Research Article - Onkologia i Radioterapia ( 2020) Volume 14, Issue 6
Hemostasis irradiation for gastric cancer: comparison of whole and partial stomach irradiation in the liver and kidney
Osamu Tanaka1*, Kousei Ono1, Takuya Taniguchi1, Naoyuki Sakamoto2, Takuji Kiryu1, Chiyoko Makita3 and Masayuki Matsuo32Asahi University Hospital, Department of Gastroenterology, Gifu, Japan
3Gifu University Hospital, Department of Radiology, Gifu, Japan
Osamu Tanaka, Department of Radiation Oncology, Asahi University Hospital 3-23 Hashimoto-cho, Gifu City, Gifu, 500-8523, Japan, Tel: +81-58-253-8001, Fax: +81-58-253-5165, Email: c.bluered@gmail.com
Received: 06-Nov-2020 Accepted: 25-Nov-2020 Published: 02-Dec-2020
Abstract
Treatment methods for advanced gastric cancer includes haemostatic radiotherapy. The usefulness of additional irradiation when re-bleeding after haemostatic irradiation has been reported. In this study, we examined the irradiation doses to the liver and kidney using partial gastric irradiation of the upper stomach and lower stomach using whole stomach irradiation as a control. Among 20 cases, the whole stomach is set as the target control. Partial stomachs were divided into 2 groups: upper stomach and lower stomach. The dose to each three PTV was set to 20 Gy/5 fractions. Whole liver and bilateral kidneys are organ at risk. The liver D5 [Dx; Dose (Gy) irradiated to x % of liver and bilateral kidney’s volume], D10, D15, D20, D30 and the kidneys D10, D15, D20, D25 were evaluated. The upper stomach was higher than the lower stomach in the high dose range of livers D20, D25, and D30. On the other hand, there was no difference between the upper and lower stomachs of livers D5, D10, and D15 in the low dose range. Renal dose was mostly significantly lower in all dose–volume histograms in the upper stomach than in the lower stomach. However, in kidney D25, the lower stomach was not significantly different from the whole stomach. In conclusion, radiation to the liver and kidneys can be reduced by partial gastric irradiation. Partial gastric irradiation is recommended if the location of the tumour can be identified using computed tomography.
Keywords
gastric malignancy, abdominal irradiation, liver dose, kidney dose
Introduction
Treatment methods for advanced gastric cancer include perioperative radiation chemotherapy and hemostatic radiation [1-5]. Perioperative irradiation is about 50 Gy, radical dose for malignant lymphoma (diffuse large B cell lymphoma, Mucosa associated lymphoid tissue) is about 24 to 36 Gy. However, hemostatic irradiation is about 20 Gy and hemostasis is often effective [6-8]. In recent years, the usefulness of additional irradiation when re-bleeding after hemostatic irradiation has been reported. In that case, it is necessary to set the dose to be irradiated to the liver and kidney by the first hemostatic irradiation for re-irradiation as low as possible. If the invasion range of gastric cancer is unclear by CT, it is necessary to irradiate the target with a wide area (whole stomach). However, if the location and progression of the tumor can be recognized by CT, it is possible to irradiate only the partial stomach, and it may be possible to reserve a liver and renal function during re-irradiation [9].
At present, when performing hemostatic irradiation for gastric cancer, it remains controversial whether to irradiate the whole stomach or only a part of the stomach. We have been irradiating the whole stomach with 20 Gy/5 fractions and have reported the usefulness of re-irradiation with 15 Gy/5 fractions for the partial stomach [6-8]. Because the use of re-irradiation will spread in the future, it is important to determine the first irradiation field in consideration of the tolerable dose to the liver and kidneys. Therefore, we examined the effect on Organs At Risk (OARs) in cases of whole stomach and partial stomach irradiations. In the upper and lower parts of the partial stomach, the positional relationship between the liver and the kidneys becomes problematic. Thus, a simulation study was performed using the whole stomach as a control, divided into the upper stomach and lower stomach.
Materials and Methods
Patients
Twenty patients with advanced gastric cancer with inoperable bleeding who had undergone RT to the whole stomach. Patients with the following conditions were excluded: high risk of febrile neutropenia (neutrophil level of ≤ 1000/μL, platelet count of <30000/mm3), distant metastases (the brain, lungs, and liver) with a life expectancy of <1 month, and past medical history of anticoagulant use or RT to the abdomen.
Radiotherapy planning
The study was conducted on 20 patients who had undergone hemostatic irradiation of the whole stomach. Moreover, we conducted retrospective partial stomach (upper and lower portion) irradiation and examined the effect for OAR. The whole stomach is set as the target control. Partial stomach cases were divided into 2 groups; upper stomach (cardiac side) and lower stomach (pylorus side). A simulation treatment plan was created targeting the upper and the lower stomach. The dose to each PTV was set to 20 Gy/5 fractions, the same as the whole stomach. We contoured the outer wall of the stomach using a butyl scopolamine injection on an empty stomach. RT was performed in the early morning, and the patients consumed a meal after RT. These methods provide good reproducibility [6-9]. The Planning Target Volume (PTV) was a 2 cm margin from the outer stomach wall in all directions. We contoured the intestine, liver, kidneys, and spinal cord as OARs. We planned treatment using the field-in-field technique. If the whole stomach is irradiated, it is unlikely that the tumour will fall out of the field. However, when irradiating the partial stomach, we did not recommend the use of Intensity-Modulated Radiotherapy (IMRT) because the movement of the stomach could cause the tumour to fall out of the field.
All treatment plans were approved if conventional dosimetric constraints were respected for the liver and kidneys: liver, mean radiation dose (Dmean)<10 Gy and maximum radiation dose (Dmax)<22 Gy and kidneys, Dmean<0 Gy and Dmax <22 Gy. Although the dose was 20 Gy/5 fractions, which is equal to 28 Gy of the Biological Effective Dose (BED 10), other strict dose regulations were not set, but the dose to the liver and kidneys was reduced as much as possible. The definition of Dx was as follows: dose (Gy) irradiated to x% of the volume of the liver and bilateral kidneys.
Evaluation
The independent samples (whole, upper and lower) were used to compare the remaining parameters. The mean liver volume (cc) and irradiated the stomach volume of three methods. The mean dose in Gray [Dmean (Gy)], maximum dose in Gray [Dmax (Gy)] and the liver D5 (relative volume receiving ≥ 5 Gy), D10, D15, D20, D30 and the kidneys D10, D15, D20, D25.
Statistics
We used two-way analysis of variance (two-way ANOVA) and Tukey’s multiple comparison procedure. All statistical analyses of the recorded data were performed using the Excel statistical software package (Excel-statistics 2015; Social Survey Research Information Co., Ltd., Tokyo, Japan). A p-value of <0.05 is regarded as statistically significant difference.
Results
In all cases, the both partial stomach dose were lower in the whole stomach than in the liver and kidney as DVH (Figure 1). All treatment plans were approved if conventional dosimetric liver and kidneys constraints were respected: Liver; Dmean is <10 Gy, Dmax is <22 Gy. Kidneys: Dmean is <10 Gy. Dmax is <22 Gy. Dx; Dose (Gy) irradiated to x% of liver and bilateral kidney’s volume (Table 1). All treatment plans were approved if conventional dosimetric liver and kidneys constraints were respected: Liver; Dmean is <10 Gy, Dmax is <22 Gy. Kidneys: Dmean is <10 Gy. Dmax is <22 Gy. Dx; Dose (Gy) irradiated to x % of liver and bilateral kidney’s volume.
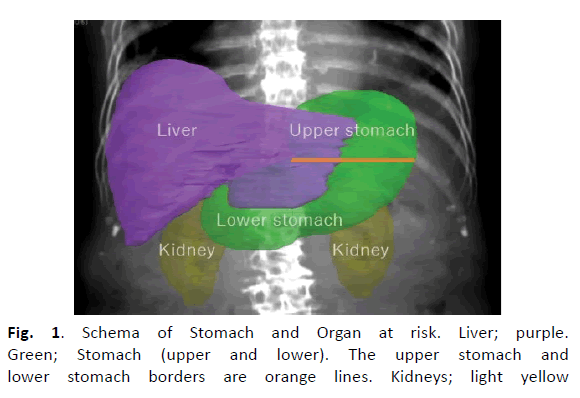
Figure 1: Schema of Stomach and Organ at risk. Liver; purple. Green; Stomach (upper and lower). The upper stomach and lower stomach borders are orange lines. Kidneys; light yellow
| Irradiated field | Whole | Upper partial | Lower partial |
| n=20 | n=10 | n=10 | |
| Mean Whole Liver Volume (cc) | 1102 ± 324 (n=20) | ||
| Irradiated Stomach Volume (cc) | 335 ± 48 | 207 ± 48 | 286 ± 32 |
| Liver | Gy | Gy | Gy |
| Dmean (<15 Gy) | 7.9 ± 3.4 | 4.5 ± 1.3 | 3.6 ± 1.0 |
| Dmax(<22 Gy) | 20.7 ± 0.4 | 20.7 ± 0.5 | 20.7 ± 0.4 |
| D30 (%) | 11.7 ± 4.9 | 6.4 ± 3.9 | 2.9 ± 2.6 |
| D25 (%) | 13.1 ± 4.8 | 6.6 ± 4.1 | 4.5 ± 3.2 |
| D20 (%) | 14.9 ± 4.1 | 8.3 ± 4.1 | 6.7 ± 3.4 |
| D15 (%) | 16.7 ± 4.3 | 9.9 ± 4.6 | 9.7 ± 2.8 |
| D10 (%) | 17.5 ± 4.0 | 13.4 ± 4.4 | 13.3 ± 3.3 |
| D5 (%) | 18.8 ± 3.2 | 17.4 ± 3.8 | 15.8 ± 5.7 |
| Kidneys | Gy | Gy | Gy |
| D25 (%) | 13.7 ± 5.8 | 3.5 ± 6.2 | 10.1 ± 3.8 |
| D20 (%) | 15.4 ± 4.5 | 5.3 ± 6.4 | 11.5 ± 2.7 |
| D15 (%) | 16.5 ± 3.6 | 8.0 ± 7.3 | 12.9 ± 1.8 |
| D10 (%) | 18.8 ± 10.1 | 10.9 ± 7.2 | 14.7 ± 2.2 |
Tab.1. Parameter of dose among three irradiated field (whole, upper, and lower stomach)
Samples of comparison of dose distribution and volume histogram between three radiotherapy methods (Figures 2 and 3). Partial radiotherapy has advantage of lowing field irradiated area. However, in lower stomach dose irradiation to the kidneys is high in some cases.
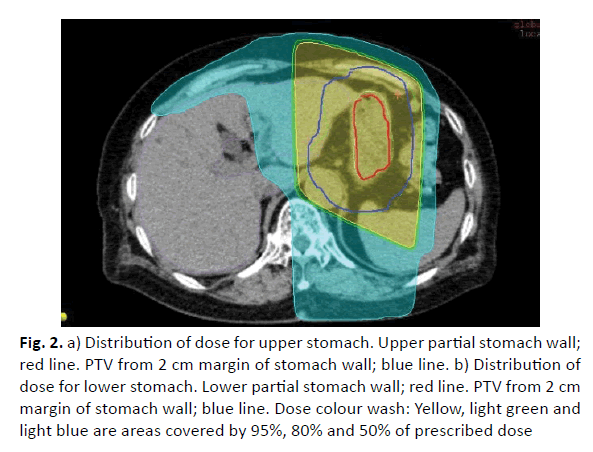
Figure 2: a) Distribution of dose for upper stomach. Upper partial stomach wall; red line. PTV from 2 cm margin of stomach wall; blue line. b) Distribution of dose for lower stomach. Lower partial stomach wall; red line. PTV from 2 cm margin of stomach wall; blue line. Dose colour wash: Yellow, light green and light blue are areas covered by 95%, 80% and 50% of prescribed dose
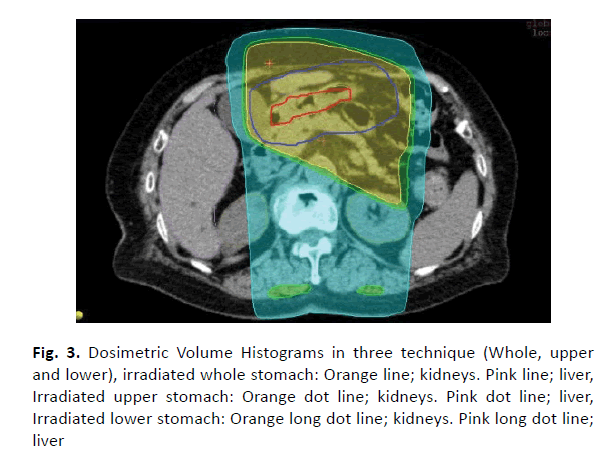
Figure 3: Dosimetric Volume Histograms in three technique (Whole, upper and lower), irradiated whole stomach: Orange line; kidneys. Pink line; liver, Irradiated upper stomach: Orange dot line; kidneys. Pink dot line; liver, Irradiated lower stomach: Orange long dot line; kidneys. Pink long dot line; liver
As the liver, irradiation to upper stomach is widely distributed. And irradiation to lower stomach is distributed bilateral kidney.
Dosimetric analysis of the Liver
The upper stomach was higher than the lower stomach in the high dose range of livers D20, D25, and D30. On the other hand, there was no difference between the upper and lower stomachs of livers D5, D10, and D15 in the low dose range.
Dosimetric analysis of the Kidneys
Regarding DVH, renal dose was significantly lower in all DVHs in the upper stomach than in the lower stomach. However, in kidney D25, the lower stomach was not significantly different from the whole stomach.
Discussion
We investigated how irradiation of the partial stomach could reduce the dose to the OAR compared with the whole stomach. The dose to the OAR was clearly lower in the partial stomach than in the whole stomach.
Chemotherapy and radiation therapy are used in addition to gastric cancer surgery and endoscopic treatment. We apply the initial dose of 20 Gy to the whole stomach for gastric cancer with bleeding. A clinical trial is conducted to re-irradiate the partial stomach with 15 Gy when re-bleeding occurs [8, 9]. Since advanced gastric cancer may have impaired liver and kidney function, it is required that the radiation dose to the liver and kidney be as low as possible. It is expected that the dose to the liver and kidneys will vary depending on the location of the irradiated partial stomach. To date, there have been no reports of dosimetric comparisons of irradiation between the whole stomach and partial stomach. Therefore, we used whole stomach as a control and conducted a simulation study of two different virtual tumors (located in the upper and lower stomach) to examine the usefulness of partial gastric irradiation.
Irradiation to the stomach includes neoadjuvant and adjuvant for gastric cancer, consolidation therapy for lymphoma, and hemostasis. The irradiation range is usually set to the whole stomach. Because it is assumed that re-irradiation is not performed. However, in the case of irradiation for hemostatic purposes, there is a possibility that it can be re-irradiated like bone metastasis. Therefore, the setting of the irradiation range of the stomach is important. When there is liver metastasis, it is necessary to set the irradiation range in consideration of liver function. Therefore, a simulation test was conducted to determine the extent to which the liver and kidneys were irradiated depending on the location of gastric cancer when irradiating the entire stomach or only the partial stomach.
The extent of tumour growth is identified from the inner surface with an endoscope, and the extent of growth outside the stomach wall is determined using contrast-enhanced CT. However, if the irradiation range is unclear on contrast-enhanced CT, diffusion weighted magnetic resonance imaging may be useful [9]. Especially useful when re-irradiation becomes necessary.
In order to maintain reproducibility, radiotherapy is given before breakfast, and butyl scopolamine is injected intramuscularly before the start of irradiation. During treatment, cone-beam CT is used for alignment every time. In our clinical trials, the initial irradiation is prescribed 20 Gy for the whole stomach, but the target for re-irradiation is the partial stomach. Then, a clip is placed in the vicinity of the tumour with an endoscope, and a GTV of the partial stomach is created using it as a landmark. This time, the clip is not detained for simulation.
Radiation-induced liver disease
RILD or radiation hepatitis is a sub-acute form of liver injury due to radiation [10-15]. It is one of the most dreaded complications of radiation which prevents radiation dose escalation and re-irradiation for hepatobiliary or upper gastrointestinal malignancies. Incidence of RILD is decreasing due to better knowledge of liver tolerance, improved investigation modalities and modern radiation delivery techniques, treatment options are still limited. Therapeutic partial liver RT (standard fractionation) Mean normal liver dose (liver minus gross tumor volume): <28 Gy in 2-Gy fractions for primary liver cancer, and <32 Gy in 2-Gy fractions for liver metastases [11].
Radiation-induced renal disease
Renal toxicity is an important complication of radiotherapy for the treatment of abdominal malignancy [16-18]. Radiotherapy with AP/ PA parallel-opposed fields raises the risk of radiation nephropathy and hypertension. Nevinny et al. reported that partial kidney irradiation; Bilateral kidneys Mean kidney dose <18 Gy. Bilateral kidneys V28 Gy < 20%. Bilateral kidneys V23 Gy < 30% [18].
Intensity modulated radiotherapy
Biancia et al. reported that the mean liver dose for 4-field 3D-CRT was 17.6 Gy compared to 6.8 Gy in AP/ PA parallelopposed fields [5]. IMRT, currently the most advanced radiotherapy technique, is considered an attractive option for the treatment of HCC, and is more widely applied because it can deliver a higher dose to the tumor than 3D-CRT while sparing surrounding normal organs [19, 20]. It is natural that the dose to the OAR is lower when the partial stomach is irradiated than the whole stomach. However, it should be noted that: In the case of irradiation of only the upper stomach, the high dose range (D15, D20, D25, D30) to the liver was low, but the low dose range (D5, D10) was not different from the whole stomach.
In the case of irradiation only in the lower stomach, the dose to the kidneys was generally not different from that in whole stomach. Therefore, it is necessary to pay attention to the dose to the kidney when irradiating the lower stomach. It is currently unknown whether the full or partial stomach is useful. However, it is highly likely that the use of re-irradiation will spread in the future. If re-irradiation becomes necessary, reducing the range of the initial irradiation can preserve liver and kidney function. Re-irradiation becomes easier as the preserved amount of OAR increases. However, in the future, it is necessary to conduct a comparative study of the local control rate and adverse events between the first irradiation in the whole stomach group and the partial stomach group.
Conclusion
High dose difference between the upper and lower stomachs was found in liver DVH. In the kidney, D25 in the high dose range was not significantly different between the whole stomach and the partial stomach. As for the lower stomach, it is necessary to pay attention to the kidneys’ DVH. Partial stomach irradiation is recommended if the tumour location can be identified by CT.
References
- Klevebro F, Nilsson K, Lindblad M, Ekman S, Johansson J, et al. Association between time interval from neoadjuvant chemoradiotherapy to surgery and complete histological tumor response in esophageal and gastroesophageal junction cancer: a national cohort study. Dis Esophagus. 2020;33:1-8.
- Hizal M, Sendur MA, Bilgin B. Expanding treatment options for resectable gastric cancer: is it a countdown for radiotherapy? J BUON. 2019;24:1367-1370.
- In-Hwan Kim. Current status of adjuvant chemotherapy for gastric cancer. World J Gastrointest Oncol. 2019;11:679-685.
- O'Connell L, Coleman M, Kharyntiuk N. Quality of life in patients with upper GI malignancies managed by a strategy of chemo radiotherapy alone versus surgery. Surg Oncol. 2019;30:33-39.
- Della Biancia C, Hunt M, Furhang E. Radiation treatment planning techniques for lymphoma of the stomach. Int J Radiat Oncol Biol Phys. 2005;62:745-751.
- Tanaka O, Yokoi R, Mukai T. Radiotherapy for gastric bleeding from tumor invasion of recurrent colon cancer with liver metastasis after resection. J Gastrointest Cancer. 2019;50:349-352.
- Tanaka O, Matsuura K, Sugiyama A. Hemostatic radiotherapy used twice for inoperable progressive gastric cancer with bleeding. J Gastrointest Cancer. 2019;50:151-155.
- Tanaka O, Yamada M, Kato T. Two sessions of radiotherapy were successful in treating gastric cancer with bleeding. J Gastrointest Cancer, 2018;50:955-959.
- Tanaka O, Omatsu T, Kariya S. Usefulness of diffusion-weighted magnetic resonance imaging for evaluating the effect of hemostatic radiotherapy for unrespectable gastric cancer. Clin J Gastroenterol. 2019;12:269-273.
- Bentzen SM, Constine LS, Deasy JO. Quantitative analyses of normal tissue effects in the clinic (QUANTEC): an introduction to the scientific issues. Int J Radiat Oncol Biol Phys. 2010;76:S3-S9.
- Pan CC, Kavanagh BD, Dawson LA. Radiation-associated liver injury. Int J Radiat Oncol Biol Phys. 2010;76:S94-S100.
- Lawrence TS, Ten Haken RK, Kessler ML. The use of 3-D dose volume analysis to predict radiation hepatitis. Int J Radiat Oncol Biol Phys. 1992;23:781-788.
- Liang SX, Huang XB, Zhu XD. Dosimetric predictor identification for radiation-induced liver disease after hypofractionated conformal radiotherapy for primary liver carcinoma patients with Child-Pugh Grade A cirrhosis. Radiother Oncol. 2011;98:265-269.
- Liang SX, Zhu XD, Xu ZY. Radiation-induced liver disease in three-dimensional conformal radiation therapy for primary liver carcinoma: the risk factors and hepatic radiation tolerance. Int J Radiat Oncol Biol Phys. 2006;65:426-434.
- Lawrence TS, Robertson JM, Anscher MS. Hepatic toxicity resulting from cancer treatment. Int J Radiat Oncol Biol Phys. 1995;31:1237-1248.
- Dawson LA, Kavanagh BD, Paulino AC. Radiation-associated kidney injury. Int J Radiat Oncol Biol Phys. 2010;76:S108-S115.
- Inaba K, Okamoto H, Wakita A. Long-term observations of radiation-induced creatinine clearance reduction and renal parenchymal volume atrophy. Radiother Oncol. 2016;120:145-149.
- Nevinny-Stickel M, Poljanc K, Forthuber BC. Optimized conformal paraaortic lymph node irradiation is not associated with enhanced renal toxicity. Strahl Onkol. 2007;183:385-391.
- Cheng JC, Wu JK, Huang CM. Radiation-induced liver disease after three-dimensional conformal radiotherapy for patients with hepatocellular carcinoma: dosimetric analysis and implication. Int J Radiat Oncol Biol Phys. 2002;54:156-162.
- Bae SH, Won Jang WI, Park HC. Intensity-modulated radiotherapy for hepatocellular carcinoma: dosimetric and clinical results. Oncotarget. 2017;8:59965-59976.



