Case Series - Onkologia i Radioterapia ( 2021) Volume 15, Issue 10
Cervical cancer brachytherapy amid the pandemic: A single institutional case series
Zahra Siavashpour1, Fatemeh Jafari2,4*, Mahdi Aghili2,4, Anya Jafari1 and Ramin Jaberi2,32Department of Radiation Oncology, Imam Khomeini Hospital Complex, Tehran University of Medical Sciences, Tehran, Iran
3Department of Physics, University of Surrey, Guildford, UK
4Radiation Oncology Research Center, Cancer Institute, Imam Khomeini Hospital Complex, Tehran University of Medical Sciences, Tehran, Iran
Fatemeh Jafari, Department of Radiation Oncology, Imam Khomeini Hospital Complex, Tehran University of Medical Sciences, Tehran, Iran, Email: jafari-f@razi.tums.ac.ir
Received: 20-Aug-2021 Accepted: 14-Oct-2021 Published: 20-Oct-2021
Abstract
Dose escalation to target volume during High Dose Rate brachytherapy was effectively employed to treat locally advanced cervical cancer patients who experienced prolongation of Overall Treatment Time (OTT) due to the COVID-19 pandemic. It was tried to achieve acceptable oncological outcomes while respecting the dose constraints of Organs at Risk (OARs).
Key Clinical Message: It was a severe challenge to obey the optimum treatment schedule of LACC cases during the coronavirus crisis. Increased risk of recurrence and mortality due to increased overall treatment time in cervical cancer patients proposes prioritizing.
http://www.surgeryjournals.com/
http://www.surgeryinsights.com/
http://www.medicineinsights.com/
http://www.medicinaljournals.com/
http://www.medicalsci.org/
http://www.eclinjournals.com/
http://www.eclinicalsci.org/
http://www.eclinicaljournals.org/
http://www.eclinicalinsight.com/
http://www.clinicalres.org/
http://www.clinicalinsight.org/
http://www.clinicalmedicaljournal.com/
http://www.clinicalmedicaljournal.org/
http://www.tradescience.org/
http://www.pharmares.org/
http://www.pharmainsights.org/
http://www.epharmajournal.org/
http://www.epharmajournal.com/
http://www.nursingres.org/
http://www.nursingres.com/
http://www.healthcareres.org/
http://www.healthcareinsights.org/
http://www.enursingcentral.com/
http://www.enursingcare.org/
http://www.ehealthjournals.org/
http://www.ehealthjournals.com/
http://www.psychiatryres.com/
http://www.neurologyres.com/
http://www.neurologyinsights.org/
http://www.neurologyinsight.com/
http://www.managjournal.com/
http://www.emedscience.org/
http://www.emedicinejournals.org/
http://www.molbioljournal.org/
http://www.molbioljournal.com/
http://www.engjournals.com/
http://www.enginsights.org/
http://www.edentalcentral.com/
http://www.dentistryjournals.org/
http://www.dentistryinsights.org/
http://www.scitechjournal.org/
http://www.jscitech.com/
http://www.pulsusjournal.com/
http://www.peerreviewjournal.org/
http://www.peerreviewjournal.com/
http://www.peerreviewedjournals.org/
http://www.journalinsight.org/
http://www.escientificjournals.com/
http://www.escienceopen.com/
http://www.esciencejournals.org/
http://www.esciencejournals.com/
http://www.emedicalscience.com/
http://www.emedicalsci.org/
http://www.emedicalsci.com/
http://www.microbiologyres.com/
http://www.microbialjournals.com/
http://www.immunologyres.com/
http://www.immunologyinsights.com/
http://www.molecularbiol.com/
http://www.esciencejournal.org/
Keywords
cervical cancer, brachytherapy, COVID-19, pandemic
Introduction
Based on the World Health Organization (WHO) report, cervical cancer is still one of the most frequent malignancies, which includes 6.6% of women cancers despite the screening campaign. This scenario is more severe in developing countries, such as Iran [1].
Chemoradiation and then Brachytherapy (BT) is the standard of care treatment for locally advanced cervical cancer patients [2, 3]. The BT boost serves to raise the gross tumor dose and enhances disease control and overall survival.
Overall Treatment Time (OTT) is a principal concern in radiation therapy, and any unnecessary radiotherapy delay should be avoided. For invasive carcinoma of the uterine cervix, especially for the SCC type, the OTT of external beam radiotherapy and BT play a significant role in pelvic tumour control and survival rates [3].
The Covid-19 pandemic is the current global threat to the healthcare system [4, 5]. The coronavirus outbreak in Iran has officially announced from the 3rd week of February 2020 [6]. Many public hospitals have been dedicated as referral centres for COVID-19 infected patients, so prevented the provision of services in another medical department. This situation has prompted the radiotherapy departments to create guidelines for categorizing some patients' treatment as a high priority and delaying other invasive procedures that need operation rooms. National Health Service (NHS) decision schemes are the worldliest used guidance in this era that classifies the patients for the anticancer therapies based on the type of tumour and radiotherapy anticipated treatment outcome. However, each radiotherapy department adapted the details of their protocols with their self-situations [5].
BT with Tandem-Cylinder-Needle (TCN) applicators for locally advanced cervical cancer patients, which needs operations rooms for applicator insertion, was one of our postponed cares in Yas hospital, Tehran, Iran, for about two weeks at the beginning of the pandemic. After two weeks, the department started to work but with a minimum possible workload and treatment delays based on their adapted guideline.
However, five years Overall Survival (OS) and five years Disease-Specific Survival (DSS) for stages IB2 to IIIB (based on International Federation of Gynecology and Obstetrics (FIGO) staging for gynecologic malignancies) of locally advanced cervical cancer are more than 50% [7]. This means that these patients belonged to the NHS scheme's priority level subset by anticipated outcome [5].
Several studies reported lower tumour control and survival rates in invasive uterine cervix carcinoma and upper risk of recurrence by increasing the treatment time procedure, especially for about seven weeks [3,8,9]. Several recurrence rate probabilities and percentage of tumour control loss per day of treatment prolongation were reported in the previous studies for different FIGO stages and OTTs, which were conveyed to be from about 0.59% to 1.1% [8].
This study aims to report an Iranian BT department's experience during the COVID-19 outbreak. It has been trying to survey and present the follow-up results for pelvic tumour control failure and investigate the risk of recurrence by prolonging the cervical cancer patient's BT during that time. Furthermore, this study tried to overview the obtained recurrence rate of locally advanced cervical cancer cases due to the treatment prolongation and compare it with the previously predicted recurrence rates by the previous reports [10].
Case Presentation
Patients' data selection
Medical files of cervical cancer patients referred to the Yas brachytherapy department from 1 February 2020 were extracted based on the following inclusion criteria:
• Patients with locally advanced cervical cancer who got EBRT of 44-50.4 Gy over five to six weeks with concomitant weekly chemotherapy (doses of 50 mg/m2 of Cisplatin) at least for five doses.
• Patients for whom the last EBRT fraction and their referral date to the Yas brachytherapy department were before February 2020.
• Patients that all their BT treatment fractions were finished before 20 April 2020.
Eventually, selected patients' age, FIGO stage, Overall Treatment Time (OTT), their EBRT EQD2, and brachytherapy details were pulled out. Overall treatment time has defined the interval between the beginning day of the EBRT and the last day of brachytherapy. The patient's treatments were finished before the study deigned. However, treatment results for each patient were gathered after a one-year follow-up period. Institutional Review Board and Ethics Committee approval was obtained for this study.
Brachytherapy procedure
The commercial treatment planning system was HDR plus 3 (Eckert & Ziegler BEBIG Gmbh, Berlin, Germany) with MultiSourceTM HDR-BT after loader treatment unit (Eckert & Ziegler BEBIG Gmbh, Berlin, Germany), which used a Cobalt-60 (Co-60) the radioactive source (model: Co 0.A86, EZAG BEBIG, Berlin, Germany). Utilizing Co-60 as a radionuclide in HDR brachytherapy increases between the brachytherapy centres, especially in Asian countries due to its relatively long half-life (i.e., t1/2=5.27 years) which, is beneficial in decreasing the source exchange and shipping expenses. There are no clinical disadvantages reported for HDR-BT with Co- 60 sources compared to 192Ir sources regarding previously published research [11].
BT applicator sets were also from the Eckert & Ziegler BEBIG Company. TCN applicator sets were used for the selected patients to cover the residual CTVs at the BT time. Three BT fractions were done for each patient.
Bladder, rectum, and sigmoid were delineated as Organs at Risk (OARs) for these patients. High-Risk Clinical Target Volume (CTVHR) and Low-Risk Clinical Target Volume (CTVIR) were also contoured. Target delineations were done using the CT scan tomography (The GE HI Speed Dual, General Electric, USA) with Intravenous (IV) contrast for all BT fractions and even Magnetic Resonance Imaging (MRI) data at the time of the first BT fraction, with and without IV gadolinium and ultrasonic gel injection to the vagina, based on the International Commission on Radiation Units and Measurements (ICRU) 89 recommendation [12].
For each patient, three fractions of insertion with a fractional prescription dose were calculated based on EQD2 (biologically equi-effective dose to dose delivered in 2 Gy fractions) formula considering α/β of 10 Gy and 3 Gy for CTVs and OARs, respectively [12]. Treatment planning aim was to deliver a minimum dose of 90 Gy (EQD2) to 90% of the CTVHR volume (D90); maximum dose to a volume of 2 cc (D2cc) and 0.1 cc (D0.1cc) of the rectum and sigmoid were below 70-75 Gy and 100 Gy, respectively. The maximum dose to a volume of 2 cc (D2cc) was less than 85-90 Gy for the bladder [12-13]. Dose- Volume Histogram (DVH) parameters of the mentioned OARs and CTVs in all three BT fractions were gathered retrospectively. 3D planning was done for each patient's BT fraction, and manual optimization was also performed.
Treatment results
All the patients were followed up every three months for one year after the end of the treatment. Treatment results, whether tumour response or complications, were conducted. Tumour response was evaluated by physical examination, imaging, and if needed, biopsy. Recurrence is considered as any local residue/recurrence or metastasis during the follow-up. Treatment complications of cystitis or proctitis were assessed using the RTOG scoring schema before, during, and after the brachytherapy for 12 months.
Outcome and follow-up
Twenty-three cervical cancer patients with different FIGO stages (Table 1) and the age range of 40-76 referred to our BT department met the current study's considered inclusion criteria. All of the pathological types of malignancy were Squamous Cell Carcinoma (SCC) of the uterine cervix. OTT of the patients ranged from 52-107 days. Demographic, therapeutic details and outcomes of the population are presented in Table 1 and Figure 1.
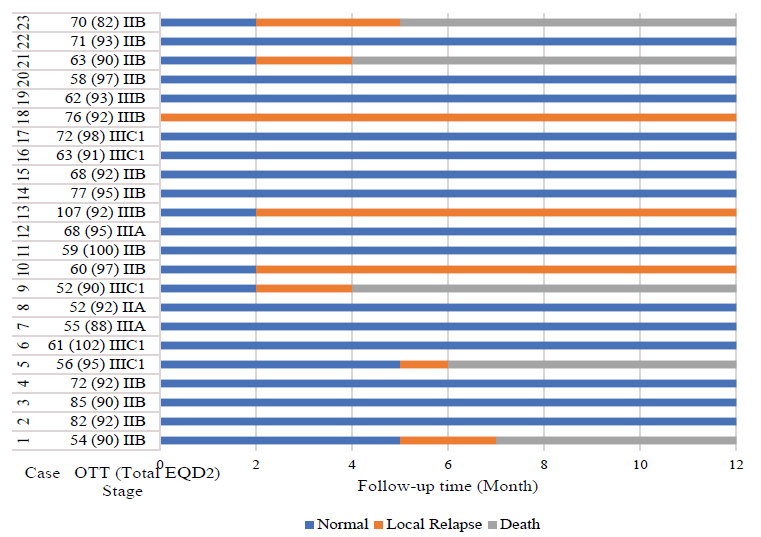
Figure 1: Patient's response to treatment during 12-months follow-up after brachytherapy
| Characteristic | Value |
|---|---|
| Total number of patients | 23 |
| Age (years) | 40-76 |
| Overall treatment time (days) | 52-107 |
| Stage | |
| IIA | 1 |
| IIB | 12 |
| IIIA | 2 |
| IIIB | 3 |
| IIIC1 | 5 |
| EQD2 of EBRT (Gy) | 44.3-54.0 |
| EQD2 EBRT+BT (Gy) | 82.0-97.0 |
| Outcome (12-month follow-up) | |
| Normal | 15 |
| recurrence | 8 |
| Months (after BT' last session) | 3-12 |
| Death | 5 |
| Months (after BT' last session) | 4-12 |
Tab. 1. Descriptive characteristic of included cases
The volume of CTVHR, CTVIR, and D2cc for rectum, bladder, and sigmoid of each patient was reported in Table 2.
| Patient ID | Target VOLUME (CC) | OAR D2CC (Gy) | Total EQD2 | ||
|---|---|---|---|---|---|
| CTVHR | CTVIR | Rectum | Sigmoid | Bladder | |
| 1 | 26.6 | 68.2 | 69.8 | 71.2 | 73.6 |
| 2 | 39.6 | 73 | 60.8 | 72.8 | 60 |
| 3 | 37 | 74.7 | 68.5 | 54.3 | 84.5 |
| 4 | 28.9 | 63.7 | 59 | 70.1 | 62.5 |
| 5 | 22.3 | 58.8 | 68.4 | 70.5 | 70.3 |
| 6 | 33 | 92.2 | 67.3 | 61.8 | 70.5 |
| 7 | 63 | 87 | 66.5 | 64.1 | 75 |
| 8 | 30.1 | 65.7 | 64.9 | 67.2 | 69.6 |
| 9 | 55.9 | 112.7 | 64.6 | 69.1 | 74.3 |
| 10 | 39.9 | 96.2 | 66.8 | 60.8 | 64.7 |
| 11 | 133.4 | - | 74.8 | 78.6 | 98.2 |
| 12 | 31.4 | - | 72.5 | 64.3 | 76.8 |
| 13 | 41.5 | 112.2 | 67.8 | 68.7 | 87.8 |
| 14 | 29.8 | 62.1 | 68.4 | 65.4 | 75.9 |
| 15 | 39.4 | 82.9 | 64.7 | 61.1 | 66.1 |
| 16 | 38.9 | 83.2 | 68.2 | 60.3 | 82.7 |
| 17 | 42.3 | 111 | 63.8 | 61.3 | 65.2 |
| 18 | 21.8 | 69.8 | 71.1 | 64.1 | 91.7 |
| 19 | 36.5 | 75.3 | 58 | 65 | 76.6 |
| 20 | 34.8 | 76.3 | 67.5 | 70.2 | 71.7 |
| 21 | 26.6 | 68.2 | 64.9 | 68 | 75.1 |
| 22 | 34.8 | 85.7 | 70.2 | 67.3 | 80.4 |
| 23 | 22.3 | 58.46 | 64.5 | 67.3 | 80.3 |
Tab. 2. Target and organs at risk brachytherapy' DVH parameters of the included cases
As illustrated in Figure 2, cystitis, eighter grade 1 or 2, was more frequent than proctitis, regarding the normal tissue tolerance dose during the planning. Grade 1 cystitis was more frequent at one month and grade 2 at one week after the final brachytherapy session, respectively. It was also the case for the proctitis, with more grade 1 proctitis at one month and more grade 2 at one week after finishing brachytherapy. Only one patient complained of grade 3 cystitis six months after treatment.
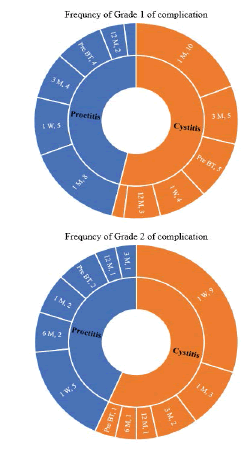
Figure 2: Frequency of proctitis and cystitis complication from pre-brachytherapy (Pre BT) checkup and during the follow-up durations (i.e., from 1 week (1 W) till 12 months (12 M) after last BT session)
Discussion and Conclusion
At the early of the Covid-19 pandemic, health resource constraints and the need to prioritize them led researchers and clinicians to provide rapid guidelines in oncology and radiotherapy to maximize the patients' care and safety while protecting radiotherapy department staff from being infected [5,6, 14-18].
To avoid increasing local recurrence risk and decreasing overall survival, BT of cervical cancer should not be delayed more than seven weeks. Nevertheless, it may prove challenging to deliver during the peak of coronavirus crisis when elective surgery and procedures would be suspended in COVID-19 referral hospitals, the same as Yas, Tehran, Iran. The hospital is a governmental university hospital dedicated to being a reference centre to treat Corona virus-affected patients. Therefore, for two weeks, all other treatments were delayed, as for the BT of our patients who needed applicator insertion under general anaesthesia or epidural sedation.
We faced 23 LACC patients whose EBRT was finished until 15 February 2020 and were introduced to the Yas hospital for BT. Their FIGO stages were from IIB to IVA (Table 1). Eight cases (34.7%) had local recurrence, and five died within the next year of treatment. Total EQD2 were in the range of 82-97 Gy, mostly more than 90 Gy. One case with local recurrence had a total EQD2 of less than 90 Gy (82 Gy). OTT of all the cases was more than seven weeks.
Based on Song, et al. and Viswanathan, et al., OTT of less than eight weeks does not influence the OS and DSS more when concomitant chemotherapy is prescribed for the patient [19,20].
Espenel, et al. in a recent retrospective study by reviewing the medical files of 94 locally advanced cervical cancer patients concluded that only 43.6% of these patients benefitted from the standard of care, which was following the American reported percentages (i.e., 44.3%). They reported that for only 25% of cases, OTT was less than 56 days, which would impact the pelvic control [7]. This means that even in non-critical situations, not all patients get the complete level of attention. This situation has gotten worse during the pandemic.
However, in a more recent study by Tanderup, et al. (the EMBRACE group), OTT was significantly associated with LC. Based on their conclusion, the 3-year local control rate is more than 93% for CTVHR with intermediate size (30 cc) and delivered dose of ≥ 85 Gy them for up to seven weeks [21].
Jhawar, et al. also reviewed the 3051 cervical cancer patients' treatment outcomes that got definitive chemoradiation therapy. Poor survival resulted when OTT of CRT duration was higher than seven weeks 8. Based on the Perez et al. study results, prolonging treatment duration for patients with stage III will increase the pelvic failure rate. Wang, et al. study results have also confirmed this statement. They followed up on 37 cervical cancer patients for whom HR-CTV was defined based on the GEC-ESTRO recommendations. They concluded that the complete treatment should be finished within seven weeks to improve the local control and clinical outcomes. 92% of their cases received concurrent chemotherapy, and 65% were SCC with FIGO stage III or higher [22]. A systematic review of 34 studies showed that treatment delays caused by pandemics increased cancer patients' mortality, including cervical cancer radiotherapy [23].
According to our study 34.7% of patients experienced local recurrence which was comparable with a study reported by Amuzegar Hashemi, et al. local recurrence of 31.8% in patients treated with two dimensional techniques [24].
Our study's high recurrence and mortality rate emphasized that the overall treatment time of chemoradiotherapy of advanced cervical cancer patients should ideally be less than seven weeks, and any treatment interruption should be avoided. Another option was obeying the ABS recommendation for single applicator insertion and twice-daily delivery during the pandemic 16. However, this was not possible due to the hospitalization of coronavirus-infected patients in our centre. The only way was to start the treatment with caution based on the patient's priority, such as their referral date, date of their last EBRT fraction, age, and health condition. Some of our patients were referred from another city to Tehran, and their treatment was postponed due to government quarantine rules.
It was a severe challenge to obey the optimum schedule of locally advanced cervical cancer patients during the peak of the coronavirus crisis. An increase of local pelvic failure would expect for these cases. Median and long-term follow-up has to be planned for all of these patients.
Conflict of Interest
The authors have no conflicts of interest to declare.
Author Contributions
Ramin Jaberi and Zahra Siavashpour were the physicians in charge of patient treatment, and contributed in data gathering, drafting and scientific revision of the manuscript. Fatemeh Jafari, Zahra Siavashpour, Anya Jafari and Mahdi Aghili contributed in writing, editing, and final revision of the manuscript.
References
- Cervical cancer. 2020.
- Banerjee R, Kamrava M. Brachytherapy in the treatment of cervical cancer: A review. Int J women's health. 2014; 6:555.
- Halperin EC, Brady LW, Wazer DE, Perez CA. Perez and Brady's principles and practice of radiation oncology. Lippincott Williams Wilkins; 2013.
- Darvishi, M., Shahali, H., & Farahani, A. A. (2021). Guillain-Barré Syndrome Associated with SARS-CoV-2 Infection: A Case Report. European Journal of Translational Myology, 31(3).
- Najafi A, Sadeghniiat K,Akbarpour S, Samadi S. The effect of apnea management on novel coronavirus infection: A study on patients with obstructive sleep apnea. Sleep Health. 2020; 7(1):14-18.
- Aghili M, Ghalehtaki R, Darzikolaee NM, Jafari F, Moshtaghian M. Radiotherapy and COVID-19: practical recommendations from Iran. Radiother Oncol. 2020; 149:70-71.
- Espenel S, Garcia MA, Trone JC, et al. From IB2 to IIIB locally advanced cervical cancers: report of a ten-year experience. Radiat Oncol. 2018;13:16.
- Jhawar S, Hathout L, Elshaikh MA, Beriwal S, Small Jr W,et al. Adjuvant chemoradiation therapy for cervical cancer and effect of timing and duration on treatment outcome. Int J Radiat Oncol Biol Phys. 2017;98:1132-1141.
- Wang C, Raince J, Park SJ, Mesko S, Demanes J,et al. Clinical Outcomes Using Image Guided Interstitial Brachytherapy for Definitive Cervical Cancer Patients with High Risk Clinical Target Volumes Greater Than 30 cc. Brachytherapy. 2017;16:S15.
- Grant MJ, Booth A. A typology of reviews: an analysis of 14 review types and associated methodologies. Health inf libr j. 2009;26:91-108.
- Strohmaier S, Zwierzchowski G. Comparison of 60Co and 192Ir sources in HDR brachytherapy. J. contemp. brachytherapy. 2011;3:199.
- Potter R, Kirisits C, Erickson B. Prescribing, Recording, and Reporting Brachytherapy for Cancer of the Cervix. Journal ICRU. 2016.
- Viswanathan AN, Kirisits C, Erickson B, Pötter R. Gynecologic radiation therapy. Viswanathan AN, editor. Springer-Verlag Berlin Heidelberg. 2011.
- COVID-19 rapid guideline: Delivery of systemic anticancer treatments. NICE guideline [NG161]; 2020.
- Achard V, Tsoutsou P, Zilli T. Radiotherapy in the time of the Coronavirus pandemic: when less is better. Int j radiat oncol Boil phys. 2020; 107:600-601.
- Mirzaee SMM.Gonçalves FG.Mohammadifard M, Tavakoli M, Vossough A. Focal cerebral arteriopathy in a pediatric patient with COVID-19. Radiology. 2020;297(2):E274-E275.
- Khan ZH, Samadi S, Makarem J, Mireskandari SM. Tests with proven value in diagnosis of COVID-19. Iranian Journal of Microbiology. 2020;12(3);261-262.
- Venkatraman P, Sahay JJ, Maidili T, Rajan R, Pooja S. Breakthrough of COVID-19 using radiotherapy treatment modalities. Radiother Oncol. 2020; 148:225-226.
- Song S, Rudra S, Hasselle MD, Dorn PL, Mell LK, et al. The effect of treatment time in locally advanced cervical cancer in the era of concurrent chemoradiotherapy. Cancer. 2013;119:325-331.
- Viswanathan AN, Thomadsen B, American Brachytherapy Society Cervical Cancer Recommendations Committee. American Brachytherapy Society consensus guidelines for locally advanced carcinoma of the cervix. Part I: general principles. Brachytherapy. 2012;11:33-46.
- Tanderup K, Fokdal LU, Sturdza A, Haie-Meder C, Mazeron R,et al. Effect of tumor dose, volume and overall treatment time on local control after radiochemotherapy including MRI guided brachytherapy of locally advanced cervical cancer. Radiother oncol. 2016; 120:441-446.
- Perez CA, Grigsby PW, Castro-Vita H, Lockett MA. Carcinoma of the uterine cervix. II. Lack of impact of prolongation of overall treatment time on morbidity of radiation therapy. Int. J. Radiat Oncol Biol Phys. 1996 ;34:3-11.
- Hanna TP, King WD, Thibodeau S, Jalink M, Paulin GA, et al. Mortality due to cancer treatment delay: systematic review and meta-analysis. bmj. 2020; 371.
- Amouzegar Hashemi F, Esmati E, Kalachi B. Pathologic Characteristics, Type of Treatment and Follow Up of Patients with Uterine Cervical Carcinoma Referred to the Radiation Oncology Department, Cancer Institute, Imam Khomeini Hospital, Tehran, Iran, 1995-2001. Asian Pac J Cancer Prev. 2007;8:86-88.



