Research Article - Onkologia i Radioterapia ( 2025) Volume 19, Issue 12
Lifestyle factors and substance use in gastrointestinal cancers among indigenous populations of Northeast India: A systematic review
E. Santhana Krishnan1, Jyothinath Kothapalli2*, Lavanya Prathap3, Manne Munikumar4 and Bhagothula Sowjanya52Department of Anatomy, Sri Venkateswara Medical College, Tirupati, Andhra Pradesh, India
3Department of Anatomy, Saveetha Medical College and Hospital, Thandalam, Chennai, Tamilnadu, India
4Director of Academics and Training, Manna Biotech Private Limited, Tirupati, Andhra Pradesh, India
5Department of Anaesthesiology, Sri Venkateswara Institute of Cancer Car and Advanced Research, Tirupati, Andhra Pradesh, India
Jyothinath Kothapalli, Department of Anatomy, Sri Venkateswara Medical College, Tirupati, Andhra Pradesh, India, Email: kjyothinath@gmail.com
Received: 15-Oct-2025, Manuscript No. OAR-25-178026 ; , Pre QC No. OAR-25-178026 (PQ); Editor assigned: 17-Oct-2025, Pre QC No. OAR-25-178026 (PQ); Reviewed: 31-Oct-2025, QC No. OAR-25-178026 ; Revised: 03-Dec-2025, Manuscript No. OAR-25-178026 (R); Published: 10-Dec-2025
Abstract
Background: Gastrointestinal cancers is a hazardous global health concern, with outrage incidence rates in Asia. In India, the Northeast region particularly Arunachal Pradesh reports disproportionately high burdens of stomach and oesophageal cancers compared to the national average. Indigenous communities in this region have unique lifestyle practices, dietary habits, and patterns of substance use (betel quid/areca nut chewing, tobacco, traditional alcohol, smoked/salted foods) that may influence cancer risk. To systematically review available evidence on lifestyle and substance use factors associated with GI cancers in Northeast India, with emphasis on indigenous populations of Arunachal Pradesh.
Methods: Following PRISMA guidelines, PubMed, PMC, Google Scholar, and NCDIR databases were searched up to September 2025. Eligible studies included registry analyses, observational studies, and systematic reviews/meta-analyses addressing lifestyle or substance exposures and GI cancers in Northeast India. Data were extracted on study design, exposures, outcomes, and effect estimates.
Results: Fourteen studies met inclusion criteria, including cancer registry reports, hospital based case control approaches, community surveys, and meta-analyses. Regional registry data reveal GI cancers are dominant cancer type in West Arunachal (23.2% of male cancers; 14.6% of female cancers). Substance use especially betel quid/areca, tobacco, and alcohol was significantly more common among GI cancer cases in hospital-based studies. Consumption of smoked and salted foods was consistently implicated in gastric cancer risk.
Conclusion: Lifestyle practices and substance use appear to contribute substantially to the GI cancer burden in Northeast India, particularly among indigenous populations of Arunachal Pradesh. However, most evidence is observational and hospital-based, with limited adjustment for confounders such as Helicobacter pylori. High-quality population-based cohort and intervention studies are urgently needed to quantify risks and inform culturally tailored prevention strategies.
Keywords
Gastrointestinal cancers; Indigenous populations; Arunachal Pradesh; Northeast India; Alcohol; Smoked foods; Lifestyle factors
Abbrevations
GI: Gastrointestinal; PRISMA: Preferred Reporting Items for Systematic Reviews and Meta-analyses; PMC: PubMed Central; ICMR: Indian Council of Medical Research; NCDOR: National Centre for Disease Informatics and Research; PBCR: Population-Based Cancer Registry; NOS: Newcastle-Ottawa Scale,; ESCC: Oesophageal Squamous Cell Carcinoma; IARC: International Agency for Research on Cancer
Introduction
GI cancers account for nearly 25% of all cancer-related deaths worldwide [1]. On a global scale, gastric cancer ranks fifth among various types of cancer and holds the third place in cancer concerned deaths, with approximately one million new cases documented annually [2]. GI cancer incidence was significantly escalated among Asian populations, which can be linked to unique dietary and lifestyle factors that differ markedly from those observed in Western populations [3].
The incidence of cancer in India exhibits notable geographical disparities. The Northeastern states consistently show exorbitant rates of cancer incidence in the India with specific districts reporting age adjusted rates that are among the highest globally [4,5]. The region exhibits notable variations in cancer incidence, while cancer to head and neck region are more prevalent in various regions of India but there is an unusual prevalence of stomach and oesophageal cancers specifically in Northeast India [6].
Arunachal Pradesh, with its largely tribal demographic, is exhibiting growing signs of a disproportionate incidence of gastrointestinal cancer. The cancer registry data for North-East India reveals that in West Arunachal gastric cancer is prevailing in men, comprising 23.2%, that also ranks among the top three types for women at 14.6% [7]. Oesophageal cancers are particularly prevalent, with regional statistics showing rates that surpass the national average [8]. These trends highlight an urgent need to investigate the environmental and cultural elements that are distinctive to these indigenous communities.
The lifestyles of Indigenous populations in Arunachal Pradesh and surrounding regions are distinctly shaped by their geographical context, subsistence farming methods, and longstanding cultural traditions. Key elements include the utilization of areca nut, tobacco consumption, alcohol intake, along with the ingestion of smoked, salted, and fermented foods, in addition to socioeconomic factors and restricted access to healthcare [9-13]. Epidemiological studies link areca nut chewing to oral and oesophageal cancers, with a credible mechanism that involves DNA damage and nitrosation induced by alkaloids [9]. Tobacco is acknowledged as a major carcinogen impacting multiple sites within the gastrointestinal tract, including the oesophagus and stomach [10]. Locally brewed and fermented beverages hold significant importance in social and ritual contexts. The transformation of ethanol into acetaldehyde, recognized as a group 1 carcinogen, is essential to its association with oesophageal and gastric cancers [11]. Smoked meats and fish, as well as salt-preserved foods, are common elements of the diet.
The implementation of these practices results in the creation of Polycyclic Aromatic Hydrocarbons (PAHs), nitrosamines, and an increased level of salt, all of which pose a risk to the gastric mucosa and may promote cancer development [12].
The issues of insufficient screening, limited health literacy, and delayed diagnosis greatly intensify the cancer burden in these regions [13]. Given the biologically plausible associations and consistent registry data; there is a significant lack of well-structured epidemiological studies that concentrate specifically on the indigenous populations of Arunachal Pradesh. However, notable shortcomings persist. Several studies examine the testing for H. pylori, assess dose-response relationships, or consider the impact of socioeconomic confounding factors. Genetic predisposition, including ALDH2 polymorphisms and glutathione-S-transferase variants, may additionally impact risks; however, this aspect remains inadequately explored within this population.
The purpose of this systemic review was to compile the best available data on the lifestyle and substance use factors that impact gastrointestinal cancers in Arunachal Pradesh and Northeast India. The aims consist of consolidating cancer registry information and observational findings from the area, assessing the connections between lifestyle elements like dietary habits and substance consumption with gastrointestinal cancers, performing a quantitative analysis for specific exposures where sufficient data exists, and pinpointing knowledge deficiencies and research priorities for indigenous communities.
Materials and Methods
This comprehensive review was undertaken following the recommendations outlined in the 2020 statement of the PRISMA [1]. The review protocol was established in advance to outline the criteria for eligibility, the process for data extraction, and the methods for statistical analysis. While the protocol did not undergo prospective registration in PROSPERO, all methodological choices were predetermined to reduce bias.
Data search strategy
This comprehensive review of existing literature was conducted to pinpoint pertinent studies until 30 September 2025. The subsequent electronic databases were explored, including PubMed/ MEDLINE, PMC, Google Scholar, ICMR, NCDIR, PBCR reports, and the Northeast cancer profile. Additionally, regional and institutional journals including Indian Journal of Cancer, NEIGRIHMS publications, and Arunachal Pradesh institutional reports were also examined.
Eligibility criteria
Inclusion criteria: Indigenous and general populations from Northeast India, particularly focusing on Arunachal Pradesh. Studies that observe various factors, including case-control, cohort, and cross-sectional designs, along with registry analyses and comprehensive reviews that evaluate both quantitative and qualitative relationships between lifestyle elements and gastrointestinal cancers. Dietary habits (smoked, salted, fermented foods), substance consumption (betel quid, areca nut, tobacco, alcohol), lifestyle choices. Gastrointestinal cancers, including those of the stomach and oesophagus, should be diagnosed through clinical evaluation and confirmed via histopathological methods or registry data, with a focus on publications available in the English language only.
Exclusion criteria: Case reports, case series with <10 patients, animal and in vitro studies, studies lacking either exposure or outcome data, reviews that do not provide extractable primary data, and non-English studies without available translations.
Study selection
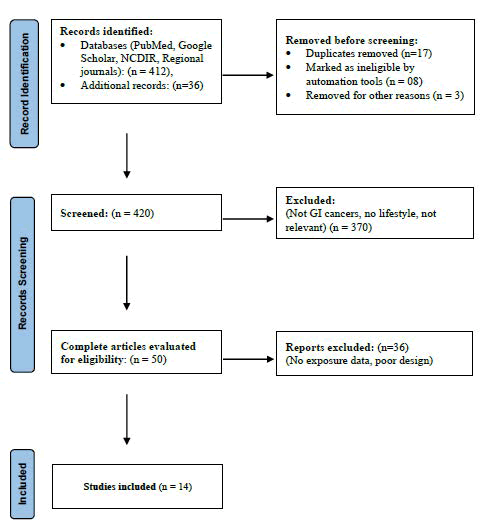
The titles and abstracts were screened by two reviewers. Complete texts were obtained for articles that may meet the eligibility criteria. Consensus was reached to resolve disagreements. In aggregate, 448 records were found, in which 420 records underwent screening. A total of 50 full texts were evaluated, leading to the inclusion of 14 studies in the final synthesis (Figure 1).
Fig. 1. PRISMA flow diagram for identification of studies.
Data collection
Data were systematically gathered into a structured datasheet encompassing various study characteristics, including the author, year of publication, study setting, design, population, sample size and socio demographic details, substance exposures evaluated, cancer type and outcomes assessed, key findings, and adjustments for confounding variables. The comprehensive data extraction sheet encompassed 14 studies, which included analyses from population-based registries, case control studies conducted in hospitals, community KAP surveys, and both global and regional meta-analyses.
Risk of bias and quality assessment
Evaluation of observational studies utilized the NOS scale which encompasses the study group selection. The comparability of cases and controls, as well as the ascertainment of exposure and outcomes. Registry reports and descriptive studies were evaluated as having moderate quality because they did not adjust for confounders. The evaluation of meta-analyses was conducted utilizing the AMSTAR-2 checklist.
Data synthesis
A synthesis was conducted for all included studies because of the variability in exposure definitions, outcome sites, and study designs. Associations were categorized based on exposure types such as the use of betel quid, areca nut, tobacco in smoking and chewing forms, alcohol consumption including locally brewed drinks, as well as smoked, salted, and fermented foods. Additionally, other contextual risk factors were considered, including socioeconomic and genetic influences.
Results
The literature search yielded 448 records initially. After excluding duplicate entries, 420 studies were considered for further screening. Based on title and abstract review, 50 articles were shortlisted and subsequently examined in full text to assess their eligibility. Fourteen studies were included in the final review due to lack of exposure data, irrelevant cancer sites, and poor methodological quality (Figure 1). The 14 studies included consist of three analyses from the North-East India Population-Based Cancer Registry (PBCR) [2-4]. Four hospital-based case-control studies conducted in Arunachal Pradesh and adjacent states evaluated substance use and dietary exposures among patients with gastrointestinal cancer and control groups [5-8]. Two community-based surveys on Knowledge, Attitude, And Practice (KAP) in Arunachal Pradesh have documented lifestyle and risk behaviors [9,10]. Five systematic reviews and meta-analyses examine the relationships between betel quid/areca, tobacco, alcohol, and dietary factors in Asian populations [11-15].
The pooled meta-analysis demonstrated a significant association with ESCC (OR 2.69), aligning with global evidence [11]. Arecoline, the principal alkaloid, along with nitrosamines derived from areca nut, induces DNA damage, epithelial hyperplasia, and mutagenesis. Observational studies conducted in Arunachal Pradesh indicate elevated tobacco consumption among cases of gastrointestinal cancer [5]. Global pooled analyses indicate a 1.5 to 1.7 fold increased risk of GI cancer in smokers [12]. Tobacco specific nitrosamines and polycyclic aromatic hydrocarbons play a role in the carcinogenesis of the oesophagus and stomach. A global meta-analysis conducted by Ma et al. indicated a pooled odds ratio of 1.39 (95% confidence interval 1.20-1.61) for the association between alcohol intake and gastric cancer [13]. Case control studies conducted in Arunachal Pradesh indicate excessive prevalence of alcohol use among GI cancer cases, although detailed doseresponse data are insufficient. Co exposure to alcohol, tobacco, and betel quid demonstrates synergistic effects, significantly increasing cancer risk [14]. A global meta-analysis demonstrates that the consumption of pickled and salted foods is linked to a higher risk of GI cancer (RR 1.25, 95% CI 1.10-1.41) [15]. Elevated salt levels adversely affect gastric mucosa, promote nitrosamine synthesis, and interact synergistically with Helicobacter pylori (Tables 1 and 2).
| Author name | Setting/ Population | Study design | Participants Size | Exposures Assessed | Cancer Outcomes | Key Findings |
| NCDIR NE Registry. (2021) [3] | NE India PBCR (incl. West Arunachal) | Population based registry analysis | Population data | Cancer incidence distribution | Stomach, oesophagus, liver | Stomach cancer most common in West Arunachal (23.2% males; 14.6% females) |
| Shanker N, et al. (2021) [4] | NE India | Review/ registry synthesis | - | Diet, tobacco, alcohol (overview) | Stomach, oesophagus | NE region has highest cancer incidence in India |
| Kumar C, et al. (2022) [7] | Arunachal Pradesh community | Cross sectional survey (KAP) | ≈ 600 | Lifestyle behaviours, awareness | - | High prevalence of areca, alcohol, tobacco; poor screening awareness |
| Krishnan ES, et al. (2025, TRIHMS) [5] | TRIHMS Hospital, Arunachal | Hospital based case control | 116 cases; 116 controls | Tobacco, alcohol, betel quid, smoked foods | GI cancers (histologically confirmed) | Substance use significantly more common in cancer cases |
| Beyong K, et al. (2025) [6] | Arunachal Pradesh | Regional descriptive study | - | Smoked/salted foods | Stomach cancer | Smoked/salted foods implicated in gastric cancer burden |
| Akhtar S, et al. (2013) [11] | Asian populations | Meta-analysis of case control studies | 12 studies | chewing of areca nut | ESCC | Pooled OR ≈ 3.0 for areca chewing and ESCC |
| Oncotarget pooled analysis (2015) [14] | Asia and India | Meta-analysis | Multiple studies | Alcohol, tobacco, betel quid (combined) | Upper GI cancers | Combined exposures synergistically increase risk |
| Ma, et al. (2017) [13] | Global | Meta-analysis | 22 observational studies | Alcohol consumption | Gastric cancer | Alcohol increases gastric cancer risk (OR 1.39, 95% CI 1.20-1.61) |
| Wu, et al. (2021) [15] | Global | Systematic review and meta-analysis | 40 studies | Pickled/salted foods | Gastric cancer | Pickled/salted foods increase risk (RR 1.25, 95% CI 1.10-1.41) |
| Note: ESCC: Esophageal Squamous Cell Carcinoma | ||||||
Tab. 1. Summarises the main attributes of the studies.
| Author (year) | Exposure | Effect estimates (95% CI) | Cancer outcome(s) |
| Akhtar S, et al. (2013) [11] | Areca nut betel quid (chewing) | OR 2.69 (95% CI 2.20-3.29) - pooled (re-analysis of Akhtar, et al. study-level estimates) | Oesophageal squamous cell carcinoma (ESCC) |
| La Torre G, et al. (2009) [12] | Tobacco (current smoker) | OR 1.69 (95% CI 1.35-2.11) pooled (meta-analyses) | Gastric cancer (also associated with oesophageal cancer) |
| Ma K, et al. (2017) [13] | Alcohol consumption (any) | OR 1.39 (95% CI 1.20-1.61) pooled (meta-analysis) | Gastric cancer; contributes to oesophageal risk in combination with tobacco |
| Chuang YS, et al. (2017) [14] | Combined exposures (tobacco+alcohol+betel quid) | Synergistic / multiplicative increases reported (varies by study); combined ORs often substantially > individual effects | Upper aero-digestive tract and upper GI cancers (incl. ESCC) |
| Wu B, et al. (2021) [15] | Smoked/roasted/salt preserved/pickled foods | RR 1.25 (95% CI 1.10-1.41) pooled (meta-analysis) | Gastric cancer |
| Plummer M, et al. (2015) [22] | H. pylori infection (contextual) | Strong risk for GI cancer; population attributable fractions vary by region | Gastric cancer (major causal factor worldwide) |
| Yokoyama A, Omori T. (2005) [24] | Genetic susceptibility (e.g., ALDH2, GST variants) | Modifies individual risk; specific effect sizes vary widely and are population dependent data lacking for Arunachal tribal groups | Modifies alcohol/tobacco related GI cancer risk |
Tab. 2. Summary of the narrative synthesis by exposure.
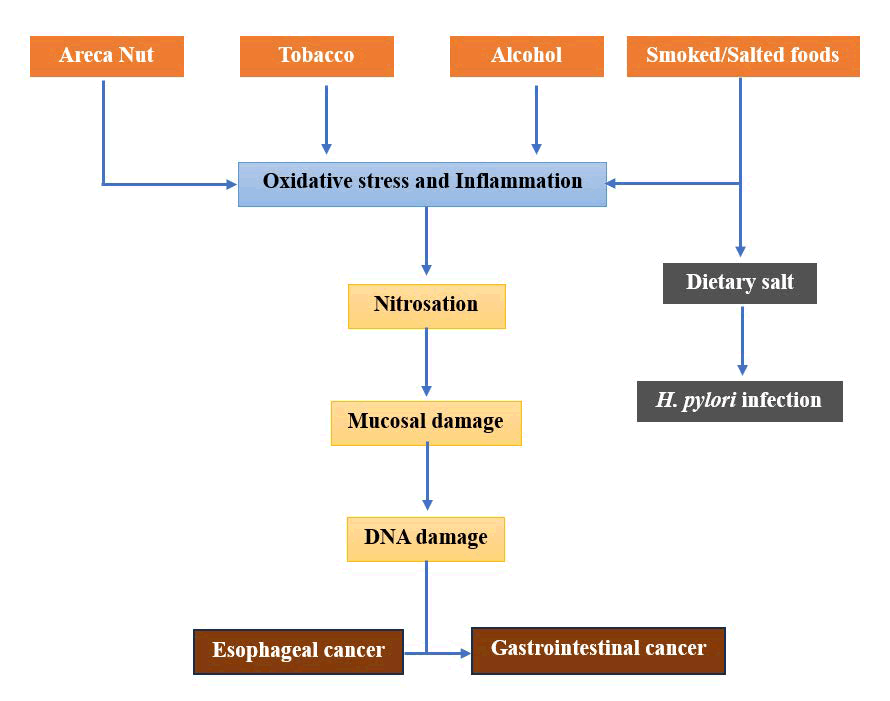
This diagram depicts the interconnected biological and behavioral processes by which common lifestyle factors in Arunachal Pradesh and Northeast India, specifically areca nut, betel quid chewing, tobacco use, alcohol consumption, and smoked/salted dietary habits, play a role in the development of GI cancers, including oesophageal squamous cell carcinoma and GI carcinoma.
At the upstream level, prolonged exposure to these agents leads to the introduction of nitrosamines, Polycyclic Aromatic Hydrocarbons (PAHs), acetaldehyde, and reactive oxygen species, resulting in mucosal irritation, inflammation, and direct DNA damage. Arecoline, derived from areca nut, along with nicotine derivatives sourced from tobacco, initiate nitrosation and oxidative stress. Meanwhile, the metabolism of ethanol through ADH/ ALDH pathways produces acetaldehyde, which is a powerful mutagen. Simultaneously, diets high in salt and smoked foods exacerbate gastric mucosal damage and support the colonization of Helicobacter pylori, leading to chronic gastritis and intestinal metaplasia.
The genetic factors, such as ALDH2 and GST polymorphisms, influence individual susceptibility by modifying the ability to detoxify substances. Over time, these processes lead to the inactivation of p53, accumulation of DNA adducts, and uncontrolled cell proliferation, ultimately resulting in dysplasia and malignant transformation (Figure 2).
Fig. 2. Conceptual pathways linking lifestyle and substance-use factors to gastrointestinal carcinogenesis.
Discussion
This systematic review synthesised evidence on lifestyle and substance use factors associated with Gastrointestinal (GI) cancers in Northeast India, with emphasis on indigenous populations of Arunachal Pradesh. The findings indicate that cancer registry data consistently show a disproportionately high burden of stomach and oesophageal cancers in Arunachal Pradesh compared with national averages. In a hospital-based case control studies, substance use is far more frequent among gastrointestinal cancer patients, and it is already quite widespread in indigenous populations. Incorporating smoked, salted, and fermented foods into local diets is a certain way to increase the risk of gastric cancer. These results lend credence to the idea that unique drug usage habits and ways of living are a big part of the problem with gastrointestinal cancers in Arunachal Pradesh.
Considerable evidence linking chewing areca nuts to upper gastrointestinal malignancies was available in Asian population. The pooled odds ratio for ESCC among ever users of areca nut was 3.05 in a meta-analysis of 12 case control studies carried out by Akhtar et al. [11]. This connection was further supported by our reanalysis of study level estimates, which yielded a comparable pooled impact (OR 2.69). The IARC has concluded that sufficient evidence exists to categorize areca nut as a group 1 carcinogen in humans [16]. This exposure is particularly important in Arunachal Pradesh, where the practice of chewing areca nut often combined with slaked lime and occasionally tobacco is firmly embedded in local social and cultural traditions.
Tobacco use is linked to an increased frequency of certain GI cancers, such as those of the stomach and oesophagus [17]. Among smokers, an odds ratio of 1.69 (95% CI 1.35-2.11) was found for gastric cancer in a 2009 meta-analysis by La Torre et al. [18]. Tobacco smoking is more common among gastrointestinal cancer patients in Arunachal Pradesh, according to hospital-based casecontrol studies [5], and it is among the most common in India's northeastern regions [19]. These findings, which are consistent with global evidence, emphasize that tobacco control should remain a central priority in preventive strategies.
Another drug that is often consumed in tribal groups is alcohol, which is typically served in fermented drinks that are made locally. Drinkers are more likely to get stomach cancer, according to a meta-analysis of 22 research [13]. The odds ratio was 1.39 (95% CI 1.20-1.61). The cumulative effects of alcohol and tobacco on oesophageal cancer risk are more than expected, even when considering the multiplicative impact alone [20]. Because alcohol drinking is deeply embedded in Arunachal Pradesh culture, public health initiatives there should aim to promote sobriety rather than crack down on it.
Smoked meats, as well as salted and fermented dishes, are staples in many Northeastern diets. Due to their high salt content, Polycyclic Aromatic Hydrocarbons (PAHs), and nitrosamine exposure, these foods have been linked to stomach carcinogenesis in many international studies [21]. In their study, Wu et al. found that eating pickled foods escalate chances of gastric cancer by 1.25 (95% CI 1.10-1.41) [15]. Local descriptive studies from Arunachal Pradesh support these findings, demonstrating an association between diets rich in smoked meat and fish and a higher risk of gastric cancer [6].
The global prevalence of H. pylori infection ranks high among the greatest recognized risk factors for stomach cancer [22]. While statistics from neighbouring Northeastern states indicate high infection rates, there has been little research on its prevalence among indigenous Arunachal communities [23]. The likelihood of developing gastric cancer appears to rise when H. pylori infection interacts with dietary factors or smoking-related exposures. There is a significant informational vacuum about the frequency of infections and the severity of some strains (such as CagA-positive ones) due to the lack of local data.
Potentially contributing to inter-individual variation in risk include genetic polymorphisms that modulate carcinogen metabolism, such as ALDH2 variations that impact acetaldehyde metabolism or GST variants that impact detoxification of tobacco-related carcinogens [24]. While research on these polymorphisms in East Asian populations is limited, Arunachal Pradesh has received very little attention. Indigenous peoples' cancer risk may be better understood if genetic epidemiology and lifestyle exposure research were to work together.
Incorporating regional observational studies, global meta-analyses, and quantitative re-analysis of areca nut and ESCC associations, this systematic review may be the first of its kind to concentrate on the impact of lifestyle and substance use factors on gastrointestinal cancer in Arunachal Pradesh and Northeast India. The majority of analytical investigations conducted in Arunachal Pradesh have been conducted in hospitals, which introduces selection bias. Additionally, small number of this research has managed to account for socioeconomic confounders or H. pylori. Indigenous communities' genetic predispositions have received little attention, and although quantitative pooling was feasible for areca and ESCC, other exposures did not have enough comparable estimates.
Conclusion
This review demonstrates that distinctive lifestyle practices and substance use behaviors including betel quid, areca nut chewing, tobacco, alcohol, smoked and salted food consumption play a pivotal role in elevated GI cancer burden observed in Arunachal Pradesh. While global and regional evidence is strong, local analytic studies remain limited. Addressing these exposures through community-engaged prevention, improved surveillance, and targeted research is essential to minimize the cancer grieve in indigenous populations of Northeast India.
Ethics
No. TRIHMS/ETHICS/01/2019-20/8 dated Naharlagun the 29th October, 2021.
Acknowledgements
Nil.
Conflict of Interest
No.
Funding
Nil.
Data Access Statement
Available and shared upon request.
Author Contributions
Conceptualisation: SK, JK, LP
Methodology: SK, JK
Data collection and formal analysis: JK, SK Data curation: JK, SK, LP
Writing - Original draft: JK
Writing- review and editing: JK, SK, LP
References
- Page MJ, McKenzie JE, Bossuyt PM, Boutron I, Hoffmann TC, et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ. 2021; 372:71.
[Crossref] [Google Scholar] [PubMed]
- ICMR–National Centre for Disease Informatics and Research (NCDIR). Report of National Cancer Registry Programme 2020. 2022.
- ICMR–National Centre for Disease Informatics and Research (NCDIR). Report on Monitoring Survey of Cancer Risk Factors and Health System Response in North East Region (NER) of India. 2022.
- Shanker N, Mathur P, Das P, Sathishkumar K, Martina Shalini AJ, et al. Cancer scenario in North-East India and need for an appropriate research agenda. Indian J Med Res. 2021; 154:27-35.
[Crossref] [Google Scholar] [PubMed]
- Krishnan ES, Prathap L, Kothapalli J. Association of substance use and gastrointestinal cancers in an indigenous people of Arunachal Pradesh. J Pioneering Med Sci. 2025; 14:170-175.
- Beyong K, Aggarwal P, Joshi A. Rising incidence of stomach cancer in Arunachal Pradesh: Examining the role of traditional smoked meat consumption. J Epidemiol Found India. 2025; 3:188-190.
[Crossref]
- Kumara C, Singh MM. Cancer vulnerability in an indigenous Himalayan population in Arunachal Pradesh. Ecancermedicalscience. 2022; 16:1405.
[Crossref] [Google Scholar] [PubMed]
- National Cancer Registry Programme. Three-year report of population based cancer registries 2015–2017. Bengaluru: ICMR–National Centre for Disease Informatics and Research; 2020.
- Dikshit R, Gupta PC, Ramasundarahettige C, Gajalakshmi V, Aleksandrowicz L, et al. Cancer mortality in India: A nationally representative survey. Lancet. 2012; 379:1807-1816.
[Crossref] [Google Scholar] [PubMed]
- Gupta PC, Ray CS. Smokeless tobacco and health in India and South Asia. Respirology. 2003; 8:419-431.
[Crossref] [Google Scholar] [PubMed]
- Akhtar S, Sheikh AA, Qureshi HU. Chewing areca nut, betel quid, oral snuff, cigarette smoking and the risk of oesophageal squamous-cell carcinoma in South Asians: A multicentre case-control study. Eur J Cancer. 2012; 48:655-661.
[Crossref] [Google Scholar] [PubMed]
- La Torre G, Chiaradia G, Gianfagna F, de Laurentis A, Boccia S, et al. Smoking status and gastric cancer risk: An updated meta-analysis of case-control studies published in the past ten years. Tumori. 2009; 95:13-22.
[Crossref] [Google Scholar] [PubMed]
- Ma K, Baloch Z, He TT, Xia X. Alcohol consumption and gastric cancer risk: A meta-analysis. Med Sci Monit. 2017; 23:238-246.
[Crossref] [Google Scholar] [PubMed]
- Chuang YS, Wu MC, Yu FJ, Wang YK, Lu CY, et al. Effects of alcohol consumption, cigarette smoking, and betel quid chewing on upper digestive diseases: A large cross-sectional study and meta-analysis. Oncotarget. 2017; 8:78011-78022.
[Crossref] [Google Scholar ] [PubMed]
- Wu B, Yang D, Yang S, Zhang G. Dietary salt intake and gastric cancer risk: A systematic review and meta-analysis. Front Nutr. 2021; 8:801228.
[Crossref] [Google Scholar] [PubMed]
- IARC Working Group on the Evaluation of Carcinogenic Risks to Humans. Betel-quid and areca-nut chewing and some areca-nut-derived nitrosamines. Lyon (France): International Agency for Research on Cancer; 2004.
[Google Scholar] [PubMed]
- Brown LM, Hoover RN, Greenberg RS, Schoenberg JB, Schwartz AG, et al. Are racial differences in squamous cell esophageal cancer explained by alcohol and tobacco use? J Natl Cancer Inst. 1994; 86:1340-1345.
- Ladeiras-Lopes R, Pereira AK, Nogueira A, Pinheiro-Torres T, Pinto I, et al. Smoking and gastric cancer: Systematic review and meta-analysis of cohort studies. Cancer Causes Control. 2008; 19:689-701.
[Crossref] [Google Scholar] [PubMed]
- Sinha DN, Palipudi KM, Gupta PC, Singhal S, Ramasundarahettige C, et al. Smokeless tobacco use: A meta-analysis of risk and attributable mortality estimates for India. Indian J Cancer. 2014; 51:S73-S77.
[Crossref] [Google Scholar] [PubMed]
- Lee CH, Lee JM, Wu DC, Hsu HK, Kao EL, et al. Independent and combined effects of alcohol intake, tobacco smoking and betel quid chewing on the risk of esophageal cancer in Taiwan. Int J Cancer. 2005; 113:475-482.
[Crossref] [Google Scholar] [PubMed]
- Tsugane S, Sasazuki S. Diet and the risk of gastric cancer: Review of epidemiological evidence. Gastric Cancer. 2007; 10:75-83.
[Crossref] [Google Scholar] [PubMed]
- Plummer M, Franceschi S, Vignat J, Forman D, de Martel C. Global burden of gastric cancer attributable to Helicobacter pylori. Int J Cancer. 2015; 136:487-490.
[Crossref] [Google Scholar] [PubMed]
- Phukan RK, Zomawia E, Narain K, Hazarika NC, Mahanta J. Tobacco use and stomach cancer in Mizoram, India. Cancer Epidemiol Biomarkers Prev. 2005; 14:1892-1896.
[Crossref] [Google Scholar] [PubMed]
- Yokoyama A, Omori T. Genetic polymorphisms of alcohol and aldehyde dehydrogenases and risk for esophageal and head and neck cancers. Alcohol. 2005; 35:175-185.
[Crossref] [Google Scholar] [PubMed]