Research - Onkologia i Radioterapia ( 2021) Volume 15, Issue 9
Asian Variant of Intra Vascular Large B cell Lymphoma of the Ad-Renal Glands with CNS Relapse-A Case Report
Sreelakshmy Baby Sarojam1, Indu R Nair2, Arun Philip1 and Keechilat Pavithran1*2Department of Pathology, Amrita Institute of Medical Science and research center, Amrita Vishwa Vidyapeetham, Kochi, Kerala, India
Keechilat Pavithran, Department of Medical Oncology, Amrita Institute of Medical Science and Research Center, Amrita Vishwa Vidyapeetham, Kochi, Kerala, India, Email: drkpavithran@hotmail.com
Received: 14-Jul-2021 Accepted: 26-Aug-2021 Published: 27-Sep-2021
Abstract
A 57-year-old gentleman presented with recurrent episodes of lowgrade fever, fatigue, and headache. Initial blood investigations showed pancytopenia with a serial decrease in counts. A laboratory workup ruled out infective pathology. Further, bone marrow aspiration was inconclusive. Provisionally, he was diagnosed with probable macrophage activation syndrome and was therefore pulsed with steroids. Subsequently, he showed improvement and was later discharged. However, after two weeks, he was readmitted with altered sensorium and lethargy. As his condition was found to be gradually deteriorating, a positron emission tomography-computed tomography was done; it showed significantly enlarged bilateral adrenal glands. Adrenal biopsy confirmed that it was non-Hodgkin’s lymphoma, specifically Intravascular Large B-Cell Lymphoma (IVLBCL). Afterwards, he was treated with R-CHOP, followed by intermediate doses of methotrexate for Central Nervous System (CNS) prophylaxis. His condition had improved on follow-up. However, after 18 months since diagnosis, he developed an isolated CNS relapse, confirmed by a brain biopsy. He was treated with modified DeAngelis protocol followed by whole-brain radiation therapy. To the best of our knowledge, this is the first reported case in India of adrenal gland IVLBCL with CNS relapse that responded to second-line chemotherapy with an overall survival of 60 months.
Keywords
lymphoma, adrenal, intravascular, large b-cell, Asian variant, and central nervous system
Introduction
Intravascular Large B-Cell Lymphoma (IVLBCL) is a rare and aggressive form of extranodal non-Hodgkin’s Diffuse Large B-Cell Lymphoma (DLBCL). It is characterized by the growth of large lymphoma cells without an extravascular tumour mass within the lumina of small to medium-sized blood vessels [1]. The disease has two variants:
(i) Western or classical variant, and
(ii) Asian variant-seen mainly in Japan
The Asian variant characteristically presents with multi-organ failure, fever, pancytopenia, bone marrow involvement, and hemophagocytic syndrome whereas the Western variant manifests itself more frequently with CNS and cutaneous symptoms and less commonly involves the bone marrow, liver, and spleen. Hemophagocytic Lymphohistiocytosis (HLH) is rare with the western variant [2].
IVLBCL shows predominance among the elderly population with a median age of 70 years across all sexes [3]. Despite its aggressive nature, early diagnosis and timely initiation of treatment usually result in a good prognosis. In our case, we report a 57-year-old man with IVLBCL of the adrenal gland and CNS relapse who responded to second-line chemotherapy with good overall survival.
Case Report
In June 2016, a 57-year-old gentleman presented to the internal medicine department with complaints of recurrent episodes of low-grade fever, headache, and lethargy for about three months. He had already undergone multiple hospital admissions for the same during this period and had been extensively tested. Other than undergoing atrial septal defect closure in December 2015, the patient (who has been on oral aspirin since then) has no other significant medical history or comorbidities. On general evaluation, he was found to have a well-built and nourished physique and was well-aware and oriented. He was febrile (temp: 100.10°F) and pallid. Furthermore, routine blood investigations showed evidence of anaemia and thrombocytopenia. Blood and urine cultures showed no significant growth. The workup for the infectious panel, including leptospirosis, dengue, malaria, and tuberculosis, was reported negative. The autoimmune workup Antinuclear Antibody (ANA) profile, antineutrophil cytoplasmic antibodies (c-ANCA, and p-ANCA) was also found to be negative. An Ultra Sonogram (USG) of the abdomen showed mild hepatosplenomegaly and enlarged bilateral adrenals. Further, a 2D echocardiography (2D ECHO) and transesophageal ECHO showed a normal study besides the previous ASD closure changes; hence, infective endocarditis was ruled out. A bone marrow aspiration and biopsy indicated hypercellular marrow with erythroid preponderance. Provisionally, it was diagnosed as probable macrophage activation syndrome and was therefore treated with steroids. The patient’s condition improved subsequently, and he was discharged afterwards; two weeks later, however, he was readmitted with altered sensorium and lethargy. Since his condition was found to be gradually deteriorating, a whole-body PET-CT was done, which showed significantly enlarged bilateral adrenal glands (Figure 1). He underwent an adrenal biopsy, which showed a neoplasm composed of cells predominantly seen in the intravascular compartment. The cells were large with moderate eosinophilia cytoplasm, round to ovoid nuclei with coarse chromatin and scattered mitosis (Figure 2a). An Immunohistochemistry (IHC) lymphoma panel was done, and the neoplastic were found to be positive for LCA, CD20 (Figure 2b), PAX-5, BCL2, and MUM-1 and negative for CK, CD3, BCL6, CD10, and TdT. Moreover, the Ki67 index was 60% focally. The neoplastic cells seen within vascular channels were highlighted by the endothelial marker, CD34 (Figure 2c), and based on these findings a diagnosis of intravascular large B cell lymphoma was made.
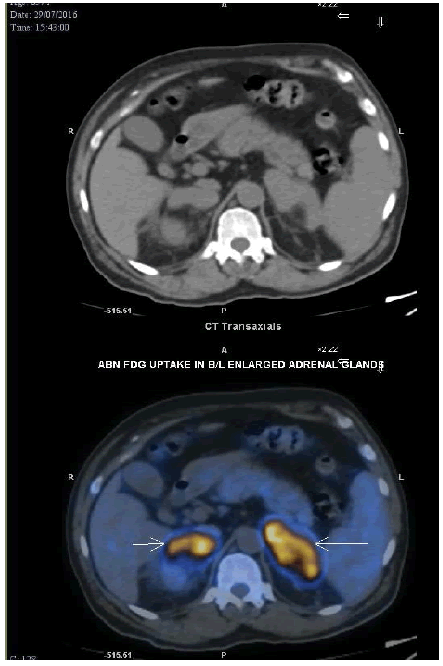
Figure 1: Enlarged adrenal glands in PET CT
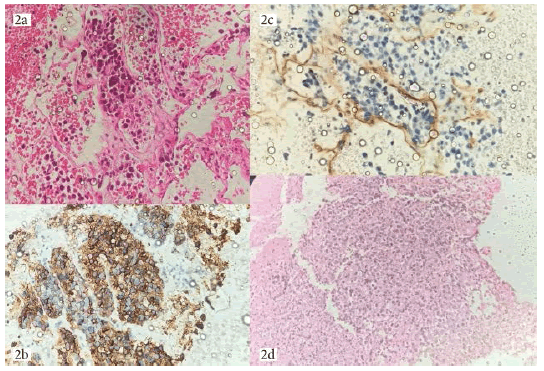
Figure 2: 2a): H&E 40x showing neoplastic lymphoid cells within vascular spaces; 2b): IHC 40x-Intravascular lymphoid cells showing CD20 positivity; 2c): IHC 40x showing vessel wall highlighted by CD34 staining; 2d): H&E 20x showing neoplastic cells infiltrating glial tissue
The patient was administered R-CHOP (rituximab 700 mg, cyclophosphamide 1500 mg, doxorubicin-100 mg, vincristine 2 mg, and prednisolone 100mg × 5 days, during the first cycle he received 75% dose as his performance status was 2) chemotherapy, followed by methotrexate 3 g/cycle once in 2 weeks for 3 courses for CNS prophylaxis.
After completing four cycles of chemotherapy (i.e. three months after initiation of chemotherapy, a repeat PET imaging was done, and it showed complete response. Treatment was completed by February 2017; and was on regular follow-up.
In July 2018, he presented with complaints of headache, swelling, and redness and pain over both lower limbs for two weeks (suggestive of cellulitis). Magnetic Resonance Imaging (MRI) of the brain with whole spine screening showed an enhancing lesion with internal blooming and signal drop in the Apparent Diffusion Coefficient (ADC) involving the posterior aspect of the lateral ventricle and splenium of the corpus callosum (Figure 3). Based on this observation, CNS lymphoma was suspected. PET-CT showed FDG uptake in bilateral tonsils and nasopharynx and enhancing lesion in the body of corpus cal-losum with involvement of ventricles which could represent relapsed lymphoma. A stereotactic biopsy of the CNS lesion showed a round cell infiltrate suggestive of large cell lymphoma (Figure 2d). IHC revealed positivity for CD20 and MUM-1 and negativity for CD3, CD10, TdT, CD5, BCL6, Cyclin D1, and c-myc. In addition, a few CD3 positive cells were seen admixed and Ki67 was found to be 50%. Based on the imaging and histopathology, the diagnosis of isolated CNS relapse of lymphoma was made. Chemotherapy with the modified DeAngelis Protocol was initiated (rituximab 700 mg, methotrexate 3.5 g, vincristine 2 mg, procarbazine 150 mg, dexamethasone in tapering doses on alternate weeks with intrathecal methotrexate for 5 cycles) following which he recovered completely. After 5 cycles of chemotherapy, consolidation radiotherapy with three-dimensional conformal radiationtherapy (3D CRT) at a dose of 36 gray in 18 fractions (200 cGy per fraction and 5 fractions a week, dose pre-scribed to 100% isodose line) to whole brain using bilateral opposed portals was given. Radiotherapy was completed in November 2018 and reassessment MRI in June 2019 showed a complete response. Currently, the patient is disease-free and on regular follow-up.
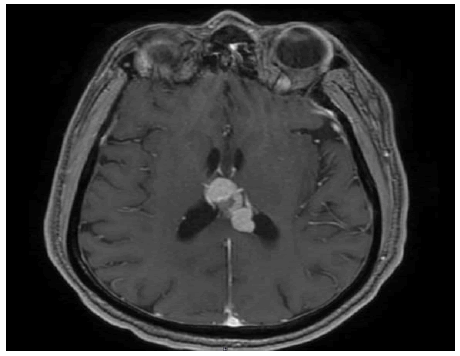
Figure 3: MRI brain which shows CNS relapse
Discussion
IVLBCL can affect almost all the organs, especially the pituitary gland, adrenal gland, kidney, skin, lungs, and CNS [4-8]. Early diagnosis of IVLBCL is challenging due to its array of nonspecific clinical symptoms. Case reports from across the world highlight the importance of its timely diagnosis and management in ensuring better survival outcomes.
The Asian variant of IVLBCL is characterized by more advanced disease symptoms, often fever, respiratory symptoms, cytopenia, and the frequent involvement of hematolymphoid organs like liver, spleen, and bone marrow with typical sparing of the skin. Additionally, hepatomegaly and/or splenomegaly were observed among most patients, whereas lymphadenopathy was less common [2]. Our patient showed the characteristic symptoms of the Asian variant, such as recurrent fever, anemia, thrombocytopenia, elevated Lactate Dehydrogenase (LDH), and ferritin, without any cutaneous manifestations. Meanwhile, most studies from Western countries, such as USA, reported intermittent fever, pancytopenia, generalized weakness, headache, arthralgia, and flu-like symptoms as commonest symptoms of IVLBCL [9,10]. Fever of unknown origin and pancytopenia justify the need for bone marrow aspiration cytology along with another infectious disease workup. However, all these substantial investigations further delay the diagnosis. In our study, the diagnosis of IVLBCL was made within a period of one month since the patient’s first consultation. Incidentally, in some of the previous reports, the diagnosis was made postmortem [5,10]. Imaging studies like PET-CT along with a histopathological examination of the affected tissue and IHC can confirm the diagnosis. Similar to our case, Li. et al reported a case in which the importance of PET-CT was highlighted in the early diagnosis of IVLBCL of the bilateral adrenal glands [11].
According to Ponzoni et al. the immunophenotype in IVLBCL shows mature peripheral B-cells with strong CD20 expression. Alternate B-cell markers like CD79a and Pax-5 can be identified in some exceptional cases in which CD20 was negative [3]. In our study, IHC-cells from the adrenal glands were positive for LCA, CD20, PAX-5, BCL2, MUM-1 and negative for CK, CD3, BCL6, CD10, and Tdt. Moreover, Ki-67 staining showed a high proliferation index of 60% focally. The neoplastic cells were seen within vascular channels, highlighted by CD34. During recurrence, a similar immune profile was seen, which was suggestive of a large B cell lymphoma. Here, the tumour cells were not restricted to the intravascular spaces and were seen to infiltrate the brain parenchyma. Such recurrences outside the vascular compartment are described in the CNS. Unlike a Japanese study that included a large series of case reports of 42 patients over 20 years, our study had distinct differences in the diagnostic tools used and the occurrence of CNS relapses. In the Japanese study, a random skin biopsy was used as a diagnostic tool among the majority of patients; moreover, CNS relapse was not common even in patients who did not receive high doses of methotrexate [12]. At the same time, our patient had a CNS relapse after 18 months of primary treatment. Even though CNS involvement is not a common characteristic of the Asian variant, our patient had a CNS relapse despite a systemic high dose of methotrexate prophylaxis.
Chemotherapy with R-CHOP is considered the standard treatment for IVLBCL [1]. A study from Italy suggested that the addition of rituximab to conventional anthracycline-based therapy showed improved survival outcomes in their patients [13]. Similarly, our patient had a progression-free period of 18 months after initial chemotherapy with R-CHOP. In a case series reported from Taiwan, six patients with rituximab-based chemotherapy achieved a 100% response rate and 75% threeyear overall survival rate [1]. Yoon et al. recently reported the median overall survival as 45 months [2]. The overall survival rate of IVLBCL in our case has been 60 months till date. The three-year overall survival rates are now as high as 60%-80% with anti CD20 chemotherapeutic regimen [14]. To the best of our knowledge, this is the first reported case from India in which IVLBCL of the adrenal gland with CNS relapse responded to second-line chemotherapy with overall survival of 60 months.
Conclusion
IVLBCL is a rare and aggressive form of extranodal non- Hodgkin’s DLBCL, which requires early identification and intervention with systemic therapy and disease management. The diagnosis is established with histopathology, IHC, and various imaging modalities. Chemotherapy with the R-CHOP regimen forms the mainstay of treatment and prognosis is largely dependent on the histological features and degree of organ involvement.
Ethics Approval
Institutional review board approval for the case report is not required at our institution. Following the ethical principles, the names of the patient were not pointed in the paper and the rights of the subject were protected. The patient received treatment consistent with the current standard of care.
Consent for Publication
Written informed consent was obtained from the patient for publication of this case report and any accompanying images.
Conflict of Interest
The author reports no conflicts of interest in this work.
References
- Ong YC, Kao HW, Chuang WY, Hung YS, Lin TL, et al. Intravascular large b-cell lymphoma: a case series and review of literatures. Biomed J. 2020;S2319-4170:30041-X.
- Yoon SE, Kim WS, Kim SJ. Asian variant of intravascular large B-cell lymphoma: a comparison of clinical features based on involvement of the central nervous system. Korean J Intern Med. 2020;35:946-956.
- Ponzoni M, Campo E, Nakamura S. Intravascular large B-cell lymphoma: a chameleon with multiple faces and many masks. Blood. 2018;132:1561-1567.
- Quintero Vega GE, Osorio D, de la Hoz Valle JA, Rodríguez Feria D. Limited Renal Intravascular Lymphoma: A Case Report and Review of the Literature. Case Rep Oncol Med. 2020;2020:7052536.
- Chen Y, Ding C, Lin Q, Yang K, Li Y, et al. Primary intravascular large B-cell lymphoma of the lung: A review and case report. J Thorac Dis. 2014;6:E242-E245.
- Renjen PN, Khan NI, Gujrati Y, Kumar S. Intravascular large B-cell lymphoma confirmed by brain biopsy: a case report. BMJ Case Rep. 2013;18; 2013:bcr2012007990.
- Anila KR, Nair RA, Koshy SM, Jacob PM. Primary intravascular large B-cell lymphoma of pituitary. Indian J Pathol Microbiol. 2012;55:549-551.
- Biji KA, Leenadevi KR. Intravascular large B cell lymphoma. Indian J Pathol Oncol. 2019;6:506-508.
- Verma A, Sharma A, Robetorye R, Porter A, Hilal T. Intravascular large b-cell lymphoma associated with systemic and central nervous system hemophagocytic lymphohistiocytosis: a case report. Perm J. 2020;24:105.
- D’Angelo CR, Ku K, Gulliver J, Chang J. Intravascular large b-cell lymphoma presenting with altered mental status: a case report. World J Clin Oncol. 2019; 10:402-408
- Li W, Lin W, Ma C, Zhang L, Sun H. A case of intravascular large B-cell lymphoma in the left adrenal and another tumor in the right adrenal detected by (18)F-FDG PET/CT. Hell J Nucl Med. 2016;19:57-59.
- Matsue K, Abe Y, Narita K, Kobayashi H, Kitadate A, et al. Diagnosis of intravascular large B cell lymphoma: novel insights into clinicopathological features from 42 patients at a single institution over 20 years. Br J Haematol. 2019;187:328-336.
- Ferreri AJM, Dognini GP, Bairey O, Szomor A, Montalbán C, et al. The addition of rituximab to anthracycline-based chemotherapy significantly improves outcome in “Western” patients with intravascular large B-cell lymphoma. Br J Haematol. 2008; 143:253-257.
- Liu Z, Zhang Y, Zhu Y, Zhang W. Prognosis of intravascular large B cell lymphoma (ivlbcl): analysis of 182 patients from global case series. Cancer Manag Res. 2020; 12:10531-10540.



