Research Article - Onkologia i Radioterapia ( 2023) Volume 17, Issue 10
Keap1, Estrogen and Progesterone Receptors as Prognostic Factors in Endometrial Carcinoma: An Immunohistochemical study
Aya S Amer, Heba A Mashhour, Hanan A AlShenawy, Hassan Tahseen Sheir and Alaa I Amer*Alaa I Amer, Department of Pathology, Faculty of Medicine, Tanta University, Tanta 31527, Egypt, Email: Alaa.amer@med.tanta.edu.eg
Received: 19-Sep-2023, Manuscript No. OAR-23-114265; Accepted: 20-Oct-2023, Pre QC No. OAR-23-114265 (PQ); Editor assigned: 21-Sep-2023, Pre QC No. OAR-23-114265 (PQ); Reviewed: 08-Oct-2023, QC No. OAR-23-114265 (Q); Revised: 17-Oct-2023, Manuscript No. OAR-23-114265 (R); Published: 25-Oct-2023
Abstract
Background: Endometrial carcinoma is the most common malignancy of the female reproductive system and represents the sixth most prevalent malignant tumour all over the world. The purpose of this study was to evaluate the expression of Keap1, estrogen and progesterone receptors immunoreactivities in a spectrum of endometrial carcinoma cases and correlate Keap1, estrogen and progesterone receptors immunohistochemistry expression in the endometrial carcinoma with available clinicopathological parameters. Paraffin blocks from 70 cases of endometrial carcinoma, 52 cases of endometrioid type and 18 cases of non-endometrioid type were subjected to H&E staining and immunohistochemical staining by Keap1, estrogen and progesterone receptors. Results: Keap1 was expressed as cytoplasmic staining mainly and its expression was related to worse prognosis as it was statistically significant associated with non-endometrioid type, high grade, higher stage, deep myometrial invasion and lymphovascular invasion. Estrogen and progesterone receptors were expressed as nuclear staining. Estrogen and progesterone receptors expression was with good prognosis. Both ER and PR expressions associated significantly with endometrioid type and low grade .ER positive tumours were also associated significantly with; early stage, less myometrial invasion and no lymphovascular invasion while PR receptor status did not exhibit significant relation between its expression, FIGO staging, depth of myometrial invasion and lymphovascular invasion. Conclusions: Keap1 could be a useful prognostic factor in endometrial carcinoma. Keap1 expression is related to non-endometrioid type, high grade, higher stage, deep myometrial invasion and lymphovascular invasion and subsequently is associated with worse prognosis. Both ER and PR expressions were related significantly with endometrioid type and low grade, thus related to good prognosis.
Keywords
keap1, estrogen receptor, progesterone receptor, immunoexpression, endometrial carcinoma
Introduction
Endometrial carcinoma is the most common malignancy of the female genital tract and is the sixth most prevalent malignant tumour all over the world [1]. In women, it accounts for the fourth-prevalent cancer [2]. The incidence rate of endometrial carcinoma is expected to increase more than 50%, regarding to the International Agency for Research on Cancer, worldwide by 2040. The mortality rate due to endometrial carcinoma was the highest between women of low socioeconomic status due to reduced evidence-based care.
A total of 604127 recent cases of endometrial carcinoma were diagnosed with an incidence of 3.1%, according to GLOBOCAN 2020 [3]. Worldwide, endometrial carcinoma is a threat to women health [4].
Endometrioid Endometrial Cancer (EEC) is the most common pathological type of endometrial carcinoma, accounting for 65-85% of all cases [5]. Even though the new guidelines advocate molecular classification for all endometrial carcinomas, particularly high-grade cancers, as a necessary step towards therapy, but when molecular classification measures are unavailable, prognosis could be based on clinicopathological features [6].
Endometrial carcinoma cases are currently treated depending on clinicopathological variables such as histological type, tumour grade and stage, with Lymphatic Vascular Space Invasion (LVSI) [7].
Immunohistochemical markers have a crucial role in the diagnosis, prognosis and treatment of endometrial cancer, in the era of targeted therapy. Therefore, there is always a continuous search for prognostic immunomarkers.
Endometrial cancers are a clinical challenge, despite wide treatment availability. This is mostly because of the development of chemo resistance and radio resistance and the diagnosis at late disease stage. Keap1, (Kelch-like ECHassociated protein 1) is related to tumour resistance to adjuvant treatment [8, 9].
The cellular protein Keap1 was considered as a suppressor of Nrf2 activity and a negative regulator of nuclear erythroid 2-related factor 2/ anti-oxidant response element. (Nrf2/ARE) signalling pathway, that increases the expression of many genes related to cytoprotective mechanisms. Following the discovery of Nrf2/ARE signalling, there is an area of ongoing research in numerous articles on this pathway [10].
Estrogen (E2) and Progesterone (Pg) are significant contributors in the formation and progression of endometrial cancer through specific receptors (ER alpha and PR) [11].
According to several research, the rates of hormone receptor expression vary depending on a variety of criteria, including the proportion of low-grade and high-grade tumours. There is still heterogeneity and publication bias among the research [12, 13].
Immune-Histochemical (IHC) assessment of steroid hormone receptors may be effective as a predictor of cancer behavior in a pre-operative environment and has value as an indicator for adjuvant treatment [13].
The aim of this work was to evaluate the expression of Keap1, estrogen and progesterone receptors immunoreactivities in a spectrum of endometrial carcinoma cases and correlate the immunohistochemical expression of Keap1, estrogen and progesterone receptors with the available clinicopathological parameters in endometrial carcinoma cases.
Methods
Study design
It is a retrospective study of 70 cases diagnosed as endometrial carcinoma collected from Department of Pathology, Faculty of Medicine- Tanta University from November 2019 till January 2021. This study was approved by the Research Ethics Committee (REC), Faculty of Medicine, and Tanta University. (Approval code: 33411/10/19).
Data collection and histopathological evaluation
Patient's age, histopathological diagnosis, grading, myometrial invasion, staging of endometrial carcinoma cases, were obtained from pathology reports. Paraffin blocks with the maximum bulk of tumour were chosen from each case for histopathological studies.
Serial sections (5 µm thickness) were prepared for Hematoxylin and Eosin stain. Stained sections were reviewed to assess the histological diagnosis. Cases were then classified according to the WHO classification of endometrial carcinoma, 2020 [14]. Also, graded according to binary grading system to reduce the inter-observer variation and improve prognostic significance. Tumour stage and myometrial invasion of endometrial carcinoma cases were determined according to FIGO stage of the Uterine Corpus tumours (American Joint Committee on Cancer) [15].
Immunohistochemical staining
The immunohistochemical staining was performed for all 70 cases using DAKO EnVision FLEX Protocol in automated Link 48 DAKO AUTOSTAINER (DAKO/Agilent Corp.). Routine FFPE sections, cut at 3 µm, were collected on positive charged slides.
Immunostaining was done using Keap1 a Rabbit polyclonal antibody (1: 300 dilutions, Biogenix), ER alpha a rabbit monoclonal antibody (Ready to use, Dako/Agilent) and PR a Mouse monoclonal antibody (Ready to use, Dako/ Agilent) were placed on each slide for 30 minutes. The Dako EnVision™ FLEX Detection system was used, without linker antibodies, regarding to standard protocol times as the following: peroxidase blocking reagent: 10 minutes then primary antibodies: 20 minutes; chromogen (diaminobenzidine; DAB): 10 minutes. Slides were flooded with distilled water and counter stain with Mayer’s hematoxylin was done, the slides were washed in tap water, then were covered by Canada balsam [16].
The expression of Keap1, ER and PR were interpreted as follows
The immunohistochemical results for Keap1were recorded based on quantitation of staining performed in cytoplasm. The expression was considered positive when moderate to strong cytoplasmic immunostaining was (>5%) in neoplastic epithelial cells [17]. The immunohistochemistry results for ER and PR expression were recorded based on the percentage of nuclear immunostained cells as the following: The marker expression was evaluated “negative” if the percentage of immunostained cells was <10%; otherwise, the expression was evaluated “positive” if the percentage (10%-100%) [18, 19].
Statistical analysis
Statistical data analysed by SPSS v26 (Statistical Package for Social Sciences) (IBM Inc., Chicago, IL, USA). Quantitative variables were presented as mean and Standard Deviation (SD). Qualitative variables were introduced as frequency and percentage (%) and were analysed by using the Chi-square test or Fisher's exact test. A two tailed p-value ≤ 0.05 was considered significant.
Results
Clinicopathologic characteristics of endometrial carcinoma cases
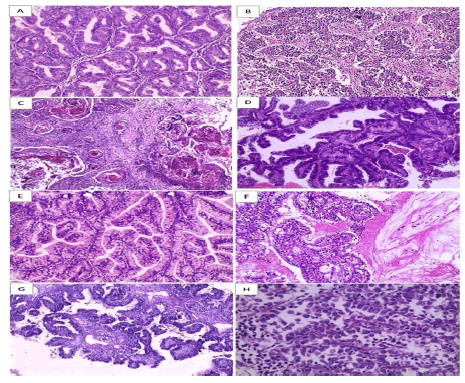
Paraffin blocks from 70 of endometrial carcinoma cases, 52 cases of endometrioid type and 18 cases of nonendometrioid type. The clinicopathological features of the studied cases of endometrial carcinoma are summarized in (Table 1). Age ranged from 35 – 84 years with a mean of 58.5 ± 14.35 years. According to binary grade classification system, studied cases were graded into: Low grade: There were 43 cases including 28 cases were endometrioid adenocarcinoma of usual type, 2 cases was endometrioid adenocarcinoma with squamous differentiation, 7 cases were endometrioid adenocarcinoma with villoglandular pattern, 2 cases were endometrioid adenocarcinoma with secretory pattern, and 4 cases were endometrioid adenocarcinoma with mucinous differentiation. High grade: There were 27 cases including 6 cases were endometrioid adenocarcinoma of usual type, 3 cases were endometrioid adenocarcinoma with squamous differentiation, 11 cases were uterine papillary serous carcinoma, and 7 cases were clear cell endometrial carcinoma. Tumour staging was determined according to FIGO stage of tumours of the Uterine Corp (American Joint Committee on Cancer) 70 cases were categorized into: stage IA was 24 cases representing (34.3%), stage IB was 34 cases representing (48.6%), stage II was 8 cases representing (11.4%), stage IIIA was 3 cases representing (4.3%) and stage IVA was 1 case representing (1.4%). Myometrial invasion ≥ 50% occurred in 46 (65.71%) cases and below 50% in 24 (34.29%) cases. Lymphovascular invasion was detected in 39 (55.7%) cases (Figure 1)
Tab. 1. Clinicopathological features of studied cases of endometrial carcinoma
| Clinicopathological features | Total (70) | (%) | |
|---|---|---|---|
| 1. | Age (Mean SD.) | 58.5 ± 14.35 | |
| 2. | Histopathological type | ||
| Endometrioid type | 52 | (74.29%) | |
| Non Endometrioid type | 18 | (25.71%) | |
| 3. | Grade | - | - |
| High grade | 27 | (38.57%) | |
| Low grade | 43 | (61.43%) | |
| 4. | Tumor staging( FIGO stage) | ||
| LA | 24 | (34.3%) | |
| IB | 34 | (48.6%) | |
| II | 8 | (11.4%) | |
| IIIA | 3 | (4.3%) | |
| IVA | 1 | (1.4%) | |
| 5. | Myometrial invasion | ||
| <50% | 24 | (34.29%) | |
| >50% | 46 | (65.71%) | |
| Vascular invasion | |||
| 6. | Yes | 39 | (55.7%) |
| No | 31 | (44.3%) | |
Figure 1: Histopathological variants of studied cases of endometrial carcinoma (H&E × 200) (A) Low grade endometrioid adenocarcinoma of usual type (B) High grade endometrioid adenocarcinoma of usual type (C) Endometrioid adenocarcinoma with squamous differentiation. (D) Low grade endometrioid adenocarcinoma with villoglandular pattern (E) Endometrioid adenocarcinoma with secretory pattern (F) Endometrioid adenocarcinoma with mucinous differentiation (G) papillary serous carcinoma (H) Clear cell carcinoma
Keap1, ER and PR immunohistochemical results
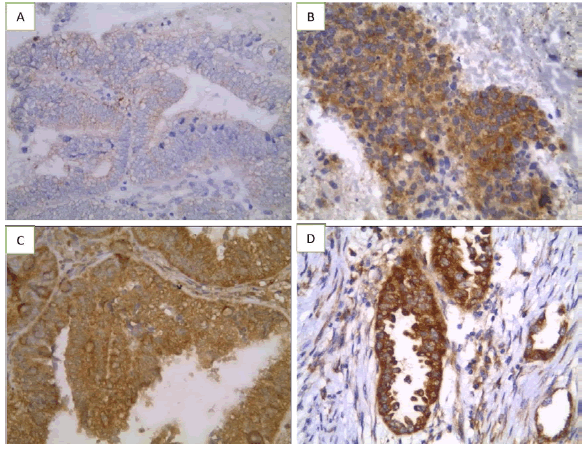
Keap1 was expressed as cytoplasmic staining mainly and its expression was related to worse prognosis as Keap1 immunoexpression was associated with non-endometrioid type as seen in 94.4% and expressed in 81.5% of high-grade endometrial carcinoma cases. Besides, its expression with higher stage (100%) of cases of stage III and IV, deep myometrial invasion representing 71.7% and showed association with lymphovascular invasion representing 74.4% as in Figure 2, Table 2.
Figure 2: Keap1 immunoexpression in studied endometrialcarcinoma cases(×400) (A) Low grade endometrioid adenocarcinoma of usual type showing negative expression (B) High grade endometrioid adenocarcinoma of usual type showing cytoplasmic positive expression (C) Papillary serous carcinoma showing positive cytoplasmic expression (D) Clear cell carcinoma showing positive cytoplasmic expression
Tab. 2. Relation between expressions of Keap1, ER and PR and available histopathologic data
| Variables | Keapl +ve | Keapl -ve | P value | ER +ve | ER -ve | P value | PR +ve | PR-ve | p-value |
|---|---|---|---|---|---|---|---|---|---|
| Histopathological type | |||||||||
| Endometroid (52) | 19 (36.5%) | 33 (63.5%) | <0.001 | 35 (67.3%) | 17 (32.7%) | <0.001 | 38(73.1%) | 14 (26.9%) | <0.001 |
| Non-endometroid (18) | 17(94.4%) | 1 (5.6%) | 2(11.1%) | 16 (88.8%) | 1(5.6%) | 17(94.4%) | |||
| Histopathological grade | |||||||||
| Low-grade (43) | 14(32.6%) | 29 (67.4%) | <0.001 | 31 (72.1%) | 12 (27.9%) | <0.001 | 34(79.1%) | 9 (20.9%) | <0.001 |
| High-grade (27) | 22(81.5%) | (18.5%) | 6(22.2%) | 21 (77.8%) | 5(18.5%) | 22 (81.5%) | |||
| Stage | |||||||||
| LA (24) | 3(12.5%) | 21 (87.5%) | <0.001 | 20 (83.3%) | 4(16.7%) | 0.002 | 15(62.5%) | 9(37.5%) | 0.051 |
| IB (34) | 22(64.7%) | 12(35.5%) | 15(44.4%) | 19 (55.9%) | 22 (64.7%) | 12 (35.3%) | |||
| II (8) | 7(87.5%) | (12.5%) | 2(25.0%) | 6(75.0%) | 1(12.5%) | 7(87.5%) | |||
| IIIA (3) | 3 (100%) | (0.0%) | 0(0.0%) | 3(100%) | 1(33.3%) | 2(66.7%) | |||
| IVA (1) | 1(100%) | 0(0.0%) | 0(0.0%) | 1(100%) | 0(0.0%) | 1(100.0%) | |||
| Myometrial invasion | |||||||||
| <50% (24) | (12.5%) | 21 (87.5%) | <0.001 | 20 (83.3%) | (16.7%) | <0.001 | 15(62.5%) | 9(37.5%) | 0.456 |
| >50% (46) | 33 (71.7%) | 13 (28.3%) | 17 (37.0%) | 29 (63.0%) | 24 (52.2%) | 22 (47.8%) | |||
| Lymphovascular invasion | |||||||||
| Yes (39) | 29(74.4%) | 10(25.6%) | <0.001 | 15 (38.5%) | 24 (61.5%) | 0.009 | 20(51.3%) | 19 (48.7%) | 0.223 |
| No (31) | 7(22.6%) | 24(77.4%) | 22 (71.0%) | 9 (29.0%) | 21 (67.7%) | 10 (32.3%) | |||
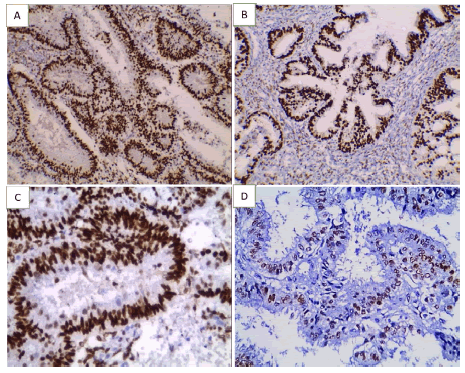
Estrogen and progesterone receptors were expressed as nuclear staining. The Estrogen receptor and PR expressions of endometroid carcinoma in our study encompassed 35 positive cases and 38 positive cases for ER and PR respectively, while all clear cell carcinoma cases showed negative expression for both ER and PR. Regarding serous carcinoma, two cases were positive for ER expression and one case showed positivity to PR expression. ER and PR-negative tumours were more often associated significantly with high-grade. ER and PR immunoexpressions and tumour staging of endometrial carcinoma cases, our study showed positivity of ER and PR expressions in stage I; (35 cases and 37 cases respectively out of 58 cases), while in stage IV negativity for both ER and PR immunoexpressions was found. Myometrial invasion below 50% was associated with positive ER expression (p-value <0.001) and nonsignificantly with PR expression. In the current study, estrogen receptor expression was associated with no lymphovascular invasion, while PR expression was insignificantly associated with lymphovascular invasion as shown in Figure 2 and 3, Table 2.
Figure 3: ER immunoexpression in studied endometrial carcinoma cases ×200 (A) Low grade endometrioid adenocarcinoma of usual type showing positive nuclear expression (B) Low grade endometrioid adenocarcinoma with villoglandular pattern showing positive nuclear expression PR immunoexpression in studied endometrial carcinoma cases ×400 (C) Low grade endometrioid adenocarcinoma of usual type showing positive nuclear expression (D) Low grade endometrioid adenocarcinoma with secretory pattern showing positive nuclear expression
Discussion
Since endometrial cancers are hormone-dependent, immunohistochemistry markers for estrogen and progest erone receptors are frequently used in prognosis. Immuno histoch-emical markers have importance in the prognosis and prediction of treatment efficacy of endometrial cancer in the age of targeted therapy. As a result, searching for prognostic markers is ongoing [20].
There are proteins and molecules that might cause a response to oxidative stress stimuli and are being researched as potential cancer biomarkers not just for diagnosis, but also as prognostic variables and potential target therapy. The NRF2/Keap1 signalling pathway is critical in cell protection from oxidative damage, which is caused by increasing Reactive Oxygen Species (ROS). Stimulation of NRF2/Keap1 signalling in cancer cells, on the other hand, lead to chemoresistance by deactivation of drug mediated oxidative stress with protection of cancer cells from drug induced cell death. Kelch-like ECH-associated protein 1 (Keap1) is an adapter of the Cullin3 (CUL3)-based ubiquitin E3 ligase which promotes NRF2 degradation and suppresses its transcription in the absence of oxidative stress [21].
Although NRF2 ordinarily protects cells from oxidative stress by co-activating genes producing antioxidant proteins, NRF2 accumulation results in chemo/radiotherapy resistance and promotion of malignancy in cancer cells. Chemotherapeutic medicines have little effect on these malignancies. These malignancies are known as NRF2-addicted/NRF2-activated cancers [22].
In the present study, Keap1 immunoexpression was associated with non-endometrioid type as seen in 94.4% and expressed in 81.5% of high-grade endometrial carcinoma cases. Besides, its expression with higher stage (100%) of cases of stage III and IV, deep myometrial invasion representing 71.7% and showed association with lymphovascular invasion representing 74.4%. These results agreed with who stated Keap1 expression in 100% of nonendometrioid type, 87.5% of high-grade cases, 56.8% of cases with deep myometrial invasion and 61% of cases with lymphovascular invasion.
Very limited literature data exists on studying Keap1 expression in endometrial carcinoma cases. When assessing Keap1 expressions on different organs, study by showed significant differences in immunohistochemical expression of Keap1 between different histological types of ovarian carcinoma cases; 108 ovarian carcinomas (serous, mucinous, endometrioid and clear cell) [23]. Patients with serous carcinoma showed highest Keap1 expression.
Regarding the histological grade, found that tumour differentiation of colorectal carcinoma cases was noticed to be significantly associated with decreased expression of Keap1 [24]. Concerning the tumour stage, reported that Keap1 was related to advanced disease and invasion of breast cancer, with increased Keap1 immunoexpression that was linked with a triple-negative subtype [25]. It was concluded that Keap1 may induce cellular proliferation and cancer progression as a transcriptional factor in the Keap-1- Nrf2 signal pathway, which may result in dedifferentiation of malignant cells.
Estrogen and progesterone receptors are key biomarkers for endometrial cancer outcome. ER and PR are members of the steroid superfamily and regulate their action in the endometrium. Binding of the receptors to its ligand results in translocation of the complex to the nucleus, where receptor dimers can bind specific hormone-responsive DNA of target genes [26].
Despite the fact that Endometrial Cancer (EC) has generally favourable outcomes with a low recurrence rate, research for its prognosis is crucial research to plan adjuvant treatment. Hormone receptors for Estrogen (ER) and progesterone are considered promising markers for prognosis of EC [27]. Besides being a predictor for adjuvant treatment, immunohistochemical assessment of hormone receptors may be beneficial in a pre-operative scenario as an indicator for tumour response.
In this study, ER positivity was substantially higher in endometrioid type than in non-endometrioid type. Furthermore, PR expression was much higher in endometrioid type.
These findings are consistent with those of, who found that the positivity of ER or PR in endometrioid type was much greater than in non-endometrioid type, with 92.1% or 91.0% of cases in type 1 being ER and PR positive, respectively [28].
The Estrogen receptor and PR expressions of endometroid carcinoma in our study encompassed 35 positive cases and 38 positive cases for ER and PR respectively, while all clear cell carcinoma cases showed negative expression for both ER and PR.
Regarding serous carcinoma, two cases were positive for ER expression and one case showed positivity to PR expression. Our results are in accordance with the findings of, who reported that out of 56 cases, 24 cases of endometrioid carcinoma were positive for ER and 33 cases were positive for PR. Out of serous carcinoma cases, two cases were PR positive. Clear cell carcinoma was negative for ER and PR expressions [29].
From the results of the current study, ER and PR-negative tumourswere more often associated significantly with highgrade in accordance with. Also, reported that ER/PRpositive tumours were in direct proportion with low grade tumours [30, 31].
Inspecting relation between ER and PR immunoexpressions and tumour staging of endometrial carcinoma cases, our study showed positivity of ER and PR expressions in stage I; (35 cases and 37 cases respectively out of 58 cases), while in stage IV negativity for both ER and PR immunoexpressions was found. Kandaswamy et al. also noticed that higher stage (stage IV) showed loss of ER or PR immunoxpression [1].
In contrary to other studies, Rima found no significant difference in ER expression and staging of the tumour [32]. Furthermore, found an insignificant association between endometrial cancer stages and receptor type. This could be explained by various factors, such as mixed histologic subtypes and varying sampling.
Our study showed that less myometrial invasion was associated with positive ER expression (p-value <0.001) and non-significantly with PR expression and this was similar to study done by [33]. In contrary to our results, documented that ER and PR expression was statistically insignificant with the depth of myometrial invasion, while [34, 35] found significance as regard ER and PR expression and depth of myometrial invasion with (p=0.024), (p=0.048) for ER and PR expression respectively. Estrogen receptor expression was associated with lymphovascular invasion in the current study. This was corresponding to a study conducted by [36].
In the current study, PR expression was insignificantly associated with lymphovascular invasion. In contrast to our results, noticed that progesterone receptor expression was associated with lymph vascular invasion (p-value = 0.022). Reported that lymphovascular invasion was not of prognostic value [37, 38].
To summarize, ER positive tumours were associated with better prognosis: endometrioid type, low grade, early stage, and less myometrial invasion. Our findings agreed with the study by that explained that by it might be related to change in normal enzyme activity, which impact the synthesis of ER and PR in cancer cell. Moreover, PR negative tumours were associated with a poor prognosis: non-endometrioid type and high grade, which is close to. ER and PR expressions are consistent because PR gene transcription is stimulated by estrogen and suppressed by progestin, this is reported by found that loss of ER expression was linked to loss of PR [39].
Conclusion
Keap1 could be a useful prognostic factor in endometrial carcinoma. Keap1 expression is related to nonendometrioid type, high grade, and higher stage, deep myometrial invasion and lymphovascular invasion and subsequently May associated with worse prognosis. Both ER and PR expressions were related significantly with endometrioid type and low grade, thus related to good prognosis.
List of Abbreviations
• ER: Estrogen Receptor
• FIGO: International Federation of Gynaecology and Obstetrics
• Keap1: Kelch-like ECH-associated protein 1
• LVSI: Lymphatic Vascular Space Invasion
• Nrf2/ARE: Nuclear Erythroid 2-Related Factor 2/Antioxidant Response Element
• PR: Progesterone Receptor • ROS: Reactive Oxygen Species
Conflicts of Interest
The authors have no conflict of interest to declare
Funding
The authors did not receive support from any organization for the submitted work.
Availability Of Data And Material
The data used and/or analysed during this study are available on request.
Author's Contributions
ASA: Collection of data, interpretation of the slides (histo pathologic and immunohistochemical staining), designed the figures and tables of this study, analysis of the statistical data, and writing the manuscript. AIA: Participated in interpretation of the slides (histopathologic and immune histochemical staining), analysis of the statistical data, writing the manuscript. HTS: Study design, interpretation of the slides (histopathologic and immune histochemical staining). HAA: Supervision of the study, revision of the draft of the manuscript. HAM: Study design, supervision of the study. All authors read, revised and approved the final version of the manuscript.
References
- S. Kandaswamy, P. Palanisamy. Immunohistochemical Expression of BCL-2 in Endometrial Carcinoma and Its Comparison with Hormone Receptor Status and Epidermal Growth Factor. Cureus. 2023.
[Crossref]
- Yang Z, Yang X, Liu X. Clinical characteristics and prognostic characterization of endometrial carcinoma: a comparative analysis of molecular typing protocol. BMC Cancer. 2023.
- Garg V, Jayaraj, K. L. Novel approaches for treatment of endometrial carcinoma. Curr Probl Cancer. 2022
[Crossref]
- Papadatou V, Tologkos S, Tsolou A, Deftereou TE, Liberis A, et al. CYLD expression in endometrial carcinoma and correlation with clinicohistopathological parameters. Taiwan J Obstet Gynecol. 2022;61:596-600.
- Ren X, Liang J, Zhang Y, Jiang N, Xu Y, et al. Single-cell transcriptomic analysis highlights origin and pathological process of human endometrioid endometrial carcinoma. Nat Commun. 2022;13:6300.
[Crossref]
- Loukovaara M, Pasanen A, Bützow R. Clinicopathologic vs. molecular integrated prognostication of endometrial carcinoma by European guidelines. Cancers. 2022;14:651.
- Li N, Jiang P, Huang Y, Estrogen Receptor-And Progesterone Receptor-Positive Thresholds in Predicting the Recurrence of Early Low-Risk Endometrial Cancer. Clin Med Insights: Oncol. 2022.
- Wong TF, Yoshinaga K, Monma Y, Ito K, Niikura H, et al. Association of keap1 and nrf2 genetic mutations and polymorphisms with endometrioid endometrial adenocarcinoma survival. Int J Gynecol Cancer. 2011.
- G. Tossetta, D. Marzioni. Targeting the NRF2/KEAP1 pathway in cervical and endomerial cancers. Eur J Pharmacol. 2023.
[Crossref]
- Ulasov A. V, Rosenkranz A. A, Georgiev GP. Nrf2/Keap1/ARE signaling: Towards specific regulation. Life Sci. 2022.
[Crossref]
- La Greca A, Bellora N, Le Dily F, Jara R, Nacht AS, et al. Chromatin topology defines estradiol-primed progesterone receptor and PAX2 binding in endometrial cancer cells. Elife. 2022;11:66034.
- Y. Y. Hsiao, H. C. Fu. Quantitative Measurement of Progesterone Receptor Immunohistochemical Expression to Predict Lymph Node Metastasis in Endometrial Cancer. Diagnostics. 2022
[Crossref]
- Srijaipracharoen S, Tangjitgamol S, Tanvanich S, Manusirivithaya S, Khunnarong J, et al. Expression of ER, PR, and Her-2/neu in endometrial cancer: a clinicopathological study. Asian Pac J Cancer Prev. 2010;11:215-220.
- Masood M, Singh N. Endometrial carcinoma: changes to classification (WHO 2020). Diagn Histopathol. 2021;27:493-499.
- VC RR, Jogi P, Bolem O, Gujju E, Sanaboina A. A review on risk factors, staging and survival rates of endometrial cancer in both black and white women in infertility patients in USA. World J Curr Med Pharm Res. 2020:152-156.
- Colley EC, Stead RH. Optimized Immunohistochemistry Workflow Facilitated by New Dako Autostainer Link 48 Software.
- Ahtikoski AM, Kangas J, Salonen R, Puistola U, Karihtala P. Cytoplasmic Keap1 expression is associated with poor prognosis in endometrial cancer. Anticancer Res. 2019;39:585-590.
- Raffone A, Travaglino A, Saccone G, Mollo A, De Placido G. Should progesterone and estrogen receptors be assessed for predicting the response to conservative treatment of endometrial hyperplasia and cancer? A systematic review and metaâ?analysis. Acta Obstetricia et Gynecologica Scandinavica. 2019;98:976-987.
- Jia M, Jiang P, Huang Z, Hu J, Deng Y. The combined ratio of estrogen, progesterone, Kiâ?67, and P53 to predict the recurrence of endometrial cancer. J surg oncol. 2020;122:1808-1814.
- M. Koskas, F. Amant, M. R. Mirza. Cancer of the corpus uteri: update. Int J Gynecol Obstet. 2021.
[Crossref]
- Uruno A, Yamamoto M. The KEAP1-NRF2 system and neurodegenerative diseases. Antioxid redox signal 2023;38:974-988.
- Panda H, Wen H, Suzuki M, Yamamoto M. Multifaceted Roles of the KEAP1–NRF2 System in Cancer and Inflammatory Disease Milieu. Antioxidants. 2022;11:538.
- Liew PL, Hsu CS, Liu WM, Lee YC, Lee YC. Prognostic and predictive values of Nrf2, Keap1, p16 and E-cadherin expression in ovarian epithelial carcinoma. Int j clin exp pathol. 2015;8:5642.
- Huang P, Wang S, Wu Z, Zhou Z, Kuang M, et al. Correlations of ALD, Keap-1, and FoxO4 expression with traditional tumor markers and clinicopathological characteristics in colorectal carcinoma. Medicine. 2022;101.
- Karihtala P, Kauppila S, Soini Y. Oxidative stress and counteracting mechanisms in hormone receptor positive, triple-negative and basal-like breast carcinomas. BMC cancer. 2011;11:1-6.
- W. J. Van Weelden, C. Reijnen, Küsters. The cutoff for estrogen and progesterone receptor expression in endometrial cancer revisited: A European Network for Individualized Treatment of Endometrial Cancer collaboration study. Hum Pathol. 2021.
[Crossref]
- Bounous V. E, Ferrero A, Campisi P. Immunohistochemical Markers and TILs Evaluation for Endometrial Carcinoma. J Clin Med. 2022.
[Crossref]
- Shen F, Gao Y, Ding J, Chen Q. Is the positivity of estrogen receptor or progesterone receptor different between type 1 and type 2 endometrial cancer? Oncotarget. 2017;8:506.
- Waqar S, Khan SA, Sarfraz T, Waqar S. Expression of Estrogen Receptors (ER), Progesterone Receptors (PR) and HER-2/neu receptors in Endometrial Carcinoma and their associations with histological types, grades and stages of the tumor. Pak J Med Sci. 2018;34:266.
- Shivkumar VB, Atram MA, Gangane NM. Expression of ER/PR receptor, Her-2/neu, Ki67 and p53 in endometrial carcinoma: clinicopathological implication and prognostic value. Indian J Gynecol Oncol. 2020;18:1-9.
- Drocas I, Craitoiu S, Stepan AE, Iliescu DG, Drocas IA. The analysis of hormonal status and vascular and cell proliferation in endometrioid endometrial adenocarcinomas. Rom J Morphol Embryol. 2022;63:113.
- Greca AL, Bellora N, Dily FL, Jara R, Silvina Nacht A, et al. Chromatin topology defines estradiol-primed progesterone receptor and PAX2 binding in endometrial cancer cells.
- Wang C, Tran DA, Fu MZ, Chen W, Fu SW. Estrogen receptor, progesterone receptor, and HER2 receptor markers in endometrial cancer. J Cancer. 2020;11:1693.
- Salama A, Arafa M, ElZahaf E, Shebl AM, Awad AA, et al. Potential role for a panel of immunohistochemical markers in the management of endometrial carcinoma. J Pathol Transl Med. 2019;53:164-172.
- Weinberger V, Bednarikova M, Hausnerová, J. A novel approach to preoperative risk stratification in endometrial cancer: the added value of immunohistochemical markers. Front Oncol. 2019.
[Crossref]
- Van Der Putten LJ, Visser NC, Van de Vijver K, et al. Added value of estrogen receptor, progesterone receptor, and L1 cell adhesion molecule expression to histology-based endometrial carcinoma recurrence prediction models: an ENITEC collaboration study. Int J Gynecol Cancer. 2018;28.
- Jiang XF, Tang QL, Li HG, Shen XM, Luo X, et al. Tumorâ?associated macrophages correlate with progesterone receptor loss in endometrial endometrioid adenocarcinoma. J Obstet Gynaecol Res. 2013;39:855-863.
- Huvila J, Talve L, Carpén O, Edqvist PH, Pontén F, et al. Progesterone receptor negativity is an independent risk factor for relapse in patients with early stage endometrioid endometrial adenocarcinoma. Gynecol oncol. 2013;130:463-469.
- Park JY, Hong DG, Chong GO, Park JY. Tumor budding is a valuable diagnostic parameter in prediction of disease progression of endometrial endometrioid carcinoma. Pathol Oncol Res. 2019;25:723-730.