Case Report - Onkologia i Radioterapia ( 2023) Volume 17, Issue 6
Synchronous cervical esophageal squamous cell carcinoma esophagus and GEJ adenocarcinoma in a post allogeneic transplant in acute lymphoblastic leukemia patient: Complex management issue of a very rare presentation
Ahmad Al Ghoche1*, Abdullah Al Rawahi2, Rami Abu Omar3, Asma Naz Nadaf4, Anjali Jain5, Sercan Yilamz6 and Mansour Al-Moundhri72Surgery department, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
3Medical Oncology department, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
4Pathology department, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
5Radiology department, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
6Radiotherapy department, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
7Professor of Medicine and medical Oncology, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman
Ahmad Al Ghoche, Medical Oncologist, Sultan Qaboos Comprehensive Cancer Care And Research Center, Muscat, Seeb | 123, Oman, Email: ahmadghauche@gmail.com
Received: 12-Apr-2023, Manuscript No. OAR-23-95260; Accepted: 01-Jun-2023, Pre QC No. OAR-23-95260 (PQ); Editor assigned: 20-Apr-2023, Pre QC No. OAR-23-95260 (PQ); Reviewed: 15-May-2023, QC No. OAR-23-95260 (Q); Revised: 28-May-2023, Manuscript No. OAR-23-95260 (R); Published: 03-Jun-2023
Abstract
The histological subtypes of Esophageal Adenocarcinoma (EAC) and Esophageal Squamous Cell Carcinoma (ESCC) continue to be fatal diseases throughout the world. Lymphoblastic Leukemia (ALL) is a heterogeneous hematologic disease characterized by the proliferation of immature lymphoid cells in the bone marrow, peripheral blood, and other organs. Bone marrow transplantation offers a curative therapy in some patients with malignant and nonmalignant diseases. However, one important late complication in transplant survivors is the development of secondary malignancies including solid tumors; among these, only few cases of EC have thus been reported. Hereby we describe a case of a 32 year old male patient who developed synchronous adeno- and squamous esophageal carcinomas of gastroesophageal junction and cervical esophagus respectively, in a known case of transplanted relapsed acute lymphoblastic leukemia in childhood. The presentation represented a management challenge in terms of employing and sequencing of treatment modalities in treatment of both esophageal carcinomas. Patient was treated with neoadjuvant FLOT4 followed by surgery then concurrent chemoradiotherapy and ended with 4 cycles of adjuvant FLOT4. To our knowledge, this is the first reported case with 2 synchronous esophageal carcinomas having two different pathologies in a cured case of relapsed ALL. As in the case presented here, prior chemotherapy, severe graft versus host disease, and other confounding variables leading to the development of secondary disease may alter the usual activity of BMDC as a possible tumor development mechanism.
Keywords
histological, esophageal, adenocarcinoma
Introduction
The histological subtypes of Esophageal Adenocarcinoma (EAC) and Esophageal Squamous Cell Carcinoma (ESCC) continue to be fatal diseases throughout the world[1]. Esophageal Cancer (EC) is usually rare in the young population. The synchronous occurrence of both subtypes is extremely rare and has been linked to genetic predisposition, immunocompromised state, or secondary malignancy due to previous cancer treatment.
Lymphoblastic Leukemia is a theterogeneous hematologic disease characterized by the proliferation of immature lymphoid cells in the bone marrow, peripheral blood, and other organs [2, 3]. Bone marrow transplantation offers a curative therapy in some patients with malignant and nonmalignant diseases. However, one important late complication in transplant survivors is the development of secondary malignancies including solid tumors; among these, only few cases of EC have thus been reported. Hereby we describe a case of a 32 yo male patient who developed synchronous adeno- and squamous esophageal carcinomas of gastroesophageal junction and cervical esophagus respectively, in a known case of transplanted relapsed acute lymphoblastic leukemia in childhood. The presentation represented a management challenge in terms of employing and sequencing of treatment modalities in treatment of both esophageal carcinomas.
Case Summary
Our patient was a 32 year old male who was diagnosed at the age of 6 years with pre-B ALL associated with CNS involvement. He was initially treated by UK ALL protocol with radiotherapy in India followed by maintenance chemotherapy. He relapsed 2 years later and was treated with modified ALL BFM protocol after which he underwent HLA matched salvage Allogeneic bone marrow transplant at Sultan Qaboos University Hospital (SQUH). He remained under follow up until June 2021 when he presented to Sultan Qaboos Comprehensive Cancer Care and Research Center (SQCCCRC) complaining of severe fatigue and black stool. Prior to presentation to our institution, he was found to have severe anemia at an outside hospital in Muscat, Oman. Gastroscopy was done there and showed a cauliflower malignant looking mass occupying the lumen near the Gastro-Esophageal Junction (GEJ) extending up 6 cm in length and protruding down to the fundus of the stomach. Biopsy revealed an invasive moderately differentiated adenocarcinoma. FDG PET scan was done on 16/6/2021 at our institution and showed an intense focal abnormal uptake in the esophagus region at the level of C6-C7 with an SUV max of 12 (Figure1).
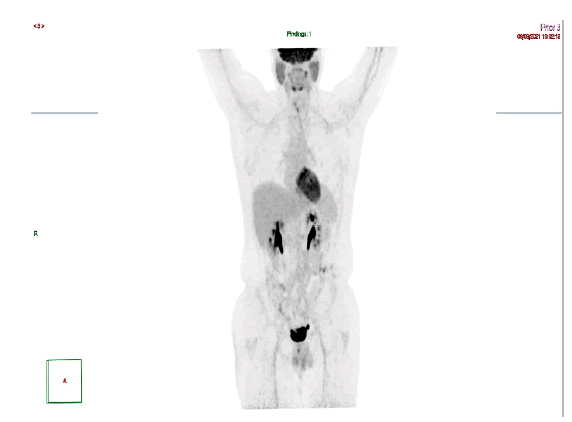
Figure 1:PET at diagnosis
It also showed an intense abnormal uptake of a gastroesophageal mass with SUV max 16 associated with an avid left perigastric lymph node (1.4x1 cm) with SUV max of 4.4. Also, a mild inhomogeneous FDG uptake was seen in the left thyroid lobe which was found to be non-malignant by FNA. MRI NECK on 20/06/2021 showed a circumferential thickening of the upper cervical esophagus with circumferential enhancement on post contrast sequences which is in correlation with the PET CT examination, with no suspicious lymph nodes seen. On the 20th of June 2021, patient had a second gastroscopy at SQCCCRC and biopsies from the upper and lower esophageal regions were taken and revealed 2 different lesions: a 1 cm polypoidal mass lesion in the upper esophagus, 18 cm from incisors just below the upper esophageal sphincter; at 35 cm from the incisors, a distal esophageal mass extending into the stomach as a large polyp like mass, non-obstructing, with no active bleeding. Biopsies for the 2 suspected lesions came as follows: moderately differentiated adenocarcinoma (Figure 2), intact MMR protein expression (MMR- proficient) (Figure 3), positive PD-L1 immune reactivity (CPS = 20%) (Figure 4), negative HER2 (Figure 5) immune reactivity in the lower GEJ mass; and a moderately differentiated squamous cell carcinoma (Figure 6) in the upper esophageal mass. EUS (endoscopic ultrasound) was done also and showed a T2 lesion (with loss of differentiation of the wall layers suggestive of infiltration of the wall in the upper esophagus with no regional adenopathies and suspicious lymph nodes in the lower esophagus. The peritoneum and the bowel were examined and were healthy during feeding jejunostomy insertion on 23rd of June 2021, with a small suspicious liver nodule found in segment 4b which was confirmed as negative by biopsy and reviewed as a hemangioma on a dedicated contrast enhanced liver MRI. A genetic analysis was requested for our patient (right leg skin biopsy) (Figures 2-6).
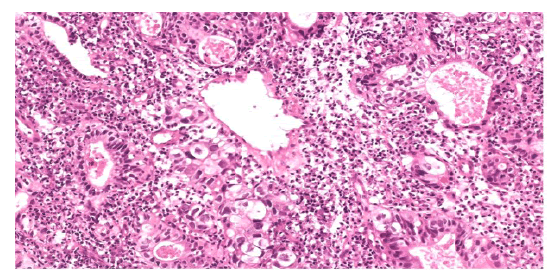
Figure 2:H&E,x100, Adenocarcinoma component with malignant glands
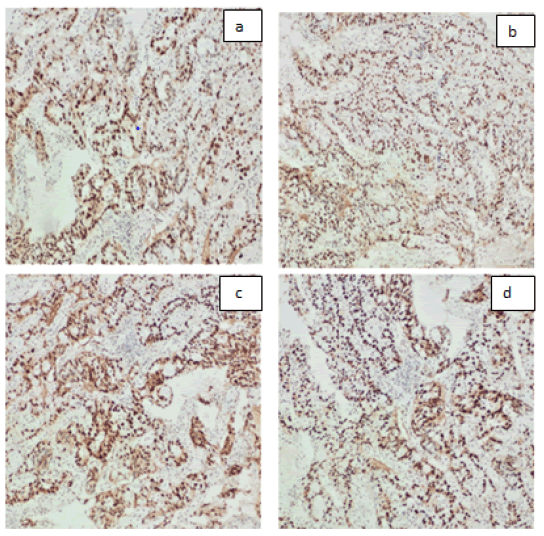
Figure 3:MMR proteins shows intact expression: a. MLH1 b. PMS2 c. MSH2 d. MSH
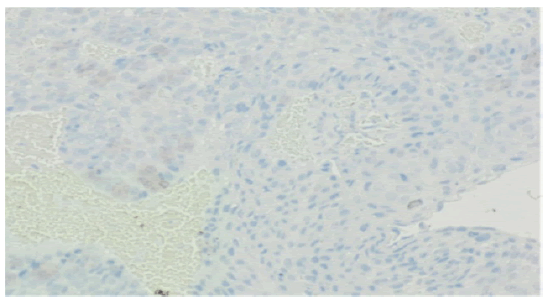
Figure 4: PDL1 Negative (No staining in tumor or immune cells )
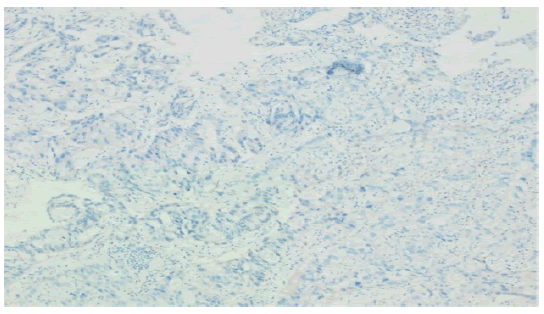
Figure 5:Her2neu: negative (score 0) in malignant adenocarcinoma
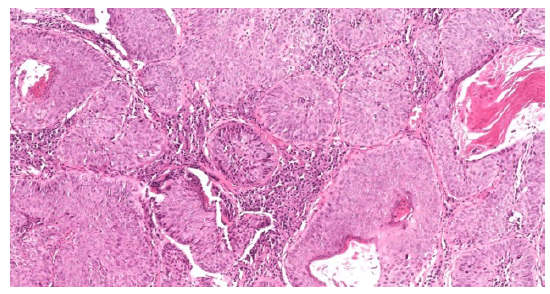
Figure 6: H&E, 10x, Neoplastic squamous cells with abundant eosinophilic cytoplasm
Patient’s case was discussed on several occasions in our center’s MDT and the plan was to give perioperative therapy tackling both lesions according to guidelines and the corresponding pathologies. The patient was treated with FLOT4 chemotherapy protocol in the neoadjuvant setting, which lead to a partial response in the lower esophageal mass. Hybrid Ivor Lewis Esophagectomy was then done, and pathology result showed R1 resection (positive margins); he then received concurrent chemo-radiotherapy involving both upper squamous esophageal lesion and the upper limit (positive margins) of the lower esophageal malignancy (Figure 7).
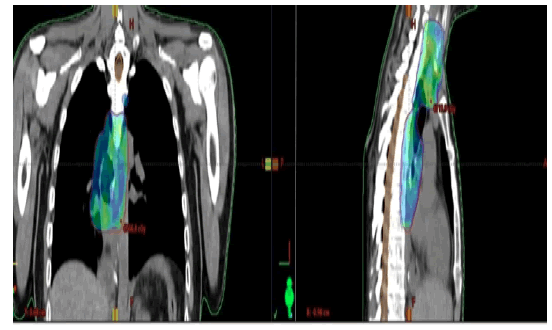
Figure 7: Radiation field
PET scan at the end of planned radiotherapy showed complete metabolic response. Patient then finished 4 cycles of adjuvant FLOT4 protocol to complete his fully planned peri-op treatment for the squamous and adenocarcinoma parts. This treatment course was complicated by recurrent neutropenia, mouth ulcers and weight loss. End of protocol PET scan (Figure 8) and a follow up scan after 4 months (Figure 9) were negative for malignancy. Latest evaluation in December 2022 was negative for recurrence by PET scan and gastroscopy with random biopsies.

Figure 8: End Of Treatment Pet
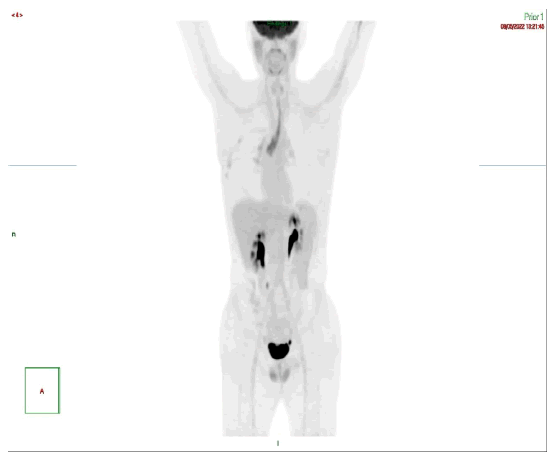
Figure 9:Follow up pet
Discussion
ALL is a rare cancer, accounting for less than 1 % of all hematologic malignancies, more predominant in females with a global lifetime incidence of less than 1 in 1000. The disease has 2 peaks of age: the first is below 5 years of age and the less frequent one above 50 [4].
Secondary malignancy post-transplant, hematologic or solid, is a lifetime problem with no peak incidence nor an age cut off after transplant. Most of these cancers were deemed related in part to the chemotherapy or to the conditioning regimen used to treat the primary tumor. While total body irradiation has been linked to development of adenocarcinoma, chronic GVHD was associated with squamous cell carcinoma [5]. Our case had both synchronically, suggesting a synergic role of multiple factors in the development of the 2 different malignancies. The development of esophageal squamous cell carcinoma may be driven by multiple events occurring during the onset or treatment course of primary hematologic disorders [6].
Furthermore, secondary malignancies can occur in some cases very late being the cause of mortality or morbidity in such patients suggesting the need for long term monitoring and cancer prevention in such category of patients [7]. Triple secondary malignancy post HLA-matched sibling donor was reported in a 31 year old male patient treated for cutaneous lymphoma. However, these 3 lesions were metachronous and no different pathologies were found at same time: he developed mandibular moderately differentiated SCC 5 years after the BMT, a well differentiated SCC of the palate one year later, concurrently with a poorly differentiated ESCC treated with subtotal esophagectomy. One year later, his disease recurred, and he was treated with radiation and chemotherapy. He remained after that free of all his cancers. Different etiologies could be behind the increased risk of secondary malignancy post allogeneic transplant in such case, including but not limited to the chemotherapy long term side effects, the chronic GVHD and the use of immunosuppressants as well as the irradiation received before transplant or all together [8]. A pathophysiologic effect of Bone Marrow Derived Cells (BMDC) post-transplant was suggested in a male transplanted by female marrow, where these donor cells were traced and 2 different mechanisms were postulated: either the BMDC lead to secondary cancer by providing cancer associated fibroblast, or it served as a niche for the myofibroblast, and endothelium required to the development of the Barret’s adenocarcinoma. This contribution of BMDC can be of effect even in very small cell numbers recruited to a given tumor developing at later stage. The effect of BMDC was shown in a mouse transplanted with bone marrow expressing beta galactosidase, followed by surgical esophagojejunostomy in a way to track BMDCs, where it went to the surgical anastomosis and resulted in Barrett's metaplasia.
However, several points to be taken in consideration: the effect of BMDC will take long time to appear as secondary tumor might take years to appear after the initial malignancy. Different patient related factors such as sex difference, immunity status, cell types, primary tumor and immune status prior to transplant could play a role in the pathogenesis and timing of development of secondary disease [9].
Another study evaluating secondary solid tumors after allogeneic HSCT from Japan showed that the risk for developing tumors was 2.16-fold higher than that of the age- and sex-adjusted general population, with a median from transplant to secondary malignancy of 5.6 years along with a 2.4% 10-year cumulative risk.
The risk was significantly higher for tumors of the skin, oral cavity and esophagus with no increase in gastric, colon or lung cancer, despite being the most prevalent neoplasm in the Japanese, was observed.
The secondary tumor development was found in a multivariate analysis to be mostly correlated with high grade lymphoma and the development of chronic GVHD [10]. A recently published study in the Journal of Cancer Research and Clinical Oncology evaluated secondary esophageal squamous cell carcinoma after allogeneic Hematopoietic Stem Cell Transplantation (alloHSCT): it was found that the irradiation received prior to transplant and the transplant itself were higher in ESCC group as well as high risk secondary ESCC patients with a mean time to secondary malignancy detection of 82.3 months. In this study, lesions were in the cervical to upper thoracic esophagus in approximately 60% of all secondary ESCC cases after allo-HSCT.
The sequencing of treatment of different histological subtypes located at distinct locations in the esophagus in the context of treated ALL paused a significant difficulty. Although, there is no standard of care in such case we opted to utilize neoadjuvant FLOT4 chemotherapy with response in both lesions followed by surgical approach to GE junction lesion with total gastrectomy, followed by chemoradiotherapy to cervical esophagus lesion, followed by 4 cycles of FLOT4. The treatment was complicated by recurrent neutropenia and required nutritional support. The patient completed his treatment and is currently in clinical remission.
Conclusion
To our knowledge, this is the first reported case with 2 synchronous esophageal carcinomas having two different pathologies in a cured case of relapsed all. As in the case presented here, prior chemotherapy, severe graft versus host disease, and other confounding variables leading to development of secondary disease. may alter the usual activity of BMDC as a possible tumor development mechanism. Shall screening for this category of patient should be required and standard practice is to be investigated in randomized trial.
References
- Kato F, Daiko H, Kanamori J, Inamoto Y, Fukuda T, et al. Esophagectomy for the patients with squamous cell carcinoma of the esophagus after allogeneic hematopoietic stem cell transplantation. Int J Clin Oncol. 2020;25:82-88.[Google Scholar]
- Jabbour EJ, Faderl S, Kantarjian HM. Adult acute lymphoblastic leukemia. In Mayo Clin Proc. 2005; 80:1517-1527. Elsevier.[Google Scholar]
- Zakir Ullah M, Zheng Y, Song J, Aslam S, Xu C. et al . An attention-based convolutional neural network for acute lymphoblastic leukemia classification. Appl Sci. 2021;11:10662.[Google Scholar]
- Danylesko I, Shimoni A. Second malignancies after hematopoietic stem cell transplantation. Curr treat options oncol.,2018;19:1-7. [Google Scholar]
- Miyawaki Y, Imoto I, Tokairin Y, Kawada K, Nakajima Y, et al Esophageal squamous cell carcinoma developed 11 years after allogeneic bone marrow transplantation for acute lymphatic leukemia. Jpn J Clin Oncol. 2013 ;43:69-73.[Google Scholar]
- Robin M, De Wreede LC, Wolschke C, Schetelig J, Eikema DJ, et. al. Long-term outcome after allogeneic hematopoietic cell transplantation for myelofibrosis. haematologica. 2019;104:1782. .[Google Scholar]
- Koharazawa H, Yamaji S, Takasaki H, Takabayashi M, Fujimaki K, et. al. Triple secondary malignancy of gingiva, palate and esophagus after an allogeneic bone marrow transplantation for cutaneous T-cell lymphoma. [Rinsho Ketsueki] Jpn J Clin Hematol. 2005;46:496-500. [Google Scholar][Cross Ref]
- Hutchinson L, Stenstrom B, Chen D, Piperdi B, Levey S, et. al. Human Barrett's adenocarcinoma of the esophagus, associated myofibroblasts, and endothelium can arise from bone marrow-derived cells after allogeneic stem cell transplant. Stem cells dev. 2011;20:11-7. [Google Scholar]
- Yokota A, Ozawa S, Masanori T, Akiyama H, Ohshima K, et al Secondary solid tumors after allogeneic hematopoietic SCT in Japan. Bone marrow transplant. 2012;47:95-100.[Google Scholar]
- Nomura K, Iizuka T, Kaji D, Asano-Mori Y, Ochiai Y, et al. Secondary esophageal squamous cell carcinoma after hematopoietic stem cell transplantation. J Cancer Res Clin Oncol. 2021;147:2137-2144.[Google Scholar][Cross Ref]



