceral | 6 (50) |
| 2 | 47 (43.1) | Bone | 1 (8.3) |
| 3 | 44 (40.4) | Visceral + bone | 5 (41.7) |
| 4 | 12 (11) | Total | 12 (100) |
| Total | 109 (100) |
Tab. 3. TNM staging results in TNBC
The most common type of surgery performed for resectable disease was modified radical mastectomy and axillary axillary lymph node dissections for 78 (80.4%). Adjuvant chemotherapy was used in 87 patients (89.7%) (anthracyclins based protocol followed by taxane in 83, and non anthracycline based regimen in 4). Neoadjuvant chemotherapy was given to 7 patients (7.2%), (3 patients received platinum based protocol). And 61 patients (62.9%) received adjuvant radiotherapy (Table 4).
| Treatment modality | No. (%) |
|---|---|
| Lumpectomy + axillary clearance | 19 (19.6) |
| Modified radical mastectomy +axillary clearance | 78 (80.4) |
| Adjuvant chemotherapy | 87 (89.7) |
| Neoadjuvant chemotherapy | 7 (7.2) |
| Radiotherapy | 61 (62.9) |
| Total | 97 (100%) |
Tab. 4. Early treatment modalities in TNBC patients
20(20.6%) of 97 patients developed recurrence after diagnosis, 7 (35%) develop locoregional recurrence, 5 (71.4%) of them within the first year of follow up. 8(40%) develop visceral metastasis, 4 (50%) of them at the 1st year of follow up. 5 (25%) develop bone metastasis, 1 (20%) of them at the 1st year of follow up (Table 5 and Figure 1).
| Progression | 1st year | 2nd year | 3rd year | 4th year | No. |
|---|---|---|---|---|---|
| Locoregional recurrence | 5 (50%) | 1 (20%) | 1 (20%) | 0 (0%) | 7 (35%) |
| Visceral metastasis | 4 (40%) | 1 (20%) | 3 (60%) | 0 (0%) | 8 (40%) |
| Bone metastasis | 1 (10%) | 3 (60%) | 1 (20%) | 0 (0%) | 5 (25%) |
| Total | 10 (50%) | 5 (25%) | 5 (25%) | 0 (0%) | 20 (100%) |
Tab. 5. The type of recurrence in the 1st fourth years of follow up
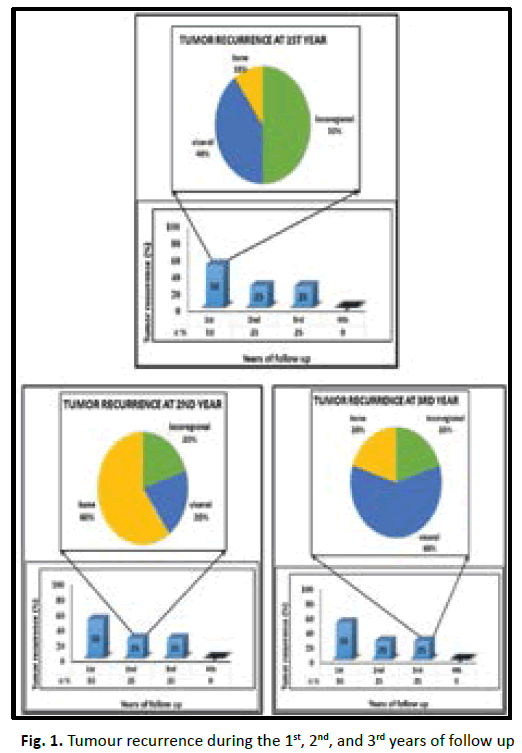
Figure 1: Tumour recurrence during the 1st, 2nd, and 3rd years of follow up
The disease free survival mean of all patients was (33.2) months and the median was (36) months. The figure 2 revealed the Disease-Free Survival (DFS) curve in TNBC patients included in our study (Figure 2).
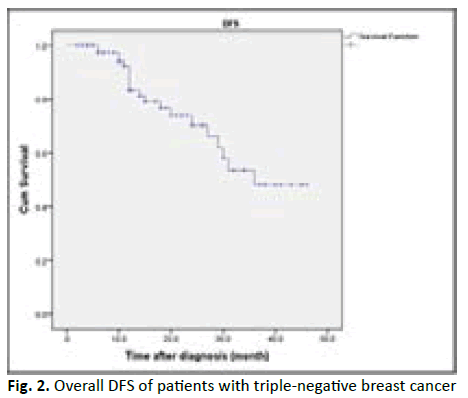
Figure 2: Overall DFS of patients with triple-negative breast cancer
In our study we found the patient who are equal or less than 50 years old have shorter DFS with high incidence of relapse in comparison to more than 50 years old in statistically insignificant (p value 0.19). Obese patients associated with better prognosis and longer DFS in TNBC patients but statistically insignificant (p value 0.53). Lymphovascular invasion associated with poor prognosis and early recurrence in TNBC patients, but statistically insignificant (p value 0.28). Higher tumour grade (grade II and III) associated with poor prognosis and shorter disease free survival, in comparison to low tumour grade (grade I) with no statistical significant (p value 0.47). In our studied patients we found increasing tumour size (T stage) associated with poor prognosis, early recurrence with shorter DFS in TNBC, with being T3 and T4 worst outcome than T1 and T2 with p value < 0.001. In our studied patients we found increasing number of lymph nodes invasion associated with shorter DFS and early recurrence in TNBC patients with (p value 0.54) (Table 6) (Figures 3-8).
| Clinical parameters | Total | Event (%) | Mean DFS (Month) | Chi-Square | p-value |
|---|---|---|---|---|---|
| Age group | 1.79 | 0.19 | |||
| ≤ 50 yr | 55 | 15 (27) | 30.15 | ||
| ˃ 50 yr | 42 | 5 (12) | 36.04 | ||
| Total | 97 | 20 (20) | |||
| BMI | 1.25 | 0.53 | |||
| Normal | 20 | 6 (30) | 30.3 | ||
| Overweight | 33 | 7 (21) | 30.4 | ||
| Obese | 44 | 7 (16) | 34 | ||
| Total | 97 | 20 (21) | |||
| Lymphovascular invasion | 1.16 | 0.28 | |||
| Present | 51 | 12 (23) | 31.7 | ||
| Absent | 36 | 5 (14) | 36.5 | ||
| Total | 87 | 17 (19) | |||
| Tumour grade | 1.48 | 0.47 | |||
| Grad I | 9 | 1 (11) | 38.9 | ||
| Grade II | 49 | 10 (20) | 31.5 | ||
| Grade III | 33 | 8 (24) | 31.8 | ||
| Total | 91 | 19 (20) | |||
| T stage | 51.5 | <0.001 | |||
| T1 | 12 | 3 (25) | 25.1 | ||
| T2 | 66 | 9 (13) | 37.9 | ||
| T3 | 14 | 6 (42) | 18.4 | ||
| T4 | 5 | 2 (40) | 6.5 | ||
| Total | 97 | 20 (20) | |||
| N stage | 2.13 | 0.54 | |||
| N0 | 37 | 4 (10) | 37.5 | ||
| N1 | 23 | 5 (21) | 34 | ||
| N2 | 24 | 7 (29) | 27.3 | ||
| N3 | 13 | 4 (30) | 28.3 | ||
| Total | 97 | 20 (20) | |||
Tab. 6. DFS according to lymphovascular invasion in TNB
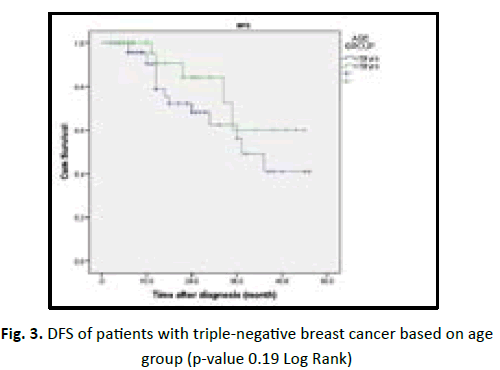
Figure 3: DFS of patients with triple-negative breast cancer based on age group (p-value 0.19 Log Rank)
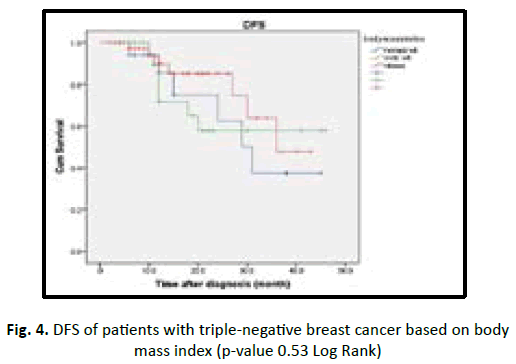
Figure 4: DFS of patients with triple-negative breast cancer based on body mass index (p-value 0.53 Log Rank)
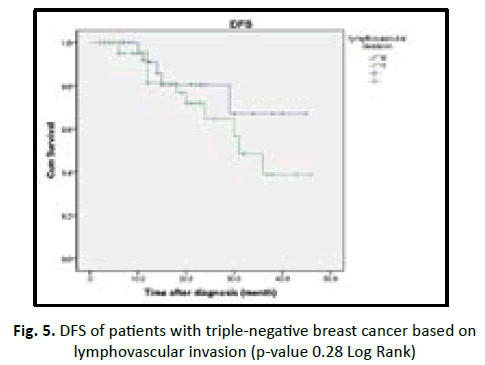
Figure 5: DFS of patients with triple-negative breast cancer based on lymphovascular invasion (p-value 0.28 Log Rank)
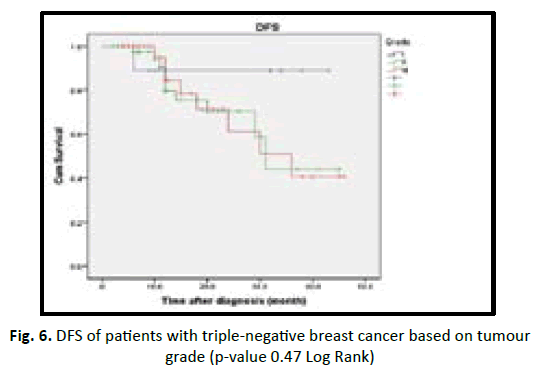
Figure 6: DFS of patients with triple-negative breast cancer based on tumour grade (p-value 0.47 Log Rank)
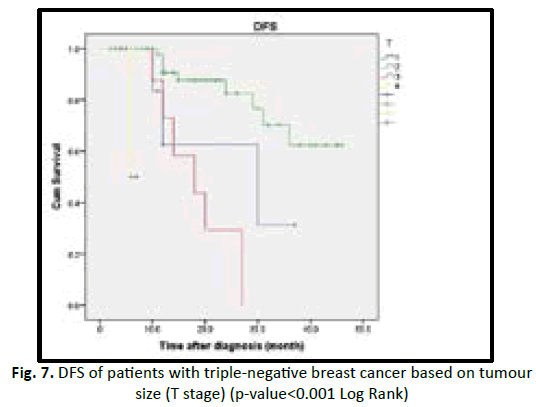
Figure 7: DFS of patients with triple-negative breast cancer based on tu m our size (T stage) (p-value<0.001 Log Rank)
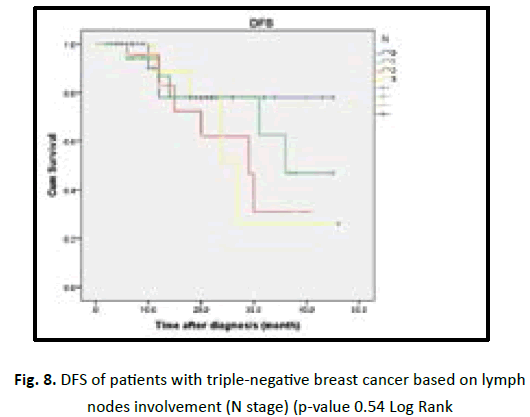
Figure 8: DFS of patients with triple-negative breast cancer based on lymph nodes involvement (N stage) (p-value 0.54 Log Rank
Discussion
In present study the age of diagnosis ranged (27-73) with a median of 50 years. Which is comparable to 50.4 years in population studied by Qiu et al [13] and Pistelli et al [14] found the median age was 54 years; range (26-83) years. A positive family history of breast cancer in about 16.5% of the patients. which is slightly more than 11.49% found by Qiu et al [13], but much more than 6.82% by Souad et al [15] and 10% studies performed in Lebanon [16, 17]. Regarding the Body Mass Index (BMI) in our study the mean was (29.85 ± 5.14) kg/m2. which is close to (30 ± 0.07) kg/m2 found by Vona Davis et al [18] in this retrospective study on patients with TNBC.
We found most of the patient are obese 45.9% and overweight 33.9% while the normal weight was 20.2% as in the Carolina breast cancer study, population-based, case-control study of American African and white women, reported that obesity (defined as a BMI ≥ 30) was associated with an increased risk of TNBC [19].
Regarding the pathological parameters, in our study we found the predominant histological type was infiltrative ductal carcinoma (89.9%) and infiltrating lobular carcinoma represented (7.4%), compared to Souad et al., [15] who found infiltrating ductal carcinoma in (96.08%) of cases, and infiltrating lobular carcinoma in (2.94%). High histological grade (grade 3) is an important poor prognostic factor. In our study we found the grade II in (52.9%), and grade III in (38.3%). A wide range of variation in the percentage of high grade histopathology was found among different studies, Fayaz et al who found high grade (grade III) in 57% of TNBC patients [20] and in Singapore, they documented a higher rate of high grade III (77%) [21]. While Sun et al., (2018) found the grade III in (19.4%) only [22] suggest a different biological behavior of TNBC among different races. The Lymph vascular invasion (LVI) was detected in 61% of patients in our study, a finding more than 39.6% that found by Rebecca Dent et al [23] and in 48.5% found by Souad et al [15].
In our study, the average tumour size was large (3.6 cm) and T2 was the predominant in 67.9% which is near to Souad et al., [15] who found average tumour size (4.4 cm) and T2 in 47.5%. While Pistelli M et al most of the patients presented pT1 tumours (53.7%) and pT2 tumours (30.9%) [14]. Suggesting a rapidly growing tumour with short time till mammographically been detected. The lymph node involvement is found in 61.5% of our studied patients, which is near the results of Souad et al [15] who found lymph nodes involvement in 69% but more than Pistelli et al who found lymph nodes were disease-positive in 38.2% of cases [14].
In our study we found (11%) of patients has metastasis at time of diagnosis. About 91.6% of metastatic patients were visceral type which is near the results at Agarwal et al who found metastasis in 15.5% with visceral type in 93.5% [24] and in Tseng et al., who found metastasis in 12.2% with 77.1% visceral type [25] suggesting visceral metastasis is the most common type in TNBC patients than bone metastasis. During the follow up of 97 patients (stage I, II and III) we found 20(20.6%) develop a recurrence after certain time from diagnosis. this was within the first year in 10(50%) patients and within the second year in 5(25%) patients and within the third year in 5(25%) patients. the most common event was visceral metastasis which occurred in (40%) followed by loco regional recurrence in 7(35%) and bone metastasis in 5(25%). These results are consistent with the results of Bauer KR et al. which have shown that TNBC is most likely to recur after treatment [7]. These risks appear to be more during the first few years after treatment. For example, a study of more than 1,600 women in Canada published in 2007 found that women with TNBC were at risk of having cancer recurrence outside breast, but only for the first three years [26]. Rakha et al. reported that in a study of 1944 breast cancer patients, local relapse and metastases occurred earlier in TNBC patients [27].
The rate of visceral metastases was higher than that of bone metastasis. In addition, Lin et al. analysed the sites of distant recurrence in 116 metastatic TNBC patients and reported that the majority of metastases were in lungs and liver (visceral metastasis) [28], which is consistent with our results.
The overall Disease Free Survival (DFS) in our study we found the median was 36 months. And all the clinical and pathological parameters showed that insignificant risk factors for reduced DFS except for the tumour size (T stage) that found higher tumour size will shorten the disease free survival and was statistically significant (p value <0.001, Log Rank test). Other studies like Qiu et al. found significant inverted relationship between DFS and age, the maximum diameter of the tumour, lymph node status, clinical stage, histological grade and pathological types [13]. While Dent et al. who conducted a long-term follow-up of 1608 breast cancer patients and found that the recurrence of TNBC had no correlation with the tumour size and the recurrence mainly occurred in the first 5 years after diagnosis, especially within the 1st 3 years after diagnosis [23].
Conclusion
Highest incidence of triple negative breast cancer is seen in the 5th and 6th decade of life and lowest in the 8th and above. Most TNBC associated with obese patients. Tumour grade II was the most common. During the follow up of stage (I, II and III). The most of the events occur within the first 3 years of follow up, and visceral metastasis was the most common type. Tumour size as a poor prognostic factor significantly shortens the disease free survival, while all the other clinicopathological data have no statistical significancee.
References
- American Cancer Society. Cancer Facts and Figures. 2017.
- Torre LA, BrayF, Siegel RL, Ferlay J, Jemal A, et al. Global cancer statistics, 2012. CA Cancer J Clin. 2015;65:87-108.
- Mohammad H. Forouzanfar KJF, Allyne M. Delossantos, Rafael Lozano, et al. Breast & cervical cancer in 187 countries between 1980 & 2010: a systematic analysis. Lancet. 2011;378:1461-1484.
- Hussain AM, Lafta RK. Cancer Trends in Iraq 2000-2016. Oman Med J. 2021;36:e219.
- Al-Hashimi MM, Wang XJ. Breast cancer in Iraq, incidence trends from 2000-2009. Asian Pac J Cancer Prev. 2014;15:281-286.
- Iraqi Cancer Board. Results of the Iraqi Cancer Registry annual reports 2016. Baghdad, Iraqi Cancer Registry Center, Ministry of Health\Environment, 2016.
- Bauer KR, Brown M, Cress RD, Parise C, Caggiano V. Descriptive analysis of estrogen receptor (ER)-negative, progesterone receptor (PR)-negative, and HER2negative invasive breast cancer, the so-called triple-negative phenotype: a population-based study from the California cancer Registry. Cancer. 2007;109:1721-1728.
- Hernandez-Aya LF, Chavez-Macgregor M, Lei X, Valero V, Hsu L, et al. Nodal status and clinical outcomes in a large cohort of patients with triple-negative breast cancer. J Clin Oncol. 2011;29:2628-2634.
- Elizabeth M, Hammond H, et al. Hormone receptors in breast cancer: Clinical utility and guideline recommendations to improve test accuracy. 2021.
- Hammond ME, Hayes DF, Dowsett M, Wolff AC, Wheeler T, et al. American Society of Clinical Oncology/College of American Pathologists guideline recommendations for immunohistochemical testing of estrogen and progesterone receptors in breast cancer. J Clin Oncol 2010;28:2784-2795.
- McGee DL, Diverse Populations Collaboration. Body mass index and mortality: a meta-analysis based on person-level data from twenty-six observational studies. Ann Epidemiol. 2005;15:87-97.
- Henry F, Wolber RA, Berean KW, Franquemont DW, Gaffey MJ, et al. Interobserver Reproducibility of the Nottingham Modification of the Bloom and Richardson Histologic Grading Scheme for Infiltrating Ductal Carcinoma. Am J Clin Pathol. 1995;103:195-198.
- Qiu J, Xue X, Hu C, Xu H, Kou D, et al. Comparison of clinicopathological features and prognosis in triple-negative and non-triple negative breast cancer. J Cancer. 2016;7:167-173.
- Pistelli M, Pagliacci A, Battelli N, Santinelli A, Biscotti T, et al. Prognostic factors in early-stage triple-negative breast cancer: lessons and limits from clinical practice. Anticancer Res. 2013;33:2737-2742.
- Souad H, Zahia F, Abdelhak L, Karima S, Dalila S, et al. Descriptive study of triple negative breast cancer in Eastern Algeria. Pan Afr Med J. 2018;29:45.
- Ghosn M, Hajj C, Kattan J, Farhat F, Karak F, et al. Triple-Negative breast cancer in Lebanon: A case series. Oncologist 2011;16:1552-1556.
- Fakhoury W. Prognostic factor in breast cancer; correlation with other factors; estrogen and progesterone receptors, MIB and histoprognostic grade. Saint-Joseph University 1997;1:1-50.
- Vona-Davis L, Rose DP. Adipokines as endocrine, paracrine, and autocrine factors in breast cancer risk and program. Endocr Relat cancer 2007;14:189-206.
- Millikan RC, Newman B, Tse CK, Moorman P, Conway K, et al. Epidemiology of basal-like breast cancer. Breast Cancer Res Treat 2008;109:123-139.
- Fayaz MS, El-Sherify MS, El-Basmy A, Zlouf SA, Nazmy N, et al. Clinicopathological features and prognosis of triple negative breast cancer in Kuwait: A comparative/perspective analysis. Rep Pract Oncol Radiother 2014;19:173-181.
- Thike AA, Cheok PY, Jara-Lazaro AR, Tan B, Tan P, et al. Triple-negative breast cancer: Clinicopathological characteristics and relationship with basal-like breast cancer. Mod Pathol 2010;23:123-133.
- Sun L, Zhu Y, Qian Q, Tang L. Body mass index and prognosis of breast cancer: An analysis by menstruation status when breast cancer diagnosis. Medicine 2018;97:e11220.
- Dent R, Trudeau M, Pritchard K, Hanna W, Kahn HK, et al. Triple-Negative Breast Cancer: Clinical Features and Patterns of Recurrence. Clin Cancer Res 2007;13:4429-4434.
- Agarwal G, Nanda G, Lal P, Mishra A, Agarwal A, et al. Outcomes of Triple- Negative Breast Cancers (TNBC) compared with Non-TNBC: Does the survival vary for all stages? World J Surg 2016;40:1362-1372
- Tseng LM, Hsu NC, Chen SC, Lu YS, Lin CH, et al. Distant metastasis in triple-negative breast cancer. Neoplasma 2013;60:290-294.
- Dent R, Hanna WM, Trudeau M, Rawlinson E, Sun P, et al. Pattern of metastatic spread in triple-negative breast cancer. Breast Cancer Res Treat 2009;115:423-428.
- Rakha EA, EI-Sayed ME, Green AR. Prognostic markers in triple negative breast cancer. Cancer 2007;109:25-32.
- Lin NU, Claus E, Sohl J, Razzak A, Arnaout A, et al. Sites of distant recurrence and clinical outcomes in patients with metastatic triple-negative breast cancer: High incidence of central nervous system metastases. Cancer 2008;113:1638-1645.



