Research Article - Onkologia i Radioterapia ( 2023) Volume 17, Issue 9
Evaluation of proteolytic enzymes in Iraqi patients with colorectal cancer
Noor Emad* and Firas A. HassanNoor Emad, Dept. of Chemistry, College of Science, Al Nahrain University, Baghdad, Iraq, Email: nooremad1441@gmail.com
Received: 14-Jul-2023, Manuscript No. OAR-23-110295; Accepted: 01-Sep-2023, Pre QC No. OAR-23-110295 (PQ); Editor assigned: 17-Jul-2023, Pre QC No. OAR-23-110295 (PQ); Reviewed: 02-Aug-2023, QC No. OAR-23-110295 (Q); Revised: 31-Aug-2023, Manuscript No. OAR-23-110295 (R); Published: 02-Sep-2023
Abstract
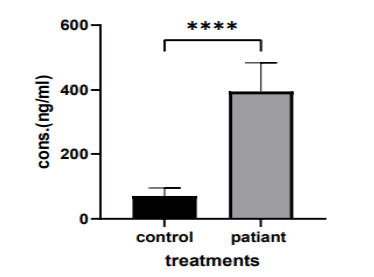
Background: Serine, aspartic and cysteine protease enzymes play a major role in Colorectal Cancer (CRC) progression. The objective of this article was to measure serine, aspartic, and cysteine protease enzymes in colorectal cancer patients and compare them with healthy subjects. Methods: Seventy newly diagnosed colorectal cancer patients were enrolled in the present study and divided as follows; Group A : (n=32) with an age range (of 20 years - 40 years); Group B : (n=38) with age range (41 years - 60 years). For comparison, a total of (30) healthy subjects were also inserted in the current study divided into patients in the same manner. Serum levels of serine, aspartic, and cysteine protease enzymes were measured for patients and controlled by the ELISA technique. Results: The current study shows that serum serine protease levels varied significantly in (the 20 years - 40 years) group when comparing the patient and control group (395.5 ng/ml vs. 71.61 ng/ml, p>0.0001) and also in (41 years - 60 years) group with mean 469.4 ng/ml vs. 106.3 ng/ml, p>0.0001, for patients and control groups respectively. Conclusion: Proteolytic enzymes including serum serine, aspartic, and cysteine protease were significantly increased in colorectal cancer patients when compared to healthy subjects.
Keywords
prostate cancer, social support, mental health, depression, life satisfaction
Introduction
Colorectal Cancer (CRC) is divided into two stages colon & rectal cancer. It is the 3rd most common kind of cancer and the fourth leading cause of cancer death. CRC is caused by the unusual development of epithelial cells that form the lining of the colon or rectum; certain tiny developments (known as polyps) have the potential to evolve into malignant tumors, while the majority of them are benign. It is believed that two-thirds of pre-malignant polyps in the colon and rectum increase the risk of CRC [1].
Protolysis is among the most crucial biological functions. Proteases [2]. A class of enzymes, have been connected to proteolytic activity. These enzymes are present almost everywhere and are crucial to many biological processes. Proteases, however, are also developed in cancer progression, according to a recent study [3]. The release of several proteases has been demonstrated to be positively correlated with tumor aggression [4]. Normal cells' proteases play a crucial role in carrying out crucial biological activities, however, altered tumor cells are the ones that do the most damage. Prognosis is also quite challenging since tumor cells secrete some particular proteases. Proteases aren't just expressed by cancerous cells, though [5]. Several times, tumor cells cause the neighboring, non-neoplastic cells to produce proteolytic enzymes, repurposing their function to support tumor growth [6].
Cysteine proteases are a subclass of proteolytic enzymes distinguished by an active site cysteine residue. Cathepsins B, L, H, and S can be found in the lysosome or the cytoplasm, and in pathological circumstances, some cell types produce them [7]. Cysteine proteases have a role in both broad processes like the breakdown of intracellular proteins and more specific ones like the selective activation of signaling molecules like interleukin, enkephalin, and protein kinase C or the destruction of extracellular proteins like bone resorption and macrophage activity. Numerous studies have linked the production of Intracellular cysteine proteases to the development of tumors [8].
Serine proteases are a particular class of proteases that are tightly connected to cell division and proliferation. They are typically located in the form of zymogens, and their activation by selective and limited proteolysis modifies the activity of the enzyme [9]. Additionally, physiological inhibitors that control cellular behavior are present. The physiological functions of the cell depend on the normal management of serine protease activities, and improper control of these activities can cause pathological illnesses. Serine proteases of the urokinase-type have been thoroughly scrutinized for their role in tumor invasion and metastasis [10]. Aspartic proteases are enzymes having two sections divided via a cleft that contains the catalytic site (two aspartate residues). Cathepsin-D (Cath-D) is a widely distributed aspartic endo-protease. It is present in lysosomes. Cath-D has been demonstrated to stimulate the pre-lysosomal recognition of specialized cells, as well as Its act as a main protein-degrading enzyme. Cathepsin D, on the other hand, has been widely researched over the last three decades, particularly in terms of its function in tumor formation and potential as a standalone tumor parameter. This research also changed the concept of Cath-D's function and supported the identification of novel Cath-D functionalities [11].
The present study aims to evaluate the photolytic enzymes in colorectal cancer.
Materials and Methods
Patients
About (70) newly diagnosed patients with colorectal cancer were enrolled in the current study. All patients were diagnosed by a physician in Al-Yarmook Teaching Hospital in a period from September 2022-January 2023. The age range of patients was (20 years - 60 years).
Patients were divided into two groups according to their ages: Group A : (n=32) with age range (20 years - 40 years); Group B: (n=38) with age range (41 years - 60 years).
For comparison, seventy healthy subjects were inserted in the present study as the control group, also divided according to their ages into two groups (20-40) (n=20) and (41-60years) (n=10).
Assessment of serum proteolytic enzymes
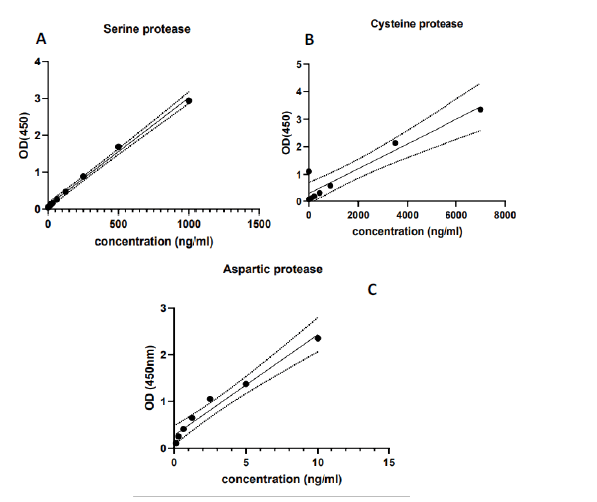
Five milliliters of blood were withdrawn from each control and patient transferred into a gel tube, allows 15 minutes to clot, and centrifuged after that at 3000 rpm, the serum was then isolated for 10 minutes' centrifugation used for measurements of serum serine, aspartic, and cysteine protease enzymes levels which were assessed in blood serum by using ELISA technique and the supplier was Abcam Co., UK (Figure 1).
Figure 1: A. Standard curve for serine protease, B. Standard curve for cysteine protease, C. Standard curve for aspart c protease
Results
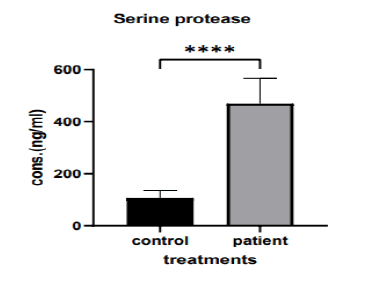
The current study shows that there was a significant vari ation in serum serine protease levels in (the 20-40 years) group when compares to the patient and control group (395.5 ng/ml vs. 71.61 ng/ml, p>0.0001), as shown in (Figure 2).
Figure 2: The mean of serine protease levels in (20 years-40 years) group
There was a significant variation in serum serine protease levels in (the 41-60 years) group when comparing the patient and control group (469.4 ng/ml vs. 106.3 ng/ml, p>0.0001), as shown in (Figure 3).
Figure 3: The mean of serine protease levels in (41 years-60 years) group
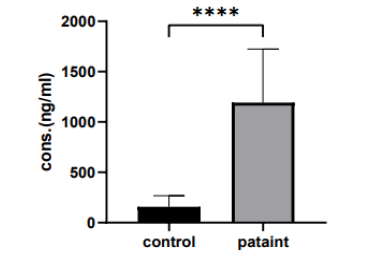
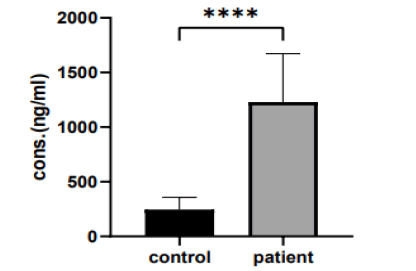
The current study shows that there was a significant va riation in serum cysteine protease levels in (20-40 years) group when comparing the patient and control group (1193 ng/ml vs. 158.3 ng/ml, p>0.0001), as shown in (Figure 4).
Figure 4: The mean of cysteine protease levels in (20 years-40 years) group
There was a significant variation in serum cysteine protease levels in (the 41-60 years) group when comparing the patient and control group (1228 ng/ml vs. 246.0 ng/ml, p>0.0003), as shown in (Figure 5).
Figure 5: The mean of cysteine protease levels in (41 years-60 years) group
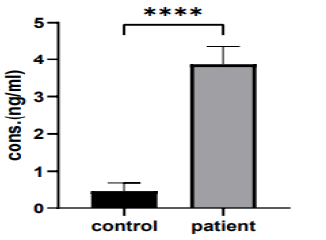
The current study shows that there was a significant variation in serum aspartic protease levels in (the 20-40 years) group when compares between the patient and control group (3.868 ng/ml vs. 0.4623 ng/ml, p>0.0199), as shown in (Figure 6).
Figure 6: The mean of aspart c protease levels in (20 years-40 years) group
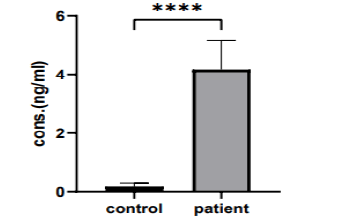
There was a significant variation in serum aspartic protease levels in (the 41-60 years ) group when compares the patient and control group (4.163 ng/ml vs. 0.1859 ng/ml, p>0.0001), as shown in (Figure 7).
Figure 7: The mean of aspart c protease levels in (41 years-60 years) group
Discussion
The current study was designed to evaluate the serum levels of proteolytic enzymes in newly diagnosed patients with colorectal cancer patients.
Serine protease levels were significantly increased in colorectal cancer patients when compared to a control group. Xie Y and his colleagues (2016) It has been discovered that serine protease has a role in colorectal carcinogenesis by promoting proliferation, invasion, and metastasis. Furthermore, colorectal tumors expressing serine protease have a worse outcome and survival rate [12]. The biological basis for serine protease drives cancer development is becoming clear. It appears to function in both directions via a 'protease-antiprotease-system' and by activating additional protease cascades [13]. Cancer cell instance for the cell membrane may be aided directly by serine protease breakdown of type I collagen [14].
The present study also showed there was a considerable rise in cysteine protease in patients when compared to healthy subjects. A recent study suggested there was a link between cysteine protease and cancer, demonstrating that the protease is secreted from human cancer tissue samples and is present in the serum of individuals with neoplasm lesions [15]. According to another study, cystine protease has been demonstrated to play a role in the remodeling and dissolution of fibrous tissue and basement wall during the growth, invasion, and metastasis of tumors, via the invasion of secreted lysosomes [16, 17]. Increased cysteine protease B and L levels have been linked to longer disease-free and Life expectancy rates and may thus be used as prognostic indicators for people who have cancer [18]. According to Patil et al. (2021), oral squamous cell carcinoma development and invasion were highly linked with cystine protease expression [19]. Chronic atrophic gastritis with dysplasia is more typically associated with cystine protease overexpression. Additionally, the role of cystine protease in the control of angiogenesis shows another unique function in the development of tumors [20].
In the current study, aspartic protease levels significantly increase in colorectal cancer when compared to control in all age groups. Human epithelial breast cancer cells have been shown to overexpress and produce large amounts of the aspartic protease cathepsin D (cath-D), a hallmark of bad fate in breast cancer, according to recent research [21, 22]. Cath-D promotes angiogenesis, metastasis, fibroblast outgrowth, and cancer cell proliferation [23]. The first evidence of cath-D's direct involvement in cancer metastasis came from rat tumor cells, whose in vivo metastatic capacity was boosted by transfectioninduced cath-D overexpression [24].
In conclusion, Protolytic enzymes significantly increase in newly diagnosed colorectal cancer so according to the present study they have a diagnostic importance.
Conclusion
Protolytic enzymes significantly increase in newly diagnosed colorectal cancer so according to the present study they have a diagnostic importance.
References
- Iversen LH. Aspects of survival from colorectal cancer in Denmark. Dan Med J. 2012;59:B4428. Google Scholar Crossref
- Slack MA, Gordon SM. Protease Activity in Vascular Disease. Arterioscler Thromb Vasc Biol. 2019;39:212-225. Google Scholar Crossref
- Martin CE, List K. Cell surface-anchored serine proteases in cancer progression and metastasis. Cancer Metastasis Rev. 2019;38(3):357-387. Google Scholar Crossref
- Vizovisek M, Ristanovic D, Menghini S, Christiansen MG, Schuerle S. The Tumor Proteolytic Landscape: A Challenging Frontier in Cancer Diagnosis and Therapy. Int J Mol Sci. 2021;22:2514. Google Scholar Crossref
- O’Connell C, VandenHeuvel S, Kamat A, Raghavan S, Godin B. The Proteolytic Landscape of Ovarian Cancer: Applications in Nanomedicine. Int J Mol Sci. 2022;23:9981. Google Scholar Crossref
- Itoh Y. Proteolytic modulation of tumor microenvironment signals during cancer progression. Front Oncol. 2022;12:744727. Google Scholar Crossref
- Verma S, Dixit R, Pandey KC. Cysteine Proteases: Modes of Activation and Future Prospects as Pharmacological Targets. Front Pharmacol. 2016;7:107. Google Scholar Crossref
- Karamanos NK, Theocharis AD, Piperigkou Z, Manou D, Passi A,et al. A guide to the composition and functions of the extracellular matrix. FEBS J. 2021;288:6850-6912. Google Scholar Crossref
- Martins A, Pfirrmann T, Heessen S, Sundqvist G, Bulone V,et al. Ssy5 is a signaling serine protease that exhibits atypical biogenesis and marked S1 specificity. J Biol Chem. 2018;293:8362-8378. Google Scholar Crossref
- Anand D, Hummler E, Rickman OJ. ENaC activation by proteases. Acta Physiol. 2022;235:13811. Google Scholar Crossref
- Song P, Cheng L, Tian K, Zhang M, Mchunu NP,et al.Biochemical characterization of two new Aspergillus niger aspartic proteases. 3 Biotech. 2020;10:303. Google Scholar Crossref
- Xie Y, Chen L, Lv X, Hou G, Wang Y, et al.The levels of serine proteases in colon tissue interstitial fluid and serum serve as an indicator of colorectal cancer progression. Oncotarget. 2016;7:32592-606. Google Scholar Crossref
- Qin S, Wang Z, Huang C, Huang P, Li D. Serine protease PRSS23 drives gastric cancer by enhancing tumor-associated macrophage infiltration via FGF2. Front Immunol. 2022;13:955841. Google Scholar Crossref
- Tagirasa R, Yoo E. Role of Serine Proteases at the Tumor-Stroma Interface. Front Immunol. 2022;11:144-152. Google Scholar Crossref
- Pogorzelska A, Å»oÅ?nowska B, Bartoszewski R. Cysteine cathepsins as a prospective target for anticancer therapies—current progress and prospects. Biochimie. 2018;151:85-106. Google Scholar Crossref
- Kciuk M, GieleciÅ?ska A, Budzinska A, Mojzych M, Kontek R. Metastasis and MAPK Pathways. Int J Mol Sci. 2022;23:3847. Google Scholar Crossref
- Iizuka S, Leon RP, Gribbin KP, Zhang Y, Navarro J, et al. Crosstalk between invadopodia and the extracellular matrix. bioRxiv. 2020.02.26.966762. Google Scholar Crossref
- Vasiljeva O, Hostetter DR, Moore SJ, Winter MB. The multifaceted roles of tumor-associated proteases and harnessing their activity for prodrug activation. Biol Chem. 2019;400:965-977. Google Scholar Crossref
- Patil R, Mahajan A, Pradeep GL, Prakash N, Patil S,et al.. Expression of matrix metalloproteinase-9 in histological grades of oral squamous cell carcinoma: An immunohistochemical study. J Oral Maxillofac Pathol. 2021;25:239-246. Google Scholar Crossref
- Park KC, Dharmasivam M, Richardson DR. The Role of Extracellular Proteases in Tumor Progression and the Development of Innovative Metal Ion Chelators that Inhibit their Activity. Int J Mol Sci. 2020;21:6805. Google Scholar Crossref
- Ashraf Y, Mansouri H, Laurent-Matha V. Immunotherapy of triple-negative breast cancer with cathepsin D-targeting antibodies. J Immunother Cancer. 2019;7:29. Google Scholar Crossref
- Basu S, Cheriyamundath S, Gavert N, Brabletz T, Haase G,et al. Increased expression of cathepsin D is required for L1-mediated colon cancer progression. Oncotarget. 2019;10:5217-5228. Google Scholar Crossref
- Alcaraz L, Mallavialle A, David T, Derocq D, Delolme F,et al.A 9-kDa matricellular SPARC fragment released by cathepsin D exhibits pro-tumor activity in the triple-negative breast cancer microenvironment. Theranostics. 2021;11:6173-6192. Google Scholar Crossref
- Pranjol MZ, Gutowski N, Hannemann M, Whatmore J. The Potential Role of the Proteases Cathepsin D and Cathepsin L in the Progression and Metastasis of Epithelial Ovarian Cancer. Biomolecules. 2015;5:3260-3279. Google Scholar Crossref