Research Article - Onkologia i Radioterapia ( 2022) Volume 16, Issue 3
Comparison study for IMRT, VMAT and 3D conformal in the treatment of gastric cancer patients
Islam Makhtar1*, Mahmoud EL Ghassaly2, Ehab Abdouh3 and Khaled El-Shahat32Department of Physics, Faculty of Science, Zigzag University, Egypt
3Department of Clinical Oncology, Faculty of Medicine, Al-Aznar University, Cairo, Egypt
Islam Makhtar, Medical Physics Unit, Radiotherapy military centre, Zigzag, Egypt, Email: eslamelaryshie1@yahoo.com
Received: 10-Dec-2021, Manuscript No. OAR-21-49433; , Pre QC No. 49433(PQ; Editor assigned: 11-Dec-2021, Pre QC No. 49433(PQ; Reviewed: 23-Dec-2021, QC No. 49433(Q); Revised: 12-Jan-2022, Manuscript No. 49433(R); Published: 24-Mar-2022
Abstract
The goal of the current study was to compare radiotherapy treatment plans for gastric cancer using double-arc volumetric (VMAT), Intensity-Modulated Radiotherapy (IMRT) versus 3DCRT delivery techniques. Materials and Methods: A total of 20 gastric cancer patients were involved in current study and each patient was scheduled IMRT VMAT and 3DCRT techniques. Dose-volume histogram statistics, Conformal Index (CI), hand Monitor Units (MUs) were analysed to compare treatment plans, treatment planning system eclipse Algorithm AAA with Energy true beam linear accelerator Varian model. Results and Discussion: The VMAT plans exceeded the other two techniques for coverage planning tumour volume dose and reduction dose for organs at risk in the kidneys, but not in the liver. VMAT exhibited a better mean CI (0.89±0.03), than the other techniques. In addition, for the kidneys the dose sparing (V13, V18 and mean kidney dose) was improved by VMAT plans. However, IMRT showed a marginal advantage in V30 and mean dose in normal liver when compared with VMAT. Conclusion: This study suggests that VMAT provides improved tumour coverage when compared with IMRT, and 3DCRT however, VMAT haven’t any advantage in liver protection when compared with IMRT. Furthermore, studies are required to establish differences in treatment outcomes among the four technologies
Keywords
gastric cancer, intensity-modulated radiotherapy, volumetric modulated arc therapy, dosimetric comparison
Introduction
Gastric disease is the fourth most regular kind of harmful tumour around the world [1] and the yearly number of novel cases is ~95 million. Every year ~70 million people surrender to gastric disease, which makes it the second most normal reason for malignant growth related mortality around the world [2]. Since the SWOG/INT-0116 preliminary [3] in 2001, adjuvant chemoradiotherapy has become a set up standard therapy for gastric disease. As opposed to the INT-0116 preliminary, which included D0-or D1-resected gastric malignancy patients, Kim et al [4] contemplated D2-resected members utilizing the equivalent chemoradiotherapy regimens, and furthermore exhibited that simultaneous chemotherapy expanded endurance and diminished repeat.
In any case, contrasted and medical procedure alone, postoperative chemoradiotherapy essentially expanded poisonousness in patients. In the INT-0116 investigation, 57% of patients experienced evaluation 3 or 4 harmfulness [3]. Ringash et al [5] found that the use of Three-Dimensional Conformal Radiotherapy (3D-CRT) in patients with gastric malignancy, which is unique in relation to the 2D radiotherapy utilized in the INT-0116 preliminary, diminished the frequency of evaluation 2 or higher poisonousness to 25%. Comparable investigations have indicated that conformal and (IMRT) accomplishes Prevalent Arranging Tumour Volume (PTV) target inclusion and improved typical tissue saving [6-8]. Besides, in spite of the fact that the National Comprehensive Cancer Network Guidelines suggest either 3D-CRT or IMRT, it is presently generally acknowledged in the clinical calling that IMRT is better than 3D-CRT regarding tumour inclusion, expanded neighbourhood tumour control likelihood and portion decrease to specific organs in danger (OARs).
Volumetric adjusted bend treatment (VMAT), as an altered rendition of IMRT, utilizes the direct quickening agents Elekta Synergy VMAT and Elekta Precise (Elekta Oncology Systems, Crawley, UK) to lead dynamic regulation turn radiotherapy. The upsides of VMAT when contrasted and IMRT, remember a decrease for the quantity of screen units (MUs), more limited conveyance times and lower presentation of OARs. Practically speaking, the VMAT advancement relies upon the quantity of circular segments and the gantry point separating between ensuing control focuses. As of now, contention exists concerning whether a solitary curve VMAT can accomplish portion circulations tantamount to IMRT plans. Bertelsen et al [9] exhibited that solitary bend is adequate to accomplish an arrangement quality like IMRT, in any case, Guckenberger et al [10] have announced that it is subject to the multifaceted nature of the objective volume.
VMAT is viewed as same as or better than IMRT for specific malignancies, including head and neck, prostate, lung, cervical and pancreatic disease [11], nonetheless, an absence of thorough examination between IMRT to VMAT exists concerning gastric malignant growth treatment. Thusly, the current investigation meant to explain the dosimetric nature of two ARC VMAT for gastric malignancy, contrasted and 7-field IMRT (7F-IMRT) and 3DCRT.
Immobilization, simulation and target delineation
All patients were immobilized in a recumbent situation, with arms crossed over the head utilizing a thermoplastic shell. Intravenous differentiation upgraded figured tomography (CT)- reenactment was performed at 3 mm timespans utilizing Gemini GXL positron outflow tomography/CT (Philips Medical Systems). Respiratory control and stomach pressure were not utilized. Following reenactment, the CT pictures were moved to the Pinnacle3 form 9.2 radiation therapy arranging framework (Philips Medical Systems). The Clinical Objective Volume (CTV) included tumor bed and per gastric lymph hubs, following the proposals delineated in the INT-0116 preliminary [3]. The CTV to PTV expansion was typically 5-10 mm to account for daily setup error and organ motion. Normal structures, including the spinal cord, liver, colon, duodenum, small intestine and kidneys were also contoured. All the contours were drawn by the same physician. Each patient had one 7F-IMRT, Two ARC-VMAT and 3DCRT plan created by the same radiation therapist A similar portion limitations were utilized for formation of 7F-IMRT, VMAT and 3DCRT plans (Table 1).
| OARs | Prescribed dose limit |
|---|---|
| Spinal Cord | Dmax<40 Gy |
| Liver | V30<30% |
| Kidney | V13<50% |
| V18<33% | |
| Small intestine | Dmax<50 Gy |
| V50<10% | |
| V45<15% | |
| Duodenum | Dmax<50 Gy |
| V50<10% | |
| V45<15% | |
| Vn, percentage of volume receving at least x Gy; OARs, organs at risk | |
Tab. 1. OARs dose constraints
Treatment planning and optimization; VMAT, IMRT and 3DCRT
The IMRT optimization was performed using the direct machine parameter optimization algorithm in the treatment planning system (Eclipse treatment planning Systems version 15.6). IMRT uses seven coplanar beams; seven beam irradiation, angles of 0º, 51º, 102º, 153º, 204 º, 255º and 306º. In the plan generation, the maximum iterations in the plan optimization were 80. There were no limitations with regard to the MUs per segment. Plans were generated for the Varian true beam with 6-MV.
VMAT
The plans were optimized in the same planning system as mentioned previously. The double arc VMAT was planned with a beam delivery time of ≤120 sec ×2, and with a gantry rotation of 181-180-181° (a control points every 4°). Plans were generated with 6-MV and all the objective parameters and algorithm used were the same as that for the single arc VMAT. All the plans were repeatedly optimized until the objectives were met.
Evaluation of the dose-volume histogram (DVH)-based parameters
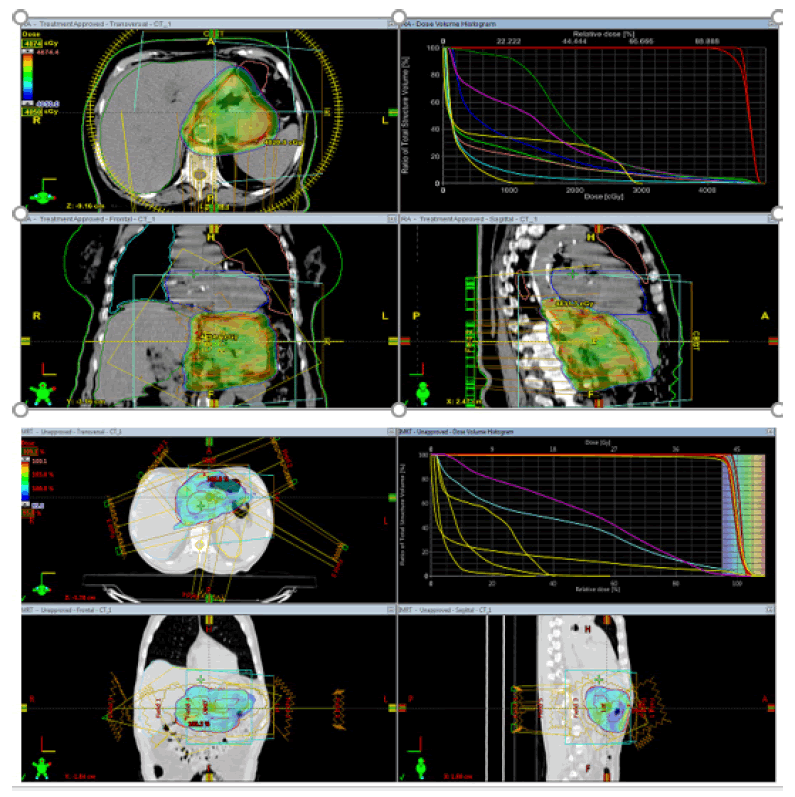
For the PTV, D98, D95, D50 and D2%, where D is the tolerant portion and n is the level of the PTV, were chosen to agree to the International Commission on Radiation Units and Measurements Report No. 83 [13]. The conformal record (CI) for PTV were determined. The CI was characterized as follows: CI= cover factor (the level of the PTV volume getting 50.4 Gy) × spill factor (the volume of the PTV accepting the 50.4 Gy comparative with the absolute remedy portion volume).The following dosimetric boundaries were reflectively d issected: V olumes o f k idney accepting a portion of ≥13 and 18 Gy (V13 and V18); volumes of liver getting a portion of ≥30 Gy; D2 of the spinal line; volumes of small digestive system and colon getting a portion of ≥50 Gy (V50); the mean portion to OARs and remaining volume in danger; the most extreme portion to 1, 5 and 10 cm3 of the pancreas and duodenum; and the volume of pancreas and duodenum accepting 5, 10, 15, 20, 25, 30, 35, 40, 45, and 50 Gy (Figure 1).
Figure 1: VMAT plan and IMRT Plan
PTV coverage
The assessment of the DVH-based boundaries of the PTV is appeared in Table 2. The D98 and D95 of the PTV were comparative among the 3DCRT, 7F-IMRT and VMAT plans, separately, and no critical contrasts were recognized between two methods (P>0.05). However, for 3DCRT arranging. For the PTV inclusion, the mean CI of the VMAT plans (0.96.5 ± 0.03) was essentially higher than that of 3DCRT (0.84 ± 0.02), IMRT (0.94 ± 0.02) individually (P<0.05). Moreover, VMAT designs additionally displayed a lower D2 (54.21 ± 49.92) when contrasted and the 3DCRT (54.52 ± 43.27) and IMRT (54.54 ± 57.63) (P<0.05). Ordinary portion dispersion in the cross over segment is appeared in Table 2.
| Radiotherapy | P-value | |||||
|---|---|---|---|---|---|---|
| Parameters | IMRT | VMAT | 3DCRT | IMRT | VMAT | 3DCRT |
| D98, Gy | 49.20±0.57 | 49.16±0.51 | 49.21±0.46 | 0.68 | 0.624 | 0.18 |
| D95, Gy | 50.45±0.44 | 50.51±0.38 | 54.31±0.50 | 0.104 | 0.446 | 0.07 |
| D50, Gy | 51.20±8.81 | 53.53±0.54 | 52.91±0.43 | <0.001 | 0.944 | <0.001 |
| D2, Gy | 54.62±0.43 | 55.43±1.10 | 54.31±0.50 | <0.001 | 0.001 | <0.001 |
| CI | 0.87±0.02 | 0.89±0.03 | 0.88±0.03 | <0.001 | 0.012 | <0.001 |
| HI | 0.14±0.17 | 0.13±0.02 | 0.11±0.01 | 0.003 | 0.013 | <0.001 |
Tab. 2. Comparisons of the dose-volume histogram-based parameters of the planning tumour volume.
OARs
VMAT essentially diminished the mean portion (14.54 ± 157.59 Gy), V13 (0.46 ± 0.04 Gy) and V18 (0.28±0.03 Gy) of the left kidney. Likewise, a lower mean portion (11.23 ± 188.43 Gy), V13 (0.26 ± 0.06 Gy) and V18 (0.18 ± 0.05 Gy) were seen in the contralateral kidney with VMAT. The mean portions to the typical liver for every strategy were 21.90 ± 138.97 Gy (DA-VMAT), 23.62±194.66 Gy (IMRT), and 19.92±196.08 Gy (3DCRT), with the mean portion to the ordinary liver with IMRT discovered to be the most reduced. Moreover, the V30 Gy (%) with VMAT (0.22 ± 0.05) was higher than that with 3DCRT (0.19 ± 0.03) (P<0.05) and IMRT (0.19 ± 0.03) (P<0.05) and DA-VMAT (0.19 ± 0.03)(P<0.05). The outcomes are appeared in Table 3.
| P-value | |||
| OARs | 3DCRT | IMRT | VMAT |
| Left kidney | |||
| V13 | 0.39±0.05 | 0.40±0.04 | 0.37±0.04 |
| V18 | 0.27±0.04 | 0.28±0.04 | 0.27±0.03 |
| Mean dose, Gy | 15.52±1.93 | 15.10±1.91 | 14.54±1.58 |
| Right kidney | |||
| V13 | 0.38±0.06 | 0.36±0.05 | 0.28±0.06 |
| V18 | 0.2±0.06 | 0.25±0.04 | 0.18±0.05 |
| Mean dose, Gy | 12.93±2.15 | 12.93±2.03 | 11.24±1.88 |
| Liver | |||
| V30 | 0.2±0.03 | 0.2±0.03 | 0.2±0.03 |
| Mean dose, Gy | 19.92±1.96 | 21.98±1.48 | 22.0±1.39 |
Tab. 3. Comparisons of the dose-volume histogram-based parameters of the kidneys and liver in present study.
Discussion
However, due to the combination of radiotherapy and chemotherapy, treatment-associated toxicities are enhanced, which often leads to relinquishment of treatment among patients. A number of studies on dosimetric comparison of 3D-CRT and IMRT have shown that IMRT exhibits improved OAR sparing. Few studies have investigated the application of VMAT in treating postoperative gastric cancer patients [14].
It is realized that the multifaceted nature of the objective volume and the quantity of VMAT circular segments are significant determinants of whether VMAT is favourable when contrasted and IMRT (11 moreover, the OARs in gastric malignant growth radiotherapy were discovered to be more radiosensitive than that in other site for some disease radiotherapy [8, 9]. True to form, the information in the current examination showed that the therapy anticipating gastric malignant growth VMAT plans accomplished unrivaled portion inclusion for PTV (CI was improved; P<0.05), a favourable position when contrasted and 3DCRT, however not IMRT.
What's more, the dosimetric boundaries of the duodenum and pancreas were analysed among the four innovations. Anatomically, the duodenum is the principal part of the small digestive tract. In any case, in the act of radiotherapy, they are distinctive with respect to portion and volume limit. As the duodenum borders the stomach, most of it is situated inside the objective volume. In this way, decreasing the portion got by the duodenum is a significant issue. Serious Gastrointestinal (GI) poisonousness has all the earmarks of being the fundamental portion restricting variable in stomach radiotherapy and it could be one reason why the IMRT is better than 3D-CRT as far as OARs saving. In any case, practically speaking, there is no distinction in intense GI harmfulness grade 2 among IMRT and VMAT. In a past report, the intense evaluation 2 or more noteworthy GI poisonousness was discovered to be 61.5 and 61.2% for 3D-CRT and IMRT, individually [7]. As indicated by Liu et al [14], the intense harmfulness was 56 and 54% for the IMRT and 3D-CRT gatherings, separately.
Taking everything into account, in the current examination, each of the three advances arrived at the norm for the marker of V25 Gy ≤45%, be that as it may, for V35 Gy≤0%, all advances fizzled. This examination is ju st astarter step and, hence, fu rther investigation is needed to improve the saving of the duodenum and distinguish dosimetric indicators for GI harmfulness. In the current examination, VMAT was discovered to be possibly more compelling than the other three advancements in D1, 5 and 10 cm3.
Conclusion
Although the fact that VMAT has been shown to display favourable circumstances in the treatment of different sorts of malignancies, the dosimetric bit of leeway of VMAT in this investigation was not generally clear when contrasted and IMRT. Moreover, it is muddled whether IMRT should be supplanted by VMAT. Thinking about the lower MUs, more limited conveyance times and decreased low-portion introduction of OARs, the utilization of VMAT in postoperative radiotherapy stays appropriate for gastric carcinoma; in any case, the clinical ramifications and result require further investigation.
References
- Kamangar F, Dores GM, Anderson WF. Patterns of, mortality, and prevalence across five continents: defining priorities to reduce cancer disparities in different geographic regions of the world. J Clin Oncol. 2006;24:2137-2150.
[Google Scholar] [CrosRef] - Parkin DM, Bray F, P. Ferlay J, Pisani Global cancer statistics, 2002. CA Cancer J Clin. 2005;55:74-108.
[Google Scholar] [CrosRef] - Macdonald JS, Smalley SR, Benedetti J, Estes CN, XHaller DG, et al. Chemoradiotherapy after surgery compared with surgery alone for adenocarcinoma of the stomach or gastroesophageal junction. N Engl J Med. 2001;345:725-730.
[Google Scholar] [CrosRef] - Kim S, Lim DH, Lee J, Kang WK, Park CH, et al. An observational study suggesting clinical benefit for adjuvant postoperative chemoradiation in a population of over 500 cases after gastric resection with D2 nodal dissection for adenocarcinoma of the stomach. Int J Radiat Oncol Biol Phys. 2005;63:1279-1285.
[Google Scholar] [CrosRef] - Murofushi K, Kitamura N, Machida H. The impact of vicryl mesh sheet placed on pelvic wall for reducing the irradiated bowel volume in VMAT of cervical cancer: Planning study. Int J Radiat Oncol Biol Phys. 2012;84:S430-S431.
[Google Scholar] [CrosRef] - Wang YB, Chen EQ, Cui YL, Zeng L, Wang YJ, et al. Seroprevalence of hepatitis B virus markers in individuals for physical examination in West China Hospital, China. Eur Rev Med Pharmacol Sci. 2011;15:592-596.
[Google Scholar] [CrosRef] - Minn AY, Hsu A, La T, Kunz P, Ford M, et al. Comparison of intensity-modulated radiotherapy and 3-dimensional conformal radiotherapy as adjuvant therapy for gastric cancer. Cancer. 2010;116:3943-3952.
[Google Scholar] [CrosRef] - Dahele M, Skinner M, Schultz B, Cardoso M, Bell C, et al. Adjuvant radiotherapy for gastric cancer: A dosimetric comparison of 3-dimensional conformal radiotherapy, tomotherapy and conventional intensity modulated radiotherapy treatment plans. Med Dosim. 2010;35:115-121.
[Google Scholar] [CrosRef] - Bertelsen A, Hansen CR, Johansen J, Brink C. Single arc volumetric modulated arc therapy of head and neck cancer. Radiother Oncol. 2010;95:142-148.
[Google Scholar] [CrosRef] - Guckenberger M, Richter A, Krieger T, Wilbert J, Baier K, et al. single arc sufficient in volumetric-modulated arc therapy (VMAT) for complex-shaped target volumes? Radiother Oncol. 2009;93:259-265.
[Google Scholar] [CrosRef] - Alvarez-Moret J, Pohl F, Koelbl O, Dobler B. Evaluation of volumetric modulated arc therapy (VMAT) with Oncentra MasterPlan® for the treatment of head and neck cancer. Radiat Oncol. 2010;5:110.
[Google Scholar] [CrosRef] - Edge SB, Compton CC. The American Joint Committee on Cancer: the 7th edition of the AJCC cancer staging manual and the future of TNM. Ann Surg Oncol. 2010;17:1471-1474.
[Google Scholar] [CrosRef] - International Commission of Radiation Units and Measurements. ICRU report 83: Prescribing, Recording, and Reporting Photon-Beam Intensity-Modulated Radiation Therapy (IMRT). J ICRU. 2010;10:1.
[Google Scholar] [CrosRef] - Stieler F, Wolff D, Schmid H, Welzel G, Wenz F, et al. A comparison of several modulated radiotherapy techniques for head and neck cancer and dosimetric validation of VMAT. Radiother Oncol. 2011;101:388-393.
[Google Scholar] [CrosRef]