Review Article - Onkologia i Radioterapia ( 2023) Volume 17, Issue 6
Prostate cancer stem cells: their role in prostate cancer and new therapies to target them
Pavan Kumar Rambatla1, Radha Krishna Nagumantri1, Satyanarayana Rentala3, Shanti Latha Pandrangi4 and Sireesha V Garimella4*2PhD student, Gandhi Institute of Technology and Management University, India
3School of Health Sciences, The Apollo University, Saketa, Murukambattu, Chittoor 517127, India
4Assistant professor, Department of Biotechnology, GITAM Institute of Science, GITAM University, Andhra Pradesh, India
Sireesha V Garimella, Assistant professor, Department of Biotechnology, GITAM Institute of Science, GITAM University, Andhra Pradesh, India, Email: garimela@gmail.com
Received: 21-Feb-2023, Manuscript No. OAR-23-89668; Accepted: 01-Jun-2023, Pre QC No. OAR-23-89668 (PQ); Editor assigned: 28-Feb-2023, Pre QC No. OAR-23-89668 (PQ); Reviewed: 18-Mar-2023, QC No. OAR-23-89668 (Q); Revised: 20-Apr-2023, Manuscript No. OAR-23-89668 (R); Published: 07-Jun-2023
Abstract
Prostate Cancer (PCa) is the second most common cancer and the sixth leading cause of cancer death in men worldwide. Prostate Cancer Stem cells (PCSC) play an important role in the relapse of PCa and its resistance to standard cancer therapeutics. This makes it important to study the role of Prostate cancer stem cells in PCa. We need to target and eradicate PCSCs in order to completely cure PCa and prevent PCa relapse. Markers that specifically identify PCSCs, such as ALDH, PSCA, CD44, will help in characterizing these cells, help in diagnostic procedures and in tracking the progression of cancer. Drugs such as Napabucasin and Trabectedin have been shown to specifically target PCSCs. Several miRNAs such as miR-34a, miR-7 have been shown to directly inhibit PCSCs. Natural metabolites like Galiellalactone and Parthenolide target PCSCs and have the advantage of reduced side effects. Nonconventional molecular pathways such as Chemokines and MMPs play critical roles in all the aspects of PCa and its progression, including PCSCs maintenance and propagation. A detailed understanding of the role of these pathways in PCa is essential to effectively curtail the disorder.
Keywords
napabucasin, trabectedin, chemokines, breast cancer, galiellalactone
Introduction
Prostate Cancer (PCa) is the second most common cancer and the 6th leading cause of cancer death in men worldwide. Around 1.1 million men were diagnosed with prostate cancer worldwide in 2012, which is 15% of all malignancies diagnosed in men. The estimated number of deaths because of PCa was almost 307,0001. 1.7 million new cases and 499000 deaths are expected as a result of PCa by 2030 worldwide 2. Despite the fact that Prostate cancer can be detected early on there has been a rising incidence of advanced-stage prostate cancer in the US population. There is stagnation in the progress made in curing prostate cancer. While the mortality rates of other cancers such as lung and breast cancer have decreased the mortality rate has remained stable for prostate cancer [1-3].
PCa is most prevalent in men of African descent, with 50% of them having latent prostate tumors by the age of 60, it is less prevalent in men of European descent with 50% of them having latent prostate tumors only by the age of 80 and is the least prevalent in Asian men with 50% of them having latent prostate tumors by the age of 904.
While the incidence of PCa was considered to be low in Asian countries including India ranging from 1 to 9/100 000 persons, because of changing lifestyles, increased awareness and access to medical facilities more cases of PCa are being reported in India. PCa is the second leading cause of cancer among males in large Indian cities like Delhi, Kolkata, Pune and the third leading cause of cancer in cities like Mumbai and Bengaluru. Data from national cancer registries show that the cases of PCa are on the rise and cancer projection data shows that this number will be doubled by the year 2020 [4-5]. PCa is the most common malignancy among men in Pakistan [6].
Despite Asia being considered a low-incidence area for prostate cancer, both incidence and mortality have steadily increased all over the continent. In 2020, Prostate cancer ranked among the top three most common cancers in men in 20 out of 47 countries [7].
The most common strategy followed for the treatment of Prostate Cancer is Radical prostatectomy, Radiation therapy, Hormonal therapy, and Combination therapy. Radical prostatectomy is a surgical procedure in which the prostate gland and seminal vesicles are removed. In Radiation therapy, a source of radiation such as X-rays is used to target and kill the tumor. Hormone therapy aims to decrease the levels of androgens in the patient, as androgens promote the growth of Prostate cancer cells. Hormone therapy can use Luteinizing Hormone-Releasing Hormone (LHRH) agonists (leuprolide, degarelix) that lower the testosterone produced by the testicles, androgen receptor antagonists (bicalutamide) and androgen biosynthesis inhibitors (Abiraterone). The sequence in which the therapies are used depends on the grade of the tumor. If cancer has been detected at an earlier grade such as Grade 1, the first treatment applied is a radical prostatectomy, followed by radiation therapy. This is followed by hormonal therapy. The next therapy suggested is combination therapy which involves hormonal therapy and radiotherapy. If cancer has metastasized to the lymph nodes or bones and other organs, then there is no standard recommendation for therapy. In such instances, therapy is decided based on the individual case [8].
Hormonal treatment for PCa involves Androgen deprivation treatment (ADT) which has been standard care for patients with advanced PCa since the 1940s. While ADT leads to an initial decrease in PCa growth and shows benefits for several patients, in most cases relapse occurs and the disease progresses to Castration Resistant Prostate Cancer (CRPC) which is resistant to ADT and other therapies used for cancer treatment. CRPC evolves through complex mechanisms and a detailed understanding of the individual mechanisms involved in the development of CRPC is lacking. It has been proposed that Prostate Cancer Stem Cells (PCSCs) play an important role in the progression and relapse of PCa
This review intends to draw attention to the various roles of Prostate Cancer Stem Cells in the progression of the cancer. Better understanding the role of the pathways and signaling molecules that play critical roles in PCSC maintenance and propogation will help develop drugs that specifically target PCSCs and prevent the progression of Prostate cancer.
Literature Review
Prostate Cancer Stem Cells
Emerging evidence suggests that Prostate Cancer stem cells play an important role in the relapse of PCa and the development of CRPC and its resistance to standard cancer therapeutics. The American Association for Cancer Research has defined a Cancer Stem cell as, “a cell within a tumor that possesses the capacity to self-renew and to cause the heterogeneous lineages of cancer cells that comprise the tumor”. The non-stem cells in the tumor have high proliferative ability but not an unrestricted proliferative ability like the stem cells. The tumor is thus arranged in a hierarchy similar to normal tissue and the cancer stem cells drive the growth and metastasis. The time between divisions of cancer stem cells is very long which makes them resistant to the current cancer therapies that are focused on the eradication of dividing cells. And hence cancer stem cells have been proposed as the source of cancer relapse
PCSCs have been shown to be resistant to hormonotherapy, chemotherapy, and radiotherapy, therefore cancer relapse may be due to more differentiated c ells b eing killed w hile l eaving the undifferentiated PCSCs intact. Th e us e of current ca ncer therapies causes an increase in the number of resistant CSCs subpopulations because they only spare the most resistant clones in a population of heterogeneous cancer cells. There is a pressing need for the development of more effective therapeutic agents that would specifically target the PCSCs [9].
Prostate Cancer stem cells and Castrationresistant phenotype
Persistent Androgen receptor (AR) signaling is being found to be a major contributor to CRPC development. Despite a castrationinduced reduction in the testosterone levels the intratumoral levels of androgens in CRPCs were found to be similar to those in hormone naïve tumors. Major mechanisms leading to AR resistant signaling are AR mutations and amplification. It was found that proteins that physically interact with AR such as FOXA1, MLL2, UTX, and ASXL1 are mutated in CRPC. Defects were found in genes such as TP53, RB1, PTEN, DDR [10].
Increasing evidence shows that the transition from androgensensitive to castration-resistant phenotype is associated with PCSCs and their expansion. ADT induces EMT which is a characteristic of PCSCs, this ,in turn, leads to CRPC development.
The IL6 pathway plays a key role in maintaining the stemness and plasticity of PCSCs especially under ADT. The Wnt/β-catenin pathway helps in the growth of PCSCs under ADT and thus helps develop resistance to ADT. ADT was also found to upregulate pluripotency genes such as SOX2 and NANOG which are essential for the renewal of PCSCs
Thus, it has been proposed that ADT leads to AR-dependent and AR independent enhancement of PCSCs expansion followed by CRPC acquisition. Further studies need to be conducted to better understand the molecular mechanisms involved in PCSC expansion and CRPC development [11].
A study found that HeyL (Hairy/enhancer-of-split related with YRPW motif-like protein) is aberrantly overexpressed in CRPC and this is directly correlated with aromatase expression. Upregulation of the HeyL-Aromatase axis led to enhancement of PCSC like properties in PC-3 cells. This also made cells resistant to androgen therapy. The HeyL-Aromatase activity suppressed PCSC apoptosis and influences CRPC development [12].
Markers for Prostate Cancer Stem Cells
Prostate Acid Phosphatase was used as a conventional diagnostic tool for PCa, however its usefulness was limited by its lack of sensitivity and specificity. More recently, Prostate Specific Antigen (PSA) was found to be a better diagnostic tool that could be detected in serum as well. The prevalence of this detection method has made it easier to detect a larger number of cases and explains the higher incidence of PCa in developed countries where this method of detection has become very common in the recent past. Levels of PSA were found to be increased in cancer patients as compared to normal males[13]. Pretreatment PSA serum level is the most significant predictor of therapy outcome after radiation therapy for PCa. A rise in the level of PSA with time after treatment is an indicator of disease relapse. Post-radiation PSA levels can serve as an early endpoint to evaluate treatment efficiency. PSA levels have also been found to be the most important prognostic factor [14].
ALDH has been identified as a marker for PCSCs. ALDH+ cells have enhanced clonogenic, migratory and metastasis inducing properties. α2, α6, and αv integrins, and CD44 are important for a tumor gaining an invasive phenotype. ALDH activity could be used as a means of classifying patients at risk of developing metastatic PCa[15]. ALDH is a stem cell marker because it has been shown to regulate several pathways involved in the stemness of cells [16]. ALDH activity was seen in a subset of prostate cells that also expressed several other stem cells markers such as Oct ¾, nestin, CD24 [17].
Recent studies have identified Prostate Stem Cell Antigen (PSCA) as a cell surface antigen in prostate epithelium. PSCA has been found to be expressed in high- grade prostate intraepithelial neoplasia and metastatic prostate cancers. PSCA marks a subpopulation of late intermediate (transit amplifying) cells which may be the target of prostate transformation in cancer [18]. PSCA can be considered as a putative stem cell marker as its expression is localized to basal cell epithelium in prostate tissue, which is considered to be the stem cell compartment of the prostate [19].
CD44 has been identified as a marker for PCSCs. Neuroendocrine (NE) cells are the only cells expressing CD44 in human PCa tissue. It is possible that NE cells are the only PCSCs and CD44 could be used as a marker to identify these cells [20]. Several studies have shown that CD44 is a stem cell marker. CD44+ cells have been shown to possess properties of self-renewal and differentiation into different cell types [21].
The expression of Hematological and Neurological expressed 1-like (HN1L) was found to be higher in CD133+ PCA tissues. HN1L expression was found to be important for the expression of other CSC markers such as Oct4, CD44, SRY-box transcription factor 2. HNL1 was found to promote stem cell-like properties and could be used as a marker for PCSCs [22].
A list of the markers for PCSCs mentioned in the review is shown in (Table 1). The table also mentions their functions and roles as markers.
Tab. 1. Markers for Prostate Cance Stem Cells
| Marker | Function | Role as a marker | Reference |
|---|---|---|---|
| PSCA | Expressed in metastatic prostate cancers | Marker for subpopulation of late intermediate (transit amplifying) cells. Putative PCSC marker. | Reiter et al., 199819 |
| ALDH | enhanced clonogenic, migratory and metastasis inducing properties | Marker for PCSCs. Can be used to classify patients at risk of developing metastatic PCa. | Clark et al., 201616 |
| CD44 | marker for cancer stem cells | Marker for PCSCs. | Palapattu et al., 200920 |
| HN1L | Expression of CSC markers such as Oct4 | Maker for PCSCs | Nong et al., 202222 |
Drugs for targeting Prostate Cancer Stem Cells
The most common drugs used in the treatment of Prostate cancer include Abiraterone, Enzalutamide and Prednisone. Abiraterone is an androgen biosynthesis inhibitor that inhibits the 17 a-hydroxylase/C17,20-lyase (CYP17) enzyme. CYP17 is essential for androgen biosynthesis and is expressed in the prostatic and testicular tissues. Enzalutamide acts as a competitive binder of androgens at the Androgen Receptor (AR) level. It prevents the translocation of AR to the nucleus and inhibits the binding of AR to chromosomal DNA.
In case of metastatic prostate cancer Luteinising Hormone-releasing Hormone (LHRH) agonists (leuprolide to degarelix) are administered. For treatment of CRPC Docetaxel is the primary drug of choice. It can be followed with other drugs including Cabazitaxel, Sipuleucel-T. In patients with CRPC and osseous metastasis, Zoledronic acid and denosumab can be used [23].
These standard drugs are not known to target PCSCs. Because of the central role that stem cells play in the relapse of PCa and its metastasis, it is important to devise new methods to target and eliminate PCSCs. Several new drug therapies are being developed which specifically target PCSCs.
Inhibitors of pathways involved in cancer
Napabucasin is a small molecule that inhibits STAT3 transcription. Treatment of PCa cell lines with Napabucasin caused decreased expression of stem cell markers like Nanog and Klf4, inhibited cell survival and proliferation, decreased cell motility and colony formation ability and increased apoptosis. This treatment had no adverse effect on normal adult stem cells [24].
Similarly, Trabectedin, a marine tetrahydroisoquinoline alkaloid, was shown to inhibit the growth of PCSCs by causing a G2/M cell cycle arrest and inducing apoptosis leading to decreased colony forming ability. Trabectedin caused this by increasing caspase expression and decreasing BCL-2 expression, it is an effective therapeutic agent to target PCSCs specifically [25].
Sunitinib, a multi tyrosine kinase inhibitor, has been shown to sensitize ALDH+CSCs to radiotherapy. Sunitinib treatment inhibited the growth of PCa cells by decreasing the expression of p-Akt, p-Erk-1/2, Id-1, and HIF-1a levels and decreased angiogenesis and vascular density in tumors. Combination therapy of Sunitinib plus radiation was shown to sensitize cells to radiation induced loss of clonogenicity, increased the rate of apoptosis, further decreased HIF-1α expression and impaired tumor vascularization[26].
Lemur Tyrosine Kinase 2 (LMTK2) has been shown to be an essential negative regulator of AR transcriptional activity and is downregulated in human PCa. LMTK2 plays an important role in pathogenesis of prostate cancer and progression to castration resistant stage. LMTK2 can be a novel therapeutic target in CRPC[27]. PCSCs could be killed with efficiency by using TNF-alpha if the NF-κB pathway was inhibited. The antioxidant pyrrolidine dithiocarbamate (PDTC) could be used to inhibit NF-κB. Inhibition of MYC/MAX signaling by using the small molecule KJ-Pyr-9 also suppressed the growth of PCSCs [28].
Integrins are known to be related with PCa growth with αvintegrins upregulated in PCSCs and αvβ3 integrins playing an important role in prostate cancer growth in the bone. GLPG0187, an αv-integrin antagonist, decreased the population of PCSCs in a dose dependent manner, prevented de novo formation and progression of bone metastasis, inhibited angiogenesis in vitro and in vivo. This shows that integrin inhibitors could be used in curative protocols in PCa [29].
The cancer stem cell antigen, Epithelial cell adhesion molecule (EpCAM) can be targeted using adoptive immunotherapy. Human peripheral blood lymphocytes expressing EpCAM specific chimeric antigen receptors can kill PCSCs in vitro and vivo. As this therapy targets CSCs specifically it is a potential treatment for cancer [30].
Micro-RNAs
Micro-RNAs are small non-coding RNAs with important functions in cellular processes and are being recognised as important agents for the diagnosis and treatment of cancer.
miR-34a is a negative regulator of CD44 and enforced expression of miR-34a in CD44+ PCSCs inhibited their growth, clonogenic expansion, sphere formation capacity, tumor regeneration and metastasis. Systemically delivered miR-34a reduced PC3 tumor burden by half and extended the survival of tumor bearing mice [31].
Similarly, miR-7 induced cell cycle arrest, inhibited prostate tumorigenesis and impaired the stem cell character of PCSCs. miR-7 suppressed a key stemness factor KLF4 and inhibited its downstream PI3K/Akt/p21 pathway[32].
miR-145 directly targeted AR and reduced the transcript and protein levels and downstream signaling of AR. Overexpression of miR-145 lead to a decrease in the number of PCSCs, decreased cell growth and the size of AR expressing tumors [33].
The miRNAs miR-145, miR-148, and miR-185 can be used as biomarkers for PCa. These miRNAs can also be used in miRNA therapy for PCa. These miRNAs were shown to regulate factors that maintain pluripotency of stem cells such as Oct4, Sox2, Klf4, CD133 and CD44. The miRNAs inhibited tumor cell viability, tumor sphere formation and bone metastases by repressing expression of CSC markers [34].
Natural products and metabolites
The adverse effects that chemotherapy agents have on the overall health has prompted research into more natural and less toxic therapeutic agents. Several natural compounds are being studied that show promise as therapeutic agents in cancer therapy
Galiellalactone, a fungal metabolite, inhibited the growth of and promoted the apoptosis of PCa cells both in vitro and in vivo, by inhibition of STAT3 signaling. Galiellalactone was shown to decrease the number of CSCs, decrease their proliferation and cause apoptosis in these cells. This shows Galiellalactone to be a potent anti-tumor drug in prostate cancers with constitutively active STAT3 [35,36].
The medicinal herb feverfew that is traditionally used to treat headaches and arthritis contains sesquiterpene lactone parthenolide (PTL), a natural phytochemical that is cytotoxic to stem cells in PCa cell lines. PTL causes cell death in tumor initiating cells and their progeny by inhibiting the NF-κB, STAT3, and JNK pathways [37].
The extract of the marine sponge Crambe crambe targets PCSCs and inhibits their self-renewal capacity, apoptosis resistance, invasive potential and tumorigenicity. Crambe crambe extract contains substances that make standard cancer therapy more effective by targeting CSCs [38]. Quercetin is a dietary bioflavonoid found in fruits and vegetables that has been shown to have antiproliferative properties and induce apoptosis in cancer cells by regulating cyclins and activating caspase-9 and caspase-3. Epigallocathechin gallate (EGCG) from tea is an important chemopreventive agent which induces growth arrest and apoptosis through various mechanisms. EECG and quercetin in combination had a greater inhibitory effect on the self-renewal and metastatic properties of PCSCs [39]
Combination treatments
Docetaxel is the first line of treatment in PCa but patients soon develop resistance to the drug. It also has a lot of side effects. It is necessary to find other drugs that are less toxic and more effective in dealing with CRPC. Two or more drugs being used in combination provides a unique strategy to use both the drugs at concentrations that are less than toxic and still provide better results. One of the major advantages of combination therapies is that they reduce the chances of development of drug resistance
ThymoQuinone (TQ) is a flavinoid extracted from Nigella sativa that is cytotoxic in several cancer cell lines. A combination treatment of Docetaxel and Thymoquinone resulted in significant cytotoxicity and apoptosis in hormone and drug resistant prostate cancer cell line DU-145. This was because of the combination blocking the PI3K/AKT pathway [40].
Piperine, the active ingredient of black pepper, suppressed the proliferation of PCa cells and induced apoptosis. Docetaxel is metabolized in the liver by the enzyme CYP3A4. Piperine has been shown to inhibit the activity of this enzyme. Co-administration of Piperin and Docetaxel led to a synergistic antitumor effect both in vitro and in xenograft animal models of CRPC [41].
A combination treatment of CRPC cells with the PI3K inhibitor GSK2126458 and MEK inhibitor, AZD6244 led to a synergistic anti-proliferative effect when compared with treatment with either compound alone. It was able to effectively prevent the growth of tumor in a DU145 xenograft model. The PI3K inhibitor GSK2126458 blocked the RAS/RAF/MEK/ERK pathway and the MEK inhibitor AZD6244 blocked the PI3K/AKT/mTOR pathway respectively[42].
Another novel strategy is to target the metabolic aberrations present in CRPC, while leaving normal cells unharmed. Simvastatin (SIM), is an inhibitor of HMG-CoA of the mevalonate pathway and Metformin (MET) acts as an indirect activator of the AMPK pathway. Treatment of C4-2B metastatic CRPC with SIM and MET led to inhibited aerobic glycolysis and macromolecule synthesis leading to impeded CRPC growth, proliferation and metastasis, cell cycle arrest and necrotic cell death mediated by Ripk-1 and Ripk-3. CRPC cells are resistant to apoptosis but not necrotic cell death, hence SIM+MET treatment is a novel treatment option for apoptosis and chemoresistant CRPC. Because normal cells do not possess the same metabolic aberrations they are not affected by SIM+MET treatment [43].
A combination treatment of Fractionated Irradiation (FIR) and B7-H3 targeting Chimeric Antigen Receptor (CAR) T cells was found to be highly effective in targeting PCSCs in hormoneinsensitive prostate cancer. FIR led to an increased expression of immune checkpoint B7-H3 (CD276) on PCSCs. When B7-H3 CAR T-cells were introduced in such tissues it led to a drastic decrease in the growth of hormone-insensitive PCa[44].
A list of the drugs that can target PCSCs which are mentioned in the review is shown in Table 2. The table also mentions their functions and targets (Table 2).
Tab.2. Drug To Target Prostate Cancer Stem Cells.
| Drug | What it targets | Function | Reference |
|---|---|---|---|
| Napabucasin | Inhibits STAT3 pathway | Inhibits expression of stem cell markers, motility and colony formation ability and increases apoptosis of cancer stem cells. | Zhang et al., 201624 |
| Sunitinib | Inhibits p-Akt, p-Erk-1/2, and Id-1, HIf-1 | Sensitizes ALDH+CSCs to radiotherapy. | Diaz et al., 201526 |
| Trabectedin | Growth of PCSCs | Inhibits the growth of PCSCs by causing a G2/M cell cycle arrest and inducing apoptosis. | Acikgoz et al., 201525 |
| Lemur Tyrosine Kinase 2 | Inhibits AR activity | Novel therapeutic target for CRPC | Shah et al., 201527 |
| GLPG 0187 | Decreases the population of PCSCs | Decreases the population of PCSCs in a dose dependent manner. | Horst et al., 201129 |
| Adaptive Immunotherapy using EpCAM specific chimeric antigen receptors | Targets PCSCs specifically | Targets and kills PCSCs | Deng et al., 201530 |
| miR-34a | negative regulator of CD44 | Inhibits PCSC growth, clonogenic and sphere formation capacity. | Liu et al.,201145 |
| miR-7 | Suppresses stemness factor KLF4 | Impairs the stem cell character of PCSCs. | Chang et al., 201532 |
| miR-145 | Regulates Androgen receptor expression | Decreases the number of PCSCs and prevents their growth. | Larne et al., 201533 |
| Galiellalactone | inhibitor of STAT3 pathway | Decreases the number of CSCs, inhibits their growth and increases apoptosis of CSCs | Hellsten et al., 201135 |
| Parthenolide | inhibits NF-KB signaling and STAT3 pathway | Cytotoxic to PCSCs. | Kawasaki et al., 200937 |
| Crambe crambe | Inhibits PCSCs | Inhibits self-renewal, apoptosis resistance, invasive potential and tumorigenicity of PCSCs | Ottinger et al., 201238 |
| Quercetin + Epigallocathechin gallate | Activates Caspase3,9 | Inhibitory effect on the self-renewal and metastatic properties of PCSCs. | Tang et al., 201039 |
| GSK2126458 +AZD6244 | Inhibit RAS/RAF/MEK/ERK pathway and PI3K/AKT/mTOR pathway respectively | synergistic anti-proliferative effect prevented the growth of tumor in a DU145 xenograft model. | Kwon et al., 201442 |
| Simvastatin+ Metformin | Inhibit the mevalonate pathway and activate AMPK pathway respectively | prolonged G1 phase cell cycle arrest and necrotic cell death in CRPC. | Babcook et al., 201443 |
| Docetaxel + Thymoquinone | block the PI3K/AKT pathway | significant cytotoxicity and apoptosis in hormone and drug resistant PCa cell lines | Dirican et al., 201440 |
Molecular Pathways involved in Prostate Cancer
Chemokines in Prostate Cancer
Chemokines are a large family of chemotactic cytokines that are known to cause inflammatory responses. Chemokines bind to G protein coupled receptors and cause a cascade of signals which activate several downstream targets depending upon the cell type and physiological and pathological response [45]. Different chemokines and their receptors have been shown to be expressed throughout PCa progression. Chemokines play a role in different aspects including proliferation, angiogenesis and metastasis [46].
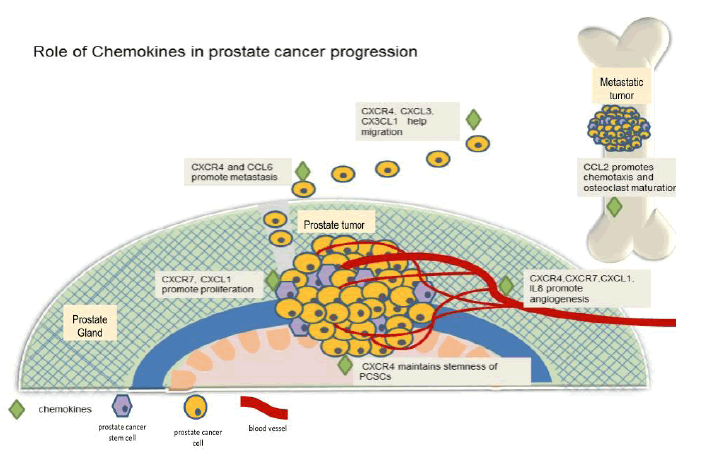
CXCR4 shows an increased expression in PCa progenitor cells. CXCR4 expression is essential for maintaining the stemness of PCa tumor initiating cells. Increased expression of CXCR4 led to a greater colony forming ability in PCa tumor initiating cells. CXCR4/CXCL12 acts as an upstream regulator of PI3K signaling and both these signaling pathways have a positive regulatory feedback loop and are both necessary for PCa stem cell renewal. Inhibition of CXCR4/CXCL12 pathway in PCa progenitors can lead to better cancer treatment [47].
CXCR4 is aberrantly expressed in several human cancers and is involved in cancer metastasis. CXCR4 expression was found in most patients (94.2%) with metastatic PCa. High-density tissue microarrays constructed from clinical samples from a large cohort of patients, showed that the expression of CXCL12 and CXCR4 expression are correlated with increasing malignant potential [48].
The CXCL12-CXCR4 ligand receptor system plays an important role in deciding the destination of metastatic cancer cells. CXCR4 expression can be used as an independent prognostic factor for bone metastasis in PCa[49]. Reactive oxygen species led to an increase in CXCR4 expression leading to increased cell invasion and migration. ROS treatment of PCa cells led to the inactivation of PTEN, thus showing that PTEN activity regulates the expression of CXCR4 indirectly. This also shows that CXCR4 enhances the proliferation and migration of cancer cells[50].
CXCR4 and CXCL12 enhance the migratory capabilities of human metastatic prostate cell lines. Primary PCa tumors also express CXCR4 mRNA at high levels. The CXCR12/CXCR4 pathway could be a novel target for therapeutic intervention[51]. CXCR4 was expressed in PCa cell lines such as PC3 and DU145 but was not expressed in normal prostate epithelial cells. CXCR12 was shown to significantly enhance the migration of PC3 and DU145 cells in a dose dependent manner. This chemotactic effect of CXCR12 was inhibited by blocking CXCR4. Thus, PCa cells metastasize to various sites mediated by the interaction of CXCR12/CXCR4 [52].
CXCR4 stimulation with SDF-1 also plays an important role in angiogenesis. SDF-1 binding to CXCR4 lead to a change in the equilibrium between pro and antiangiogenic factors in favor of the proangiogenic factors. CXCR4 knockdown led to the deregulation of PI3K/AKT and MEK/ERK signalling[53]. Downregulation of CXCR4 led to a decrease in the expression of VEGF and MMP-9, and this in turn led to a decrease in tumor growth and metastasis especially bone metastasis [54].
CXCL2 was shown to promote cancer stem cell and neuroendocrine phenotypes in prostate cancer cells. CCL12 did this through CXCR-4 mediated PKCa/NFkB signaling. This signaling led to metastasis and chemoresistance [55]
CCR6 chemokine receptor is expressed in various tumors and is implicated in tumor progression and metastasis. CCL20 which is the ligand of CCR6, is also upregulated in PCa compared to normal prostatic tissue. There is a significant association between the expression of CCR6 and the clinical and pathological features of more advanced PCa, including Gleason score. There is an association between CCR6 expression and lymph node metastasis [56].
The chemokine receptor CXCR7 was found to be highly expressed in undifferentiated PCa and bone lesions from metastatic disease. This indicates that CXCR7 plays a role in PCa progression. The proinflammatory chemokine receptor CXCR7 is elevated in several human cancers. CXCR7 is capable of modulating a number of cellular functions without being activated by its ligands. IL-8 was shown to upregulate CXCR7 expression and increase the proliferative ability of PCa cells. CXCR7 was able to increase cell proliferation, and angiogenesis by coupling with EGFR [57].
Chemokine CXCL3 and its receptor CXCR2 have been implicated in the progression of malignant tumors. CXCL3 and CXCR2 are overexpressed in PCa cell lines and PCa tissues. CXCL3 had a greater expression in more aggressive PCa cell line such as RWPE-1 and a lower expression in less aggressive cell like LNCaP, thus indicating that CXCL3 may be involved in the aggressiveness of PCa. Exogenous CXCL3 promoted the migration of PCa cell lines in a concentration dependent manner [58]. CXCR3 played key roles in PC cell proliferation and invasion. CXCR3A downregulation and CXCR3B upregulation led to the inhibition of PC-3 cell proliferation and invasion ability. The downregulation of CXCR3A led to increased expression of PLCβ, MMP-1, and MMP-3 except MMP-7 in PC-3 cells indicating that aberrant CXCR3 signaling affects prostate cells through downstream signaling pathways [59].
Chemokine CX3CL1 was shown to regulate cell adhesion, migration and survival of human PCa cells. Exposure to CX3CL1 led to occurrence of EMT and increased cell migration and invasion through a mechanism involving activation of TACE/ TGF-α/EGFR signaling. This shows that CX3CL1 may be used as a new target for PCa treatment [60].
A large-scale study found increased expression of CXCL1 in human PCa as compared to benign controls. Higher Gleason score tumors showed higher expression of CXCL1 as compared to lower Gleason score tumors. CXCL1 may enhance epithelialstromal interactions supporting tumor growth and invasion[61]. CXCL1 expression chemoattracts Adipose Stromal Cells (ASC) to the tumor and promotes prostate cancer progression. The ASCs secrete a smooth muscle actin which promotes tumor vascularization and growth. Obese patients with PCa have an increased expression of CXCL1 [62].
Tumor-associated macrophages (TAMs) are the most common immune cells in tumor microenvironment. It was found that TAMs secrete CCL5. TAM derived CCL5 was found to promote PCSCs self-renewal and cancer metastases by activating the β-catenin/STAT3 signaling pathway. TAMs and CCL5 could be potential molecular targets for the elimination of PCSCs [63]
Human prostate cancer cell lines have been shown to secrete their own angiogenic CXC chemokines such as interleukin (IL-)8. IL8 was found to be a major positive regulator of neovascularization and tumorigenesis of PCa tumors. Different cell lines use distinct angiogenic chemokines to promote tumorigenesis, PC3 cell line uses IL-8 and Du145 uses GRO-α [64]. TGF-β signaling plays an important role in the growth of PCa tumors in nude mice and it does this by regulating the expression of IL-8. TFG-β stimulated IL-8 expression increased angiogenesis in PCa tumors contributing to their growth and progression [65].
The anti-androgen drug enzalutamide led to an increase in the metastatic phenotype by increasing the TNF-α signaling. The increased TNF-α signaling leads to autocrine CCL2 expression which binds to cognate receptor CCR2 and induces chemotaxis and pro-metastatic effects in prostate cancer [66]. CCL2 activates survivin through a PI3K/AKT dependent mechanism and helps protect PCa cells from autophagic death. The several functions of CCL2 in PCa make it an attractive target in metastatic PCa [67]. CCL2 leads to promotion of osteoclast maturation in the bone tumor microenvironment and suppression of cytotoxic lymphocytes [68].
Chemokines play crucial roles in all the stages of prostate cancer. Chemokines are involved in the survival, proliferation of Prostate cancer cells (CCL5,CXCR4, CCR6, CXCR7,CXCL1, CXCR2, CXCR3) Several chemokines play a role in metastasis and angiogenesis (CCR6,CXCR4,CXCR7,IL-8,CCL2). Increased chemokine expression correlated with increased malignant cancer potential. Downregulating chemokines slowed the growth of prostate cancer. This makes them targets for chemotherapeuticagents. The various rolesthat chemokines play in PCa are shown in (Figure 1).
Figure 1: Role of Chemokines in prostate cancer progression Chemokines play an important role in the progression of prostate cancer. Chemokines help maintain stemness of PCSCs, promote proliferation of prostate cancer cells. Chemokines help in metastasis and migration of ancer cells.
Matrix Metalloproteinases in Prostate Cancer
Matrix Metalloproteinases (MMP) are a group of 24 protease enzymes that are so named because of their dependence on metal ions for catalytic activity and their ability to degrade structural proteins of the extracellular matrix (ECM). MMPs play important roles in several cancers and this has made them the focus of anticancer research.
MMPS are inhibited specifically by Tissue inhibitors of MMPs (TIMPs) which are a family of small extracellular proteins. TIMPs have been shown to have biological functions apart from inhibiting MMPs with functions in cell growth, differentiation, angiogenesis and apoptosis.
Prostate cancer tissue exhibits a decreased expression of TIMPs and an increased expression of MMPs. Elevated MMP activity in tumors promotes cell proliferation, angiogenesis, epithelial to mesenchymal transition (EMT) and metastasis. Studies have consistently demonstrated an increased expression of MMP2 and MMP9 in PCa tissues [69].
MMP-10 was found to to play an important role in the maintenance of highly tumorigenic, cancer-initiating, metastatic stem-cell like cell population in lung cancer. MMP-10 is a critical lung cancer stem cell gene [70].
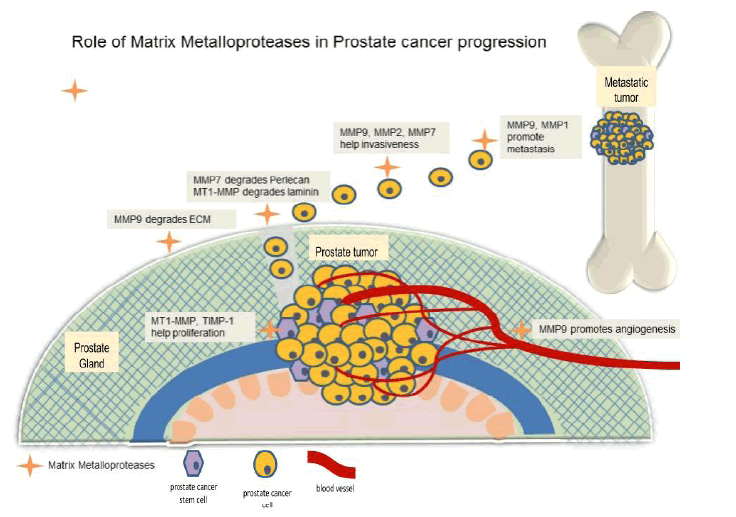
Notch1 controls the expression of MMP9 directly by enhancing its transcriptional activity[71]. MMP9 has been shown to play a major role in PCa metastasis. There is a direct relationship between expression levels of MMP9 and uPAR in different PCa cell lines and their invasiveness. Downregulation of MMP9, uPAR and Cathepsin B in PCa cell lines led to a marked decrease in their tumor invasiveness, migration and angiogenic ability[72].
MMP9 is always found to be present in higher concentrations in malignant PCa tumors. There is a positive correlation between MMP9 expression, disease progression and clinical outcome for patients in several kinds of tumors. MMP9 activates urokinase plasminogen activator in both soluble and tumor cell receptor bound forms. MMP9 is an important protease that participates in invasion and metastasis [73].
MMP9 was shown to play an important role in the angiogenesis of PCa tumors. Endothelial cells found in the microvascules of PCa tumors during angiogenesis were shown to secrete IL-6. This IL6→AR→TGFβ→MMP9 signaling pathway mediated by endothelial cells can be a good therapeutic target to prevent PCa metastasis [74].
Some studies have shown a relationship between the chemokine pathway and metalloproteinases. In ovarian cancer stem-like cells (CSLCs) there was an autocrine activation of CCR1 and CCR3 receptors by chemokine CCL5 which led to an activation of the NF-κB signaling and in turn led to an elevation of MMP9 levels. This was responsible for the enhanced invasiveness and metastasis of ovarian cancers [75].
Kindlin-2(Mig-2) is a focal adhesion protein that functions in integrin mediated cell-extracellular matrix adhesion and spreading. It was found that Kindlin-2 promoted the binding of NF-ΚB to MMP-9 and MMP-2 promoters which led to the upregulation of MMP9 and MMP-2 expression and in turn led to increased invasiveness of PCa cells [76].
ADAM 17 (tumor necrosis factor-α converting enzyme (TACE) is a member of metzincin superfamily of Zn-dependent matrix metalloproteinases and has been implicated in several malignancies. ADAM17 expression levels are correlated with the invasive ability of androgen independent PCa cell lines. ADAM17 contributes to PCa cell invasion by shedding EGFR ligand TGF-α, which subsequently activates the EGFR-MEK-ERK signaling pathway, leading finally to over- expression of MMP-2 and MMP-9 [77]. MMP-2 and MMP-9 levels could be used in association with PSA to monitor advanced PCA.
MMP-13 was found to be the most carcinoma specific MMP with levels of MMP-13 being elevated to 7-8-fold in patients with PCa. MMP-13 could be used as a diagnostic marker for PCa [78].
MMP1 was shown to directly influence the metastatic potential of PCa cells. Histone deacetylase inhibitors are being studied as novel agents for cancer chemotherapy. The HDAC inhibitor MHY219 was shown to cause downregulation of MMP1 and MMP2 and induce expression of TIMP-1 by inhibiting the expression of HDAC1. Treatment of PCa cells with MHY219 led to a decrease in the migration of PCa cells [79].
TGF-β1 upregulates the expression of MMP2 and MMP9 which are closely associated with tumor invasion. TGF-β1 was shown to induce secretion, activation and cell surface association of MMP9 in PCa cells. TGF-β1 was also shown to increase the expression of MMP3 and induce the expression of MMP7 [80].
Membrane-Type 1 Matrix Metalloproteinase (MT1-MMP) has been implicated in the aggressiveness of a variety of cancers. Cell surface expression of MT1-MMP has come to be associated with tumor growth, invasion and metastasis. MiR-335 is a miRNA that is being studied for tumor associated properties. In some cell lines MiR-335 led to an increased expression of MT1-MMP accompanied with increased proliferation and motility[81].
IGF-1R tyrosine kinase signaling, which plays an important role in proliferation and differentiation of normal cells during development has also been implicated in tumor progression and metastasis. IGF-1R regulates the expression of MT1-MMP in PCa cell lines [82].
Laminin-10 is found in the basal lamina and is a substrate for cell migration and cell adhesion. MT1-MMP can cause cleavage of Laminin-10 and this helps in the increase of PCa transmigration and linear migration through the processed Laminin-10 [83].
PCa varieties overexpressing MMP7 have been shown to have increased invasiveness. Perlecan, a multimodular five domain heparan sulfate proteoglycan, is an important part of ECM involved in several biological functions. MMP7 has been shown to degrade perlecan. MMP7 is the strongest candidate protease that can degrade perlecan in the basal membrane and destabilize the stromal matrix which is an essential step for cancer invasion and migration to occur [84]. In most invasive tumors perlecan expression was upregulated along with upregulation of its MMP7 destruction. This enzyme substrate relationship between MMP7 and perlecan can be used as an index for tissue invasion [85]. Expression of MMP-7 mRNA was found to be upregulated in primary and invasive PCa.
Serum levels of MMP7 can be used as a potential marker to identify patients with metastatic PCa. MMP7 serum levels were found to be significantly elevated in metastatic PCa. High serum concentration of MMP7 is an independent predictor of disease specific mortality in men with localized PCa [86]. Overexpressing MMP7 in a human PCa cell line changed its in vivo phenotype and enabled it to become more invasive and colonize the underlying stroma [87]. High MMP7 level is an independent predictor of disease specific mortality in men with localized prostate cancer [86].
Tissue Inhibitor of Metallo Proteinase-1 (TIMP-1) has been shown inhibit the catalytic activity of MMPs but at the same time also inhibits apoptosis and promotes tumor growth and angiogenesis. Elevated levels of TIMP-1 have been associated with poor prognosis in several cancers. Higher expression of TIMP-1 was associated with decreased survival in patients with metastatic castration resistant PCa . Neuroendocrine PCa is an aggressive form of PCa, which is characterised with elevated levels of neuroendocrine markers such as chromogranin A [CGA]. The increased TIMP-1 levels were associated with higher expression of CGA suggesting that TIMP-1 plays a role in the neuroendocrine differentiation of CRPC.
Matrix Metalloproteinases are involved in invasiveness and metastasis of prostate cancer (MMP9, MMP2, MMP1, MT1- MMP, MMP7). MMPs such as TIMP-1 are involved in angiogenesis. MMP expression (MMP9 and MMP2) can be used as markers to monitor advanced Prostate cancer. As they are involved in metastasis, they are ideal targets to prevent the spread of Prostate cancer. The different functions of MMPs in PCa are shown in Figure 2.
Figure 2: Role of Matrix Metalloproteinases in prostate cancer progression Matrix Metalloproteinases (MMPs) play an important role in prostate cancer progression. MMPs help in degradation of extracellular matrix and laminin, promote angiogenesis, promote invasiveness and metastasis.
Conclusion
It is now well established that we need to target and eradicate PCSCs in order to completely cure PCa and prevent development of CRPC and PCa relapse. Markers that specifically identify PCSCs will help in characterizing these cells, help in diagnostic procedures and in tracking the progression of the cancer. There is an urgent need to understand the detailed molecular pathways through which ADT leads to the propagation of PCSCs and development of CRPC. This will aid the development of effective therapy agents that target PCSCs and prevent CRPC. Several natural metabolites are being found to be effective in targeting cancer and are a desirable option as therapeutic agents because of their easy availability and non-toxicity.
Nonconventional molecular pathways such Chemokines and MMPs play critical roles in all the aspects of PCa and its progression, including PCSCs maintenance and propagation. A detailed understanding of the role of these pathways in PCa is essential to effectively curtail the disorder.
Therapies that target stem cells can be used at each stage of Prostate cancer treatment. Drugs such as Napabucasin, Trabectedin that target PCSCs can be used in chemotherapy. Drugs such as Sunitinib which sensitize cells to radiotherapy, can be used in combination with radiotherapy to increase the efficacy of radiotherapy and specifically target PCSCs. Therapeutics such as the m RNas miR-34a, miR-7 can be administered locally after surgery to eradicate any remaining PCSCs. The natural metabolites that target PCSCs ((Galiellalactone , PTL, Crambe crambe EECG and quercetin ) have few side effects and can be used in combination with traditional chemotherapy agents. In treatment of CRPC, Piperin can be used in combination with Docetaxel for a synergestic antitumor effect. The combination therapies mentioned in the review can be used in patients who have relapsed or have CRPC. Therapies that target stem cells should be incorporated at every stage of Prostate Cancer treatment to give patients an edge over Prostate cancer.
References
- Jacques Ferlay IS, Dikshit R, Eser S, Mathers C, Rebelo M,et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer. 2014; 136:29.
- Ferlay J. Cancer incidence, mortality and prevalence worldwide. GLOBOCAN2002. 2004.
[Google Scholar] [Crossref]
- Siegel RL, Miller KD, Jemal A. Cancer statistics. Cancer Surgery. 2018.
- Rebbeck TR, Haas G. Temporal trends and racial disparities in global prostate cancer prevalence. The Can j urol. 2014; 21:7496.
- Jain S, Saxena S, Kumar A. Epidemiology of prostate cancer in India. Meta Gene. 2014; 2: 596–605.
- Bashir MN. Epidemiology of prostate cancer. Asian Pacific journal of cancer prevention. 2015;16:5137-5141.
- Zhu Y, Mo M, Wei Y, Wu J, Pan J,et al. Epidemiology and genomics of prostate cancer in Asian men. Nat Rev Urol. 2021;18:282-301.
- European Association of Urology. EAU guidelines on prostate cancer. EAU https://uroweb. org/guideline/prostate cancer. 2019.
- Jaworska D, Król W, Szliszka E. Prostate cancer stem cells: research advances. International journal of molecular sciences. 2015 ;16:27433-49.
- Tong D. Unravelling the molecular mechanisms of prostate cancer evolution from genotype to phenotype. Critical Reviews in Oncology/Hematology. 2021; 163:103370.
- Ojo D, Lin X, Wong N, Gu Y, Tang D. Prostate cancer stem-like cells contribute to the development of castration-resistant prostate cancer. Cancers. 2015;7:2290-308.
- Lin Q, Cao J, Du X, Yang K, Shen Y,et al. The HeyL-aromatase axis promotes cancer stem cell properties by endogenous estrogen-induced autophagy in castration-resistant prostate cancer. Frontiers in Oncology. 2022;11:5590.
- Wang MC, Papsidero LD, Kuriyama M, Valenzuela LA, Murphy GP,et al. Prostate antigen: a new potential marker for prostatic cancer. The Prostate. 1981;2:89-96.
- Zagars GK, von Eschenbach AC. Prostateâ?specific antigen. An important marker for prostate cancer treated by external beam radiation therapy. Cancer. 1993;72:538-548.
- van den Hoogen C, van der Horst G, Cheung H, Buijs JT, Lippitt JM,et al. High aldehyde dehydrogenase activity identifies tumor-initiating and metastasis-initiating cells in human prostate cancer. Cancer research. 2010;70:5163-5173.
- Clark DW, Palle K. Aldehyde dehydrogenases in cancer stem cells: potential as therapeutic targets. Ann transl med. 2016 ;4:. 787-798
- Burger PE, Gupta R, Xiong X, Ontiveros CS, Salm SN,et al. High aldehyde dehydrogenase activity: a novel functional marker of murine prostate stem/progenitor cells. Stem cells. 2009;27:2220-2228.
- Tran CP, Lin C, Yamashiro J, Reiter RE. Prostate stem cell antigen is a marker of late intermediate prostate epithelial cells. Mol Cancer Res. 2002 ;1:113-121.
- Palapattu GS, Wu C, Silvers CR, Martin HB, Williams K,et al. Selective expression of CD44, a putative prostate cancer stem cell marker, in neuroendocrine tumor cells of human prostate cancer. The Prostate. 2009;69:787-98.
- Du L, Wang H, He L, Zhang J, Ni B,et al. CD44 is of functional importance for colorectal cancer stem cells. Clin cancer res. 2008;14:6751-6760.
- Nong S, Wang Z, Wei Z, Ma L, Guan Y,et al. HN1L promotes stem cellâ?like properties by regulating TGFâ?β signaling pathway through targeting FOXP2 in prostate cancer. Cell biol int. 2022 ;46:83-95.
- Heidenreich A, Bastian PJ, Bellmunt J, Bolla M, Joniau S,et al. EAU guidelines on prostate cancer. Part II: treatment of advanced, relapsing, and castration-resistant prostate cancer. Eur urol. 2014 ;65:467-479.
- Zhang Y, Jin Z, Zhou H, Ou X, Xu Y,et al. Suppression of prostate cancer progression by cancer cell stemness inhibitor napabucasin. Cancer medicine. 2016;5:1251-1258.
- Acikgoz E, Guven U, Duzagac F, Uslu R, Kara M,et al. Enhanced G2/M arrest, caspase related apoptosis and reduced E-cadherin dependent intercellular adhesion by trabectedin in prostate cancer stem cells. PLoS One. 2015 ;10:e0141090. PLoS One. 2015;10:1-17.
- Diaz R, Nguewa PA, Redrado M, Manrique I, Calvo A. Sunitinib reduces tumor hypoxia and angiogenesis, and radiosensitizes prostate cancer stemâ?like cells. The Prostate. 2015;75:1137-1149.
- Shah K, Bradbury NA. Lemur Tyrosine Kinase 2, a novel target in prostate cancer therapy. Oncotarget. 2015; 6:14233.
- Kaltschmidt B, Witte KE, Greiner JF, Weissinger F, Kaltschmidt C. Targeting NF-κB Signaling inCancerStemCells:ANarrativeReview.Biomedicines.2022;10:261.
- van der Horst G, van den Hoogen C, Buijs JT, Cheung H, Bloys H,et al. Targeting of αv-integrins in stem/progenitor cells and supportive microenvironment impairs bone metastasis in human prostate cancer. Neoplasia. 2011 ;13:516-519.
- Deng Z, Wu Y, Ma W, Zhang S, Zhang YQ. Adoptive T-cell therapy of prostate cancer targeting the cancer stem cell antigen EpCAM. BMC immunology. 2015;16:1-9.
- Li J, Lam M, Reproducibility Project: Cancer Biology. Registered report: the microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Elife. 2015 ;4:211-215.
- Chang YL, Zhou PJ, Wei L, Li W, Ji Z,et al. MicroRNA-7 inhibits the stemness of prostate cancer stem-like cells and tumorigenesis by repressing KLF4/PI3K/Akt/p21 pathway. Oncotarget. 2015 ;6:24017.
- Larne O, Hagman Z, Lilja H, Bjartell A, Edsjö A,et al. miR-145 suppress the androgen receptor in prostate cancer cells and correlates to prostate cancer prognosis. Carcinogenesis. 2015 ;36:858-866.
- Coradduzza D, Cruciani S, Arru C, Garroni G, Pashchenko A,et al. Role of miRNA-145, 148, and 185 and stem cells in prostate cancer. Int J Mol Sci. 2022; 23:1626.
- Hellsten R, Johansson M, Dahlman A, Sterner O, Bjartell A. Galiellalactone inhibits stem cell-like ALDH-positive prostate cancer cells. PLoS One. 2011;6:1-9.
- Don-Doncow N, Escobar Z, Johansson M, Kjellström S, Garcia V,et al. Galiellalactone Is a Direct Inhibitor of the Transcription Factor STAT3 in Prostate Cancer Cells. J Biol Chem. 2014; 289:15969-15978.
- Ottinger S, Kloppel A, Rausch V, Liu L, Kallifatidis G,et al.Targeting of pancreatic and prostate cancer stem cell characteristics by Crambe crambe marine sponge extract.Int j cancer. 2012 Apr 1;130(7):1671-81.
- Tang SN, Singh C, Nall D, Meeker D, Shankar S, Srivastava RK. The dietary bioflavonoid quercetin synergizes with epigallocathechin gallate (EGCG) to inhibit prostate cancer stem cell characteristics, invasion, migration and epithelial-mesenchymal transition. J Mol Signal. 2010;5:1-5.
[Google scholar][Cross ref]
- Dirican A, Atmaca H, Bozkurt E, Erten C, Karaca B,et al. Novel combination of docetaxel and thymoquinone induces synergistic cytotoxicity and apoptosis in DU-145 human prostate cancer cells by modulating PI3K–AKT pathway. Clin Transl Oncol. 2014;17:145-151.
- Makhov P, Golovine K, Canter D, Kutikov A, Simhan J et al. Co-Administration of Piperine and Docetaxel Results in Improved Anti-Tumor Efficacy via Inhibition of CYP3A4 Activity. 2013;72:661-667.Co-Administration.
- Kwon T, Kim Y, Park H, Song SH, Suh N,et al. 5 Synergistic anticancer effect of combination treatment with MEK inhibitor and PI3K inhibitor in castration-resistant prostate cancer. European Urology Supplements.2014; 1:e5.
- Babcook MA, Sramkoski RM, Fujioka H, Daneshgari F, Almasan A,et al. Combination simvastatin and metformin induces G1-phase cell cycle arrest and Ripk1-and Ripk3-dependent necrosis in C4-2B osseous metastatic castration-resistant prostate cancer cells. Cell death dis. 2014 ;5:e1536
- Zhang Y, He L, Sadagopan A, Ma T, Dotti G, et al .Targeting radiation-resistant prostate cancer stem cells by B7-H3 CAR T cells. Mol cancer ther.2021;20:577-588.
- Liu C, Kelnar K, Liu B, Chen X, Calhoun-Davis T,et al. The microRNA miR-34a inhibits prostate cancer stem cells and metastasis by directly repressing CD44. Nat. med. 2011 ;17:211-215.
- Vindrieux D, Escobar P, Lazennec G. Emerging roles of chemokines in prostate cancer. Endocrine-related cancer. 2009 ;16:663.
- Dubrovska A, Elliott J, Salamone RJ, Telegeev GD, Stakhovsky AE,et al. CXCR4 expression in prostate cancer progenitor cells. PloS one. 2012 ;7:e31226.
- Sun YX, Wang J, Shelburne CE, Lopatin DE, Chinnaiyan AM,et al. Expression of CXCR4 and CXCL12 (SDFâ?1) in human prostate cancers (PCa) in vivo. Journal of cellular biochemistry. 2003 ;89:462-73.
- Akashi T, Koizumi K, Tsuneyama K, Saiki I, Takano Y,et al. Chemokine receptor CXCR4 expression and prognosis in patients with metastatic prostate cancer. Cancer science. 2008 ;99:539-542.
- Chetram MA, Don-Salu-Hewage AS, Hinton CV. ROS enhances CXCR4-mediated functions through inactivation of PTEN in prostate cancer cells. Biochem biophys res commun. 2011 ;410:195-200.
- Masters JR, Albrecht M, Mittler A. The Role of the Chemokine. 2003;2:2003.
[Google Scholar][Cross ref]
- Mochizuki H, Matsubara A, Teishima J, Mutaguchi K, Yasumoto H,et al. Interaction of ligand–receptor system between stromal-cell-derived factor-1 and CXC chemokine receptor 4 in human prostate cancer: a possible predictor of metastasis. Biochem biophys res commun. 2004;320:656-663.
- Wang J, Wang J, Sun Y, Song W, Nor JE,et al. RETRACTED: Diverse signaling pathways through the SDF-1/CXCR4 chemokine axis in prostate cancer cell lines leads to altered patterns of cytokine secretion and angiogenesis.
- Wang Q, Diao X, Sun J, Chen Z. Regulation of VEGF, MMPâ?9 and metastasis by CXCR4 in a prostate cancer cell line. Cell biol int. 2011; 35:897-904.
- Jung Y, Cackowski FC, Yumoto K, Decker AM, Wang J,et al. CXCL12γ Promotes Metastatic Castration-Resistant Prostate Cancer by Inducing Cancer Stem Cell and Neuroendocrine PhenotypesCXCL12γ Promotes Development of m-CRPC. Cancer research. 2018 Apr 15;78(8):2026-39.
- Ghadjar P, Loddenkemper C, Coupland SE, Stroux A, Noutsias M,et al. Chemokine receptor CCR6 expression level and aggressiveness of prostate cancer. J. cancer res. clin. oncol. 2008 ;134:1181-9.
[Google Scholar][Cross ref]
- Singh RK, Lokeshwar BL. The IL-8–Regulated Chemokine Receptor CXCR7 Stimulates EGFR Signaling to Promote Prostate Cancer GrowthCXCR7 Promotes Cell Proliferation via EGFR Activation. Cancer research. 2011;71:3268-77.
- Gui SL, Teng LC, Wang SQ, Liu S, Lin YL,et al.Overexpression of CXCL3 can enhance the oncogenic potential of prostate cancer. International urology and nephrology. 2016 May;48:701-9. Int Urol Nephrol. 2016;48:701-709.
- Shen D, Cao X. Potential role of CXCR3 in proliferation and invasion of prostate cancer cells. Int. J Clin Exp Pathol. 2015; 8:8091.
- Tang J, Xiao L, Cui R, Li D, Zheng X,et al. CX3CL1 increases invasiveness and metastasis by promoting epithelial-to-mesenchymal transition through the TACE/TGF-α/EGFR pathway in hypoxic androgenâ??independent prostate cancer cells. Oncology Reports. 2016;35:1153-1162.
- Miyake M, Lawton A, Goodison S, Urquidi V, Rosser CJ. Chemokine (CXC motif) ligand 1 (CXCL1) protein expression is increased in high-grade prostate cancer. Pathol.-Res. Pract. 2014 ;210:74-78.
- Zhang T, Tseng C, Zhang Y, Sirin O, Corn PG,et al. CXCL1 mediates obesity-associated adipose stromal cell trafficking and function in the tumour microenvironment. Nat commun.2016;7:11674.
- Huang R, Wang S, Wang N, Zheng Y, Zhou J,et al. CCL5 derived from tumor-associated macrophages promotes prostate cancer stem cells and metastasis via activating β-catenin/STAT3 signaling. Cell death dis.. 2020 ;11:234.
- Moore BB, Arenberg DA, Stoy K, Morgan T, Addison CL,et al. Distinct CXC chemokines mediate tumorigenicity of prostate cancer cells. Am j pathol. 1999; 154:1503-1512.
- Zhang F, Lee J, Lu S, Pettaway CA, Dong Z. Blockade of transforming growth factor-β signaling suppresses progression of androgen-independent human prostate cancer in nude mice. Clin cancer res. 2005 ;11:4512-4520.
- Sha K, Yeh S, Chang C, Nastiuk KL, Krolewski JJ. TNF signaling mediates an enzalutamide-induced metastatic phenotype of prostate cancer and microenvironment cell co-cultures. Oncotarget. 2015 ;6:25726.
- Zhang J, Lu Y, Pienta KJ. Multiple roles of chemokine (CC motif) ligand 2 in promoting prostate cancer growth. J Natl Cancer Inst. 2010 ;102:522-528.
- Zhang J, Patel L, Pienta KJ. CC chemokine ligand 2 (CCL2) promotes prostate cancer tumorigenesis and metastasis. Cytokine growth factor rev. 2010 ;21:41-48.
- Gong Y, Chippada-Venkata UD, Oh WK. Roles of matrix metalloproteinases and their natural inhibitors in prostate cancer progression. Cancers. 2014 Jun 27;6:1298-1327.
- Justilien V, Regala RP, Tseng IC, Walsh MP, Batra J,et al. Matrix metalloproteinase-10 is required for lung cancer stem cell maintenance, tumor initiation and metastatic potential.PloS one. 2012;7:e35040.
- Bin Hafeez B, Adhami VM, Asim M, Siddiqui IA,et al. Targeted knockdown of Notch1 inhibits invasion of human prostate cancer cells concomitant with inhibition of matrix metalloproteinase-9 and urokinase plasminogen activator. Clin cancer res. 2009 ;15:452-459.
- Nalla AK, Gorantla BH, Gondi CS, Lakka SS, Rao JS. Targeting MMP-9, uPAR, and cathepsin B inhibits invasion, migration and activates apoptosis in prostate cancer cells. Cancer gene ther. 2010;17:599-613.
- Aalinkeel R, Nair BB, Reynolds JL, Sykes DE, Mahajan SD,et al.Overexpression of MMP-9 contributes to invasiveness of prostate cancer cell line LNCaP. Immunol investig. 2011 ;40:447-464.
- Wang X, Lee SO, Xia S, Jiang Q, Luo J,et al. Endothelial Cells Enhance Prostate Cancer Metastasis via IL-6→ Androgen Receptor→ TGF-β→ MMP-9 SignalsEndothelial Cells Increase Metastatic Potential of Prostate Cancer. Mol cancer ther. 2013;12:1026-1037.
- Long H, Xie R, Xiang T, Zhao Z, Lin S,et al. Autocrine CCL5 signaling promotes invasion and migration of CD133+ ovarian cancer stem-like cells via NF-κB-mediated MMP-9 upregulation.Stem cells. 2012;30:2309-2319.
- Yang JR, Pan TJ, Yang H, Wang T,et al. Kindlin-2 promotes invasiveness of prostate cancer cells via NF-κB-dependent upregulation of matrix metalloproteinases.Gene. 2016 ;576:571-576.
- Xiao LJ, Lin P, Lin F, Liu X, Qin W,et al. ADAM17 targets MMP-2 and MMP-9 via EGFR-MEK-ERK pathway activation to promote prostate cancer cell invasion. Int j oncol. 2012;40:1714-1724.
- Morgia G, Falsaperla M, Malaponte G, Madonia M, Indelicato M,et al. Matrix metalloproteinases as diagnostic (MMP-13) and prognostic (MMP-2, MMP-9) markers of prostate cancer. Urol res. 2005;33:44-50.
- De U, Kundu S, Patra N, Ahn MY, Ahn JH,et al. A new histone deacetylase inhibitor, MHY219, inhibits the migration of human prostate cancer cells via HDAC1. Biomolecules & Therapeutics. 2015;23:434-441.
- Festuccia C, Angelucci A, Gravina GL, Villanova I, Teti A,et al.Osteoblastâ?derived TGFâ?β1 modulates matrix degrading protease expression and activity in prostate cancer cells. Int j cancer 2000 ;85:407-415.
- Rojas F, Hernandez ME, Silva M, Li L, Subramanian S,et al. The oncogenic response to miR-335 is associated with cell surface expression of membrane-type 1 matrix metalloproteinase (MT1-MMP) activity. PLoS One. 2015 ;10:1-12.
- Sroka IC, McDaniel K, Nagle RB, Bowden GT. Differential localization of MT1â?MMP in human prostate cancer tissue: role of IGFâ?1R in MT1â?MMP expression. The Prostate. 2008 ;68:463-476.
- Bair EL, Chen ML, McDaniel K, Sekiguchi K, Cress AE, et al. Membrane type 1 matrix metalloprotease cleaves laminin-10 and promotes prostate cancer cell migration. Neoplasia. 2005;7:380-389.
- Grindel BJ, Martinez JR, Pennington CL, Muldoon M, Stave J,et al. Matrilysin/matrix metalloproteinase-7 (MMP7) cleavage of perlecan/HSPG2 creates a molecular switch to alter prostate cancer cell behavior. Matrix Biology. 2014 ;36:64-76.
- Jin S, Xu B, Yu L, Fu Y, Wu H,et al. The PD-1, PD-L1 expression and CD3+ T cell infiltration in relation to outcome in advanced gastric signet-ring cell carcinoma, representing a potential biomarker for immunotherapy. Oncotarget. 2017;8:38850.
- Szarvas T, Becker M, Vom Dorp F, Meschede J, Scherag A,et al. Elevated serum matrix metalloproteinase 7 levels predict poor prognosis after radical prostatectomy. Int J Cancer 2011;128:1486-1492.
- Powell WC, Knox JD, Navre M, Grogan TM, Kittelson J,et al.Expression of the metalloproteinase matrilysin in DU-145 cells increases their invasive potential in severe combined immunodeficient mice. Cancer research. 1993 Jan 15;53:417-422.
- Oh WK, Vargas R, Jacobus S, Leitzel K, Regan MM, et al. Elevated plasma tissue inhibitor of metalloproteinaseâ?1 levels predict decreased survival in castrationâ?resistant prostate cancer patients. Cancer. 2011 Feb 1; 117:517-525.
- Gong Y, Chippadaâ?Venkata UD, Galsky MD, Huang J, Oh WK. Elevated circulating tissue inhibitor of metalloproteinase 1 (TIMPâ?1) levels are associated with neuroendocrine differentiation in castration resistant prostate cancer. The Prostate. 2015 ;75:616-627.