Research Article - Onkologia i Radioterapia ( 2021) Volume 15, Issue 7
Intensity-modulated radiotherapy with cisplatin or cetuximab in patients undergoing chemoradiation for squamous cell carcinoma head and neck
Shruthi Venkateshulu1* and Kiran Kumar BR22Department of Radiation Oncology, Bangalore Medical College and Research Institute, Bangalore, India
Shruthi Venkateshulu, Department of Radiation Oncology, VTSM Peripheral Cancer Centre, Kalaburgi, India, Email: dr.v.shruthi@gmail.com
Received: 08-Jul-2021 Accepted: 22-Jul-2021 Published: 26-Jul-2021
Abstract
To study the efficacy, response of weekly cetuximab used concurrently with radiotherapy versus weekly cisplatin with concurrent radiotherapy in loco regionally advanced Head and Neck Squamous Cell Carcinoma (HNSCC). Materials and Methods: Fifty eligible patients were randomly assigned to study arm (Cetuximab arm) and control arm (Cisplatin arm) to receive either intravenous cetuximab 400 mg/m2 or weekly intravenous cisplatin 40 mg/ m2, during RT. RT dose received was 70Gy in 35 fractions with 2Gy/fraction in both the arms. Treatment evaluation was done by assessing tumor response after 6 months follow up period after completion of chemo radiotherapy. Results: Overall Response (OR) rates, which include Complete response (CR) and Partial Response (PR), were assessed in all the treated patients of locally advanced HNSCC at 6 months after completion of treatment. 76% overall response was achieved (CR-56%+PR-20%) in cetuximab arm and 80%(CR52%+PR-28%) in Cisplatin arm (p=1.000). Conclusion: Weekly Cisplatin shows slightly better results in terms of overall response comparing to weekly Cetuximab in our study. Large sample size and longer duration of follow up are needed for strong evaluation of efficacy, to draw inferences on Loco regional Control (LRC) Disease Free Survival (DFS) and Overall Survival (OS).
Keywords
cetuximab, cisplatin, HNSCC, head and neck cancer, chemotherapy.
Introduction
Head and Neck Squamous Cell Cancer (HNSCC) is the seventh most common malignancy worldwide, with a global incidence of. 800,000 new cases annually [1]. In India, it constitutes almost one third of all cancer cases [2]. Mortality in India due to head and neck cancer is at least half the incidence due to its late presentation for treatment (stage III- 39%, stage IV-23%) [3]. Surgery combined with Radiotherapy (RT) with or without chemotherapy is the preferred treatment in loco regionally advanced HNSCC. However, because of unrespectable disease, and because of an ambition to preserve affected organs and their function, definitive RT often remains the treatment of choice [4]. The established standard treatment for patients, which are unsuitable for surgical treatment, is concurrent systemic therapy with RT. There are currently 2 common treatment strategies supported by guidelines, concurrent platinum, or cetuximab with RT. Platinum based chemo radiotherapy is the standard of care for locally advanced HNSCC in many countries, and concurrent high-dose cisplatin is the preferred systemic agent which is used most widely in the world.
Many large phase 3 trials and meta-analysis have shown that concurrent Cisplatin with Radiotherapy (CRT) improves Overall Survival (OS) compared with RT alone. However, CRT leads to numerous toxicities. [5-7] According to updated Meta-Analysis of Chemotherapy in Head and Neck Cancer (MACH-NC) met analysis, addition of concomitant cisplatin to RT improves outcome, with an absolute gain in Overall Survival(OS) of 6.5% at 5 years [8].Cisplatin administered intravenously at a dose of 100 mg/m2 every third week is the most established regimen, although several other schedules, mainly weekly low-dose regimens, have been reported [9-11].
As patients may might develop serious toxicity of cisplatin that could affect their quality of life. Radiotherapy can induce the expression of Epidermal Growth Factor Receptor (EGFR) in HNSCC, leading to acquired resistance [12]. Cetuximab, a monoclonal antibody that targets the EGFR, is the first targeted treatment that shows therapeutic efficacy in HNSCC and may help to overcome this resistance. Cetuximab has been approved by the Food and Drug Administration Agency (FDA), for use in patients with locally advanced HNSCC [13]. Similarly, some phase 3 trials have demonstrated that concurrent cetuximab with radiotherapy improves OS, LRC, and the quality of life compared with RT alone[14-15]. Updated results with 5-year survival reported by Bonner et al. showed that RT combined with the Epidermal Growth Factor Receptor (EGFR) antibody cetuximab rendered an absolute benefit of 9.2% (5-year survival of 45.6% in the cetuximab plus RT group versus 36.4% in the radiotherapy alone group) which is similar to that of concomitant cisplatin [15]. Cetuximab has been increasingly used to treat patients who concern about the toxicity of platinum chemotherapy, such as elderly or frail patients. Cetuximab appears to have less toxicity than high dose cisplatin. In the phase III study of cetuximab and radiotherapy for locally advanced non-operative HNSCC, 93% of patients received the prescribed cetuximab dose, which compares very favourably to the compliance rate of high dose cisplatin in RTOG 95-01 (61%) [16].
Recently, 2 randomized studies showed that Cetuximab with radiotherapy was inferior to Cisplatin with Radiotherapy for patients with Human Papillomavirus (HPV) positive (+) or pharyngeal carcinoma [17-18] However, except for its highly selected group, there are no robust trials with direct comparison of efficacy of weekly cetuximab against cisplatin with concurrent radiotherapy in locally advanced HNSCC. Most of the data published in various journals are of retrospective studies,which comprise the majority in HNSCC. Furthermore, several studies suggest that EGFR inhibition might be more effective in HPV Negative disease than in HPV positive disease [19-20]. Hence, we conducted a prospective study comparing efficacy of the Cetuximab versus Cisplatin in the treatment of Locally advanced HNSCC.
Materials and Methods
It was a hospital based prospective randomized comparative study. Patients aged above 18 years with previously untreated locally advanced HNSCC of the stage III-IV according to American Joint Committee on Cancer Staging (AJCC) TNM classification, 7th edition, without distant metastases, Performance status of 0-2 according to ECOG and aimed for curative treatment with definitive RT were eligible for study. Patients previously treated with surgery, chemotherapy or radiotherapy and Patients with severe cardiac illness, previous malignancies, poor performance status (ECOG 3 and 4), pregnancy and lactating females were excluded.
Fifty biopsy proven cases of locally advanced HNSCC were taken into the study, after taking the written informed consent. The procedure for staging included a detailed history and a physical examination, as well as common laboratory tests and standard chest radiographs, Ultrasonography abdomen, Echocardiogram (ECG), complete ENT evaluation, CECT head and neck. All the patients were randomized to study arm (Cetuximab) and Control arm (Cisplatin) with 25 patients in each arm.
Both the arms were treated with a Definitive RT delivered by Linear Accelerator with Intensity Modulated Radiotherapy (IMRT) with a dose of 70Gy in 35 Fractions, 5 days in a week with 2Gy per fraction. Control arm received concurrent Cisplatin 40 mg/m2 intravenously 1-hour infusion with full hydration and supportive medications 4 hours-6 hours before radiation, repeated weekly for 5 cycles. Study arm received concurrent cetuximab 400 mg/m2 as loading dose over 120 minutes infusion, one week prior to radiotherapy followed by weekly dose of 250mg/m2 intravenously 1-hour infusion with prior premedication. Treatment evaluation was done by assessing tumour response according to Response Evaluation Criteria in Solid Tumours (RECIST) version 1 [21]. After 6 months follow up period after completion of chemo radiotherapy.
Data analysis was performed with Statistical Package for Social Sciences (SPSS) version 21.0 statistical package. We have used Chi-Square test for the analysis. The probability value p<0.05 was considered as significant (Table 1).
| Gender | Control arm(N=25) | Study arm(N=25) | ||
|---|---|---|---|---|
| No. | % | No. | % | |
| Male | 24 | 96 | 23 | 92 |
| Female | 1 | 4 | 2 | 8 |
| Primary site | ||||
| Oral cavity | 1 | 4 | 2 | 8 |
| Oropharynx | 13 | 52 | 11 | 44 |
| Hypopharynx | 4 | 16 | 6 | 24 |
| Larynx | 7 | 28 | 5 | 20 |
| Nasopharynx | 0 | 0 | 1 | 4 |
| Tumour stage | ||||
| T1 | 5 20 |
0 | 0 | |
| T2 | 16 | 64 | 14 | 56 |
| T3 | 3 | 12 | 9 | 36 |
| T4 | 1 | 4 | 2 | 8 |
| Nodal Stage | ||||
| N0 | 3 | 12 | 3 | 12 |
| N1 | 3 | 12 | 5 | 20 |
| N2 | 19 | 76 | 17 | 68 |
| N3 | 0 | 0 | 0 | 0 |
| AJCC Stage | ||||
| III | 5 | 20 | 7 | 28 |
| IVA | 20 | 80 | 18 | 72 |
Tab. 1. Patient and Tumour characteristics
Results
Fifty patients with locally advanced HNSCC (AJCC 7th edition classification stage III/IV) were entered into the study. The patients were divided into two groups Study arm and Control arm with 25 patients in each group by online computergenerated randomization.The commonest histopathology was moderately differentiated squamous cell carcinoma, with 20 patients in the study arm and 19 Patients in the control group. Twelve patients (7 in study arm and 5 in control arm) were of stage III and thirty-eight patients (18 in study group and 20 in control arm) were of stage IVA according to AJCC staging system. Fourty four patients had ECOG performance status 0 (21 in study group and 23 in control group) and six patients had ECOG performance status 1 (4 in study group and 2 in control group) (Table 2).
| ECOG-Performance Status | Control group (N=25) | Study group (N=25) | ||
|---|---|---|---|---|
| No | % | No | % | |
| 0 | 23 | 92 | 21 | 84 |
| 1 | 2 | 8 | 4 | 16 |
| Histology | ||||
| Well Differentiated Squamous | 4 | 16 | 3 | 12 |
| Cell Carcinoma | ||||
| Moderately Differentiated Squamous | 19 | 76 | 20 | 80 |
| Cell Carcinoma | ||||
| Poorly Differentiated Squamous | 2 | 8 | 2 | 8 |
| Cell Carcinoma | ||||
Tab. 2. Performance Status and Histology Distribution of Patients
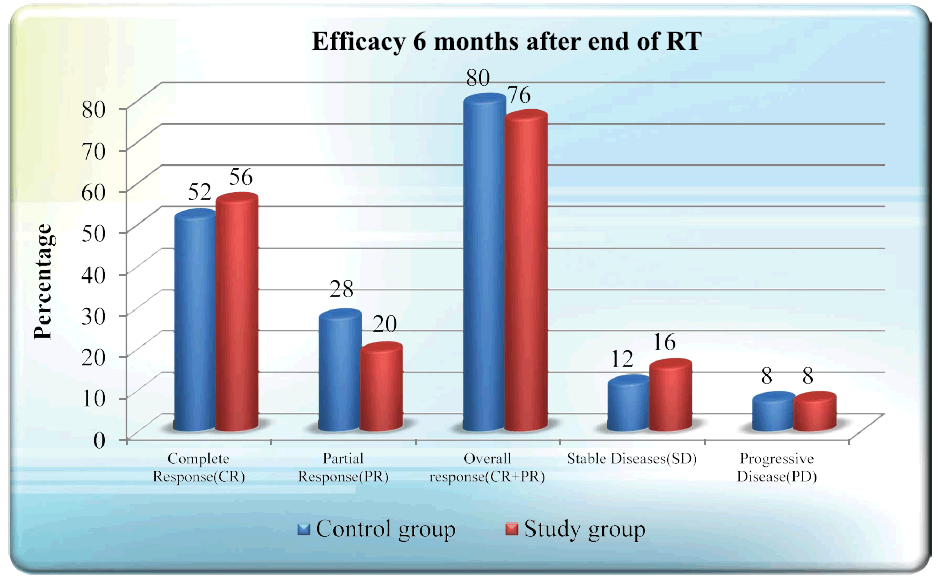
The clinical response rates obtained after 6 months of treatment follow up revealed that Complete Response (CR) was achieved in 14 patients (56%) in the study arm and 13 patients (52%) in the control group. Overall Response (OR) rates (CR+PR) were 76% in the study group and 80% in the arm. Though the CR and OR rates are slightly more in the control group as compared to that of the study group the results are statistically not significant.In the study group 2 patients had Progressive Disease (PD) and 4 patients had Stable Disease (SD). In the control group 2 patients had Progressive Disease (PD) and 3 patients had Stable Disease (SD) during the 6 months follow up period. (Table 3, Figure 1) The Complete Response Rates according to Tumour and Nodal Staging are mentioned in Table 4.
| Disease Response | Control group | Study group | ||
|---|---|---|---|---|
| No | % | No | % | |
| Complete Response (CR) | 13 | 52 | 14 | 56 |
| Partial Response (PR) | 7 | 28 | 5 | 20 |
| Overall response (CR+PR) | 20 | 80 | 19 | 76 |
| Stable Diseases (SD) | 3 | 12 | 4 | 16 |
| Progressive Disease (PD) | 2 | 8 | 2 | 8 |
| Chi-square =0.444 with 3 degrees of freedom; p=1.000 | ||||
Tab. 3. Efficacy 6 months after end of RT
Figure 1: Efficacy 6 months after end of RT
| Tumour and | No. of Complete Response | |||||
|---|---|---|---|---|---|---|
| Nodal Stage | Control group | Study group | ||||
| Total | CR | % | Total | CR | % | |
| T1 | 5 | 3 | 60 | 0 | 0 | 0 |
| T2 | 16 | 8 | 50 | 14 | 10 | 71.42 |
| T3 | 3 | 1 | 33.3 | 9 | 3 | 33.33 |
| T4 | 1 | 1 | 100 | 2 | 1 | 50 |
| N0 | 3 | 2 | 40 | 3 | 3 | 100 |
| N1 | 3 | 3 | 100 | 5 | 4 | 80 |
| N2 | 19 | 8 | 42.1 | 17 | 7 | 41.17 |
| N3 | 0 | 0 | 0 | 0 | 0 | 0 |
Tab. 4. Complete response according to tumour and nodal stage
Discussion
The study was intended to compare anti-EGFR monoclonal antibody cetuximab with concurrent radiotherapy (Bioradiotherapy- BRT) versus concurrent chemo-radiation with weekly cisplatin in loco-regionally advanced HNSCC. In this study we have shown that weekly cetuximab with concurrent radiotherapy is a promising and treatment regimen which is comparable to standard chemotherapy regimen Cisplatin in terms of overall response.
CRT and BRT are both the standard of treatment for patients with inoperable locally advanced HNSCC. Since there were no randomized phase 3 trials to compare these two strategies for a long time, the opinion that BRT was comparable to CRT has been challenged all the time. Some clinical studies and metanalyses that have addressed this issue have conflicting conclusions. Fausto et al. conducted a meta-analysis to evaluate the efficacy of platinum-based chemo radiotherapy compared with cetuximab-based bio-radiotherapy in locally advanced HNSCC [22] and they found cisplatin had better OS and PFS. However, the risk ratio was defined as the primary measurement of treatment outcome in this study, but the outcome of time However, the risk ratio was defined as the primary measurement of treatment outcome in this study, but the outcome of timeto event was not considered. A meta-analysis by Huang et al. observed a better OS in patients with HPV (+) HNSCC and better PFS in patients.
With or pharyngeal cancer treated with BRT than in patients treated with CRT [23].
A study on outcomes of cetuximab concurrent with radiotherapy in advanced HNC unsuitable for platinum-based chemoradiotherapy was carried out by JP Agarwal [24]. 37 patients were included in the study, median age of the patients was 59 years. Thirty-four (92%) patients had advanced stage disease (stage III-IV). At a median follow-up of 16 months, the 2-year LRC, DFS and OS was 35.5%, 29.5%, and 44.4% respectively. They concluded that Cetuximab concurrent with radiotherapy is a reasonable alternative in advanced head-neck cancer patients with acceptable compliance and outcomes, but with higher skin toxicity.
In Our study, in the cetuximab arm OR is 76% (19 patients), which is comparable to the results in a study by S Dattatreya et al. [25] in which OR rate is 68.42% (13/19 patients). CR rate of 56% is comparable to the results obtained in a study by JP Agarwal [24], in which the CR rate is 47% (14/30 patients). In cisplatin arm OR is 80% (22 patients), CR 52% (15 patients) and PR 28% (7 patients), which is comparable to theresults in a study by Lone M Maqbool et al. [26], with an overall response rate of 88.8% (40 patients), complete response rate of 57.7% (26 patients), partial response rate of 31.1% (14 patients), the response is also comparable to the results in a study by Dimriet al. [27], with complete responses at the primary site, regional nodes and overall diseasein 86%, 89% and 83% patients respectively.
A meta-analysis conducted by Wen-Hua Tang et al. on Concurrent cisplatin or cetuximab with radiotherapy in patients with locally advanced head and neck squamous cell carcinoma. The primary outcome that was Overall Survival (OS), whereas the secondary outcomes were Progression-Free-Survival (PFS), Locoregional Control (LRC), and Distant Metastasis-Free Survival (DMFS). Twenty-three studies, with a total of 8701 patients, were considered eligible and included in this metaanalysis. Their results revealed that patients treated with CRT (Chemoradiotherapy) had longer OS (HR=0.51, 95%CI, 0.41- 0.64, p<.001), PFS (HR=0.37, 95%CI, 0.23-0.60, p<.001), LRC (HR=0.46, 95%CI, 0.37-0.57, p<.001) than those treated with BRT (Bio Radiotherapy).They concluded that CRT had better OS, PFS, LRC, and DMFS in locally advanced HNSCC than BRT. Thus, concurrent cisplatin should remain the standard of treatment for patients in this setting. Concurrent cetuximab may still be administered to patients who cannot tolerate cisplatin [28].
There are no robust trials for long time with head to head comparison of efficacy of weekly cetuximab against cisplatin with concurrent radiotherapy in locally advanced HNSCC. Most of the data published in various journals are of retrospective studies. So, we are unable to compare the results of our study with any of large prospective studies where there is a direct comparison between the two regimens with respect to response assessment. But in our study the overall response rates are slightly better towards Cisplatin arm, though the OR rates of 80% is achieved in cisplatin arm, whereas OR in the cetuximab arm is 76%, the results are statistically not significant. (p=1.00).
As the sample size was less in our study, and the follow up period of only six months and these are preliminary results, larger prospective randomized studies with longer duration of follow up with direct comparison of both the regimens are needed for strong evaluation of efficacy and to draw inferences about the late toxicities and also LRC, DFS and OS, which are both the standard of care in locally advanced HNSCC in the current era.
Conclusion
Weekly Cisplatin shows slightly better results in terms of overall response comparing to weekly Cetuximab in our study with this regimen in locally advanced HNSCC. Although follow up period is short, loco-regional control rates are impressive with both the treatments. Larger prospective randomized studies with longer duration of follow up with direct comparison of both the regimens are needed for strong evaluation of efficacy.
Conflicts of Interest
Nil.
References
- Ferlay J, Ervik M, Lam F, Colombet M, Mery L, et al: Cancer Today (powered by GLOBOCAN 2018). Lyon, France, 2018.
- Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, et al. Global Burden of Disease Cancer Collaboration, Fitzmaurice C, Allen C, et al. Global, Regional, and National Cancer Incidence, Mortality, Years of Life Lost, Years Lived with Disability, and Disability-Adjusted Life-years for 32 Cancer Groups, 1990 to 2015. A Systematic Analysis for the Global Burden of Disease Study. JAMA Oncol. 2017;3:524-548.
- Mishra A, Singh VP, Verma V.Environmental effects on head and neck cancer in India. J ClinOncol. 2009;27:e17059.
- Colevas AD, Yom SS, Pfister DG, Spencer S, Adelstein D,et al. NCCN guidelines insights: head and neck cancers, version 1.2018. J NatlComprCancNetw. 2018;16:479-490.
- Cooper JS, Pajak TF, Forastiere AA, Jacobs J, Campbell BH, et al. Postoperative concurrent radiotherapy and chemotherapy for high-risk squamous-cell carcinoma of the head and neck. N Engl J Med. 2004;350:1937-1944.
- Pignon JP, leMaitre A, Maillard E, Bourhis J, MACH-NC Collaborative Group. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): an update on 93 randomised trials and 17,346 patients. RadiotherOncol 2009;92:4-14.
- Adelstein DJ, Li Y, Adams GL, Wagner H, Kish JA, et al. An intergroup phase III comparison of standard radiation therapy and two schedules of concurrent chemoradiotherapy in patients with unresectable squamous cell head and neck cancer. J ClinOncol. 2003;21:92-98.
- BlanchardP, Landais C, Landais C, Petit C, Hackshaw A, et al. Meta-analysis of chemotherapy in head and neck cancer (MACH-NC): An update on 100 randomized trials and 19,248 patients, on behalf of MACH-NC group. Ann Oncol. 2016;27:vi328-vi350.
- Ho KF, Swindell R, Brammer CV. Dose intensity comparison between weekly and 3-weekly Cisplatin delivered concurrently with radical radiotherapy for head and neck cancer: A retrospective comparison from New Cross Hospital, Wolverhampton, UK. ActaOncol. 2008;47:1513-1518.
- Lau H, Brar S, Hao D, MacKinnon J, Yee D, et al. Concomitant low-dose cisplatin and three-dimensional conformal radiotherapy for locally advanced squamous cell carcinoma of the head and neck: Analysis of survival and toxicity. Head Neck. 2006;28:189-196.
- Rutten H, Pop LA, Janssens GO, Takes RP, Knuijt S, et al. Long-term outcome and morbidity after treatment with accelerated radiotherapy and weekly cisplatin for locally advanced head-and-neck cancer: Results of a multidisciplinary late morbidity clinic. Int J RadiatOncolBiol Phys. 2011;81:923-929.
- Dent P, Reardon DB, Park JS, Bowers G, Logsdon, et al. Radiation-induced release of transforming growth factor alpha activates the epidermal growth factor receptor and mitogen-activated protein kinase pathway in carcinoma cells, leading to increased proliferation and protection from radiation induced cell death. MolBiol Cell. 1999;10:2493-2506.
- https://www.medscape.org/cetuximab
- Bonner JA, Harari PM, Giralt J, Azarnia N, Shin DM, et al. Radiotherapy plus cetuximab for squamous-cell carcinoma of the head and neck. N Engl J Med. 2006;354:567-578.
- Bonner JA, Harari PM, Giralt J, Cohen RB, Jones CU, et al. Radiotherapy plus cetuximab for locoregionally advanced head and neck cancer: 5-year survival data from a phase 3 randomised trial, and relation between cetuximab-induced rash and survival. Lancet Oncol. 2010;11:21-28.
- Cooper JS,Zhang Q, Pajak TF, Forastiere A, Jacobs J, et al. Long-term follow-up of the RTOG 9501/intergroup phase III trial: postoperative concurrent radiation therapy and chemotherapy in high-risk squamous cell carcinoma of the head and neck. Int J RadiatOncolBiol Phys. 2012;84:1198-1205.
- Mehanna H, Robinson M, Hartley A, Kong A, Foran B, et al. Radiotherapy plus cisplatin or cetuximab in low-risk human papillomavirus-positive oropharyngeal cancer (De-ESCALaTE HPV): an open-label randomised controlled phase 3 trial. Lancet. 2019;393:51-60.
- Gillison ML, Trotti AM, Harris J, Eisbruch A, Harari HM, et al. Radiotherapy plus cetuximab or cisplatin in human papillomavirus-positive oropharyngeal cancer (NRG Oncology RTOG 1016): a randomised, multicentre, non-inferiority trial. Lancet. 2019;393:40-50.
- Vermorken JB, Stohlmacher-Williams J, Davidenko I,Licitra L, Winquist E,et al. Cisplatin and fluorouracil with or without panitumumab in patients with recurrent or metastatic squamous-cell carcinoma of the head and neck (SPECTRUM): an open-label phase 3 randomised trial. Lancet Oncol. 2013;14:697-710.
- Fayette J, Wirth L, Oprean C, Udrea A, Jimano A, et al. Randomized phase II study of duligotuzumab (MEHD7945A) vs. cetuximab in squamous cell carcinoma of the head and neck (MEHGAN Study). Front Oncol. 2016;6:232.
- Eisenhauer EA, Therasse P, Bogaerts J, Schwartz LH, Sargent D, et al. New response evaluation criteria in solid tumours: revised RECIST guideline (version 1.1).Eur J Cancer. 2009;45:228-247.
- Petrelli F, Coinu A, Riboldi V, Borgonov, GhilardiM,et al. Concomitant platinum-based chemotherapy or cetuximab with radiotherapy for locally advanced head and neck cancer: a systematic review and meta-analysis of published studies. Oral Oncol. 2014;50:1041-1048.
- Huang J, Zhang J, Shi C, Liu L, Wei Y,et al. Survival, recurrence and toxicity of HNSCC in comparison of a radiotherapy combination with cisplatin versus cetuximab: a meta-analysis. BMC Cancer. 2016;16:689.
- Agarwal JP, Budrukkar A, Chaturvedi P, Kalyani N, Laskar G, et al. Mini Symposium Cetuximab with radiotherapy in patients with loco-regionally advanced squamous cell carcinoma of head and neck unsuitable or ineligible for concurrent platinum-based chemo-radiotherapy: Ready for routine clinical practice? Indian J Cancer. 2011;48:148-153.
- S Dattatreya, C Goswami.Mini Symposium Cetuximab plus radiotherapy in patients with unresectable locally advanced squamous cell carcinoma of head and neck region - A open labelled single arm phase II study. Indian J Cancer. 2011;48:154-157.
- Maqbool LM, Malik TR, Afroza F, Khan NA. Treatment Outcome with Weekly Cisplatin Concurrent with Radiation Therapy in Locally Advanced Head and Neck Squamous Cell Carcinoma. J Int Med Sci Acad. 2012;25:3159.
- Dimri K, Pandey AK, Trehan R, Rai B, Kumar A, Conventional radiotherapy with concurrent weekly Cisplatin in locally advanced head and neck cancers of squamous cell origin-a single institution experience. Asian Pac J Cancer Prev. 2013;14:6883-6888.
- Wen-Hua Tang, Wei Sun, Guo-Xian Long. Concurrent cisplatin or cetuximab withradiotherapy in patients with locally advanced head and neck squamous cell carcinoma. A meta-analysis. Medicine. 2020;99:36(e21785).