Research Article - Onkologia i Radioterapia ( 2023) Volume 17, Issue 10
Apoptotic effects of Phenolic extract from Spirulina platensis on Esophagus cancer cell line SK-GT-4
Rasha. N. Aljabery1*, Mohammed A. Auda1 and Hussain Yosif Al-Rekabi22Technical Institute Al-Nassiriah, Southern Technical University, Iraq
Rasha. N. Aljabery, Department of Chemistry, College of Science, University of Thi-Qar, Thi-Qar, 64001, Iraq, Email: rasha_na.ch@sci.utq.edu.iq
Received: 07-Sep-2023, Manuscript No. OAR-23-113027; Accepted: 03-Oct-2023, Pre QC No. OAR-23-113027 (PQ); Editor assigned: 09-Sep-2023, Pre QC No. OAR-23-113027 (PQ); Reviewed: 22-Sep-2023, QC No. OAR-23-113027 (Q); Revised: 30-Sep-2023, Manuscript No. OAR-23-113027 (R); Published: 08-Oct-2023
Abstract
Due to their strong antioxidant qualities and their notable impact in the prevention of numerous oxidative stress-related disorders like cancer, phenolic extract from Spirulina platensis has attracted increasing attention. Phenolic compounds were extracted from spirulina platensis by using an ultrasonic-assisted extraction method. The yield of Phenolic extract by this method was (0.21% w/w). Additionally, phenolic components for phenolic extract were identified using the HPLC technique. The findings indicated that the extract contained a number of important compounds, including vanillic acid, gallic acid, Ferulic acid, cinnamic acid. Then, using a fluorescent microscope, the Acridine-orange-Ethidium bromide (AaO/ EtBr) double labelling was utilized to identify the alterations in nuclear morphology during apoptosis. SK-GT-4 cells were exposed to phenolic extract for 72 hours. The findings demonstrated that treatment with the extract causes somewhat more apoptosis in treated cells than in untreated cells.
Keywords
spirulina platensis, acridine-orange-ethidium bromide, apoptosis, SK-GT-4
Introduction
Cancer is a category of diseases characterized by unchecked cell development and the spread of aberrant cells, which results in a mass of cells known as a neoplasm or tumour. If the spread is unchecked, this condition could ultimately result in death [1]. One characteristic of cancer is the quick development of aberrant cells that expand outside of their normal borders, infiltrate other body components, and eventually move to other organs. This process is known as metastasis. The main reason for dying from cancer is widespread metastases [2]. Oesophageal cancer is also one of the main causes of cancer-related death globally [3, 4]. It is a tumour that develops in the oesophageal lining. Adenocarcinoma is a type of cancer that develops in the cells responsible for producing and secreting mucus and other fluids [5]. Phenolic extracts play an important role in preventing cancer, simple phenols, flavonoids, lignins and lignans, tannins, xanthones, and coumarins are among the phenolic chemicals identified from algal sources [6]. These phenolic compounds have been shown to have strong anti-cancer properties as well as to be effective against a number of other disorders that are oxidative stress-related [7]. Numerous investigations have shown that dietary phenols' protective benefits on health are caused by their anti-inflammatory and antioxidant properties [8]. The main mechanism by which phenolic compounds have anti-carcinogenic effects is their capacity to induce cell cycle arrest and inhibit oncogenic signalling cascades regulating cell proliferation, angiogenesis, and apoptosis. Other mechanisms include modulating ROS levels, promoting tumour suppressor proteins like p53, and enhancing the capacity to differentiate and transform into normal cells. According to, Spirulina platensis phenolic compounds exhibit anticancer action against the human fibroblast normal cell line HepG2, but have no effect on normal human fibroblasts [9]. Furthermore, flavonoids, phenolic compounds, or polyphenols interact with enzymes and proteins in a number of biological processes that either render them toxic to cells or prevent cell growth [10, 11]. The improvement of the efficiency of standard chemo and radio therapeutic treatment regimens and the prevention of resistance to these therapies are additional major effects of phenolic compounds that warrant further research [12]. The ability of phenolic compounds to actively act as free radical scavengers, reducing agents, singlet oxygen quenchers, metal chelators, and hydrogen donors has recently attracted research attention. This is primarily due to the redox properties of their hydroxyl groups and the structural relationship between different functional groups in their structure [13]. The aim of this research was to discover bioactive natural products from Spirulina platensis in order to implement an alternative strategy in the treatment of oesophageal malignancies given that the active phenolic compound that was extracted from Spirulina platensis has been documented to have potential anticancer activity
Methods
Spirulina platensis
Spirulina platensis was purchased in powder form ALIBAB Company and was stored in air tight packages at room temperature in the dark prior to use.
Extraction of phenolic compounds
With slight modifications, the idea and method established by Kredy et al. were used to the extraction process. 500 mL of 70% ethanol were added to each brown bottle containing 40 g of Spirulina platensis [14]. The mixture was placed in an ultrasonic bath, and then, using a vacuum pump, was filtered through a Buchner funnel using Whatman No. 1 filter paper. The filtrate solution was then dried after being evaporated via a rotary evaporator with reduced pressure at 40°C and then dried. After that, the alcoholic extract is diluted in 30 ml of distilled water, and 50 ml of chloroform is added to it in a separation funnel to separate it into two layers (water and organic layer). This process is repeated to add 50 ml of chloroform to the aqueous layer, and the mixture is stirred for 48 hours on a magnetic stirrer. The aqueous layer was separated when the mixture was transferred to the separating funnel. The four-step aqueous layer was then extracted using the same volume of ethyl acetate while being stirred for 48 hours on a magnetic stirrer. The mixture was then transferred to the separating funnel for layer separation, producing two layers (aqueous and ethyl acetate layer). The ethyl acetate layer was then separated and dried to produce the phenolic extract.
Identification of phenolic compounds by using the High Performance Liquid Chromatography (HPLC) technique
In laboratories of the Ministry of Science and Technology, phenols extracted from a Spirulina platensis were identified using a method called HPLC (HighPerformance Liquid Chromatography) column. Individual phenolic components were quantified using reversed-phase HPLC processing utilizing a SYKAMN HPLC chromatographic apparatus equipped with a UV detector, Chemstation, and a Zorbax Eclipse Plus-C18- OSD. 25 cm, 4.6 mm column The column had a temperature of 30°C. The gradient elution procedure was carried out as follows: initial 0 min-4 min, 40% B; 4 min-10 min, 50% B; and flow rate of 0.7 mL/min. Eluents A (methanol) and B (1% formic acid in water (v/v)) were used. 100 litres of samples and 100 litres of standards were automatically injected using an auto sampler. The 280 nm range was used to acquire the spectra [15].
Cell line and culture
Esophagus cancer cell line SK-GT-4 was obtained from the tissue culture laboratory in the College of Education for Pure Sciences of Basra University. The cells were cultured in RPMI-1640 medium containing 10% FBS, 100 V/ml of penicillin, and 100 μg/ml of streptomycin at 37°C in a humidified 5% CO2 incubator.
Acridine orange/ethidium bromide (AO/EB) double staining method
According to, the Acridine Orange/Ethidium Bromide double staining (AO/EB) assay was carried out [16]. The esophageal cancer cell line SK-GT-4 was grown in 6-well plates and incubated overnight. The cell suspension (3 × 104 cells/well) of cancer cell line SK-GT-4 were cultured in 6-well plates and incubated overnight. After that, 36.5 g/ml of phenolic extract was added to the cells, and incubated for 24 hr. Cells were harvested, washed in Phosphate Buffer Saline (PBS), and stained with a dye solution containing acridine orange and ethidium bromide (1:1, 100 g/mL each). A clean microscope slide was used for the stained cell solution (10 L), which was then immediately viewed using a fluorescence microscope.
Results
Algae extracts
As shown in Table, the weight of dry powder material of Spirulina platensis was (40 g). The product of dry powder phenolic extracts (0.084 g) (Table 1). Extraction percentage of extract was calculated from equation as
Tab. 1. Percentage of extracts
| Algae Weight (g) | 40 |
|---|---|
| phenolic extract (g) | 0.084 |
| Percentage (%) | 0.21 |
Extraction
Determination of Phenolic Compounds in Phenolic Extract by HPLC Technique
In order to analyse phenolic compounds, High Performance Liquid Chromatography (HPLC) is combined with various detectors, such as UltravioletVisible (UV) and Photodiode Array Detector (PDA) [17]. HPLC is a separation and characterization method that has been widely accepted by many scientists. The extract was examined to determine how many phenolic chemicals were present. The retention time of each component was compared to pure commercial standards in order to identify it [18]. The figures explained that the retention time of sample agrees with the standard retention time of most contents in the extract, and the tables showed the retention time of standards and the phenolic compounds (Figures 1-6) (Tables 2-7).
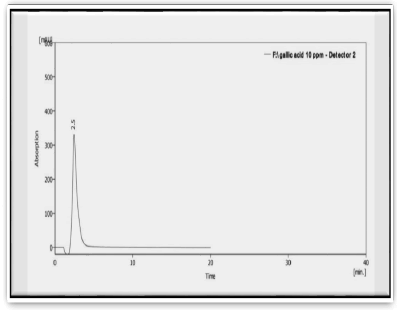
Figure 1: HPLC of standard Gallic acid compound
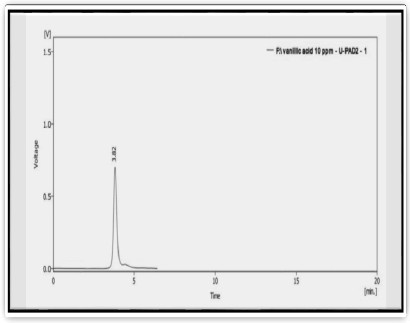
Figure 2:HPLC of standard Vanillic acid compound
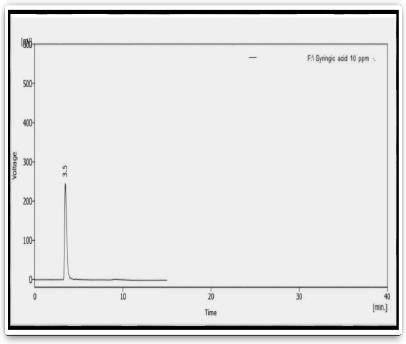
Figure 3: HPLC of standard Syringic acid compound
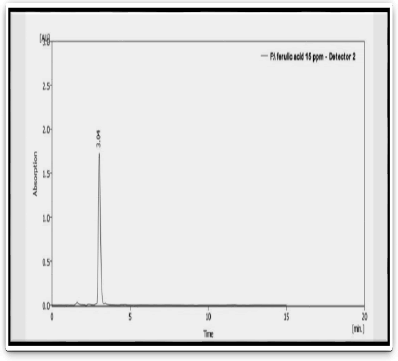
Figure 4: HPLC of standard Ferulic acid compound
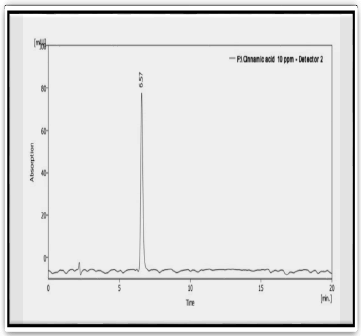
Figure 5: HPLC of standard Cinnamic acid compound
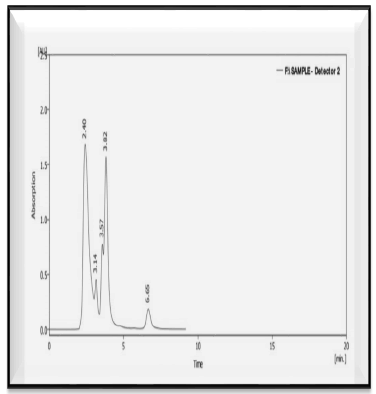
Figure 6: HPLC of phenolic extract of Spirulina platensis
According to this study's findings and HPLC analysis, Spirulina platensis phenolic extract contains several important compounds, including gallic acid, Ferulic acid, cinnamic acid, syringic acid, and vanillic acid.
Tab. 2. The retention time of standard gallic acid compound
| Peak | Standard | Retention time (min) | Area |
|---|---|---|---|
| 1 | Gallic acid | 2.453 | 476.079 |
Tab. 3. The retention time of standard vanillic acid compound
| Peak | Standard | Retention time (min) | Area |
|---|---|---|---|
| 1 | Vanillic acid | 3.82 | 318.876 |
Tab. 4. The retention time of standard syringic acid compound
| Peak | Standard | Retention time (min) | Area |
|---|---|---|---|
| 1 | Syringic acid | 3.464 | 675.603 |
Tab. 5. The retention time of standard Ferulic acid compound
| Peak | Standard | Retention time (min) | Area |
|---|---|---|---|
| 1 | Ferulic acid | 3.037 | 2159.052 |
Tab. 6. The retention time of standard cinnamic acid compound
| Peak | Standard | Retention time (min) | Area |
|---|---|---|---|
| 1 | Cinnamic acid | 6.573 | 139.672 |
Tab. 7. The concentration of phenolic compounds in phenolic extract of Spirulina platensis
| Peak | Phenolic contents in the extract | Retention time (min) | Area | Conc. of phenolic compounds (µg / gm) |
|---|---|---|---|---|
| 1 | Gallic acid | 2.4 | 46074.82 | 52.9 |
| 2 | Vanillic acid | 3.817 | 28121.595 | 14.58 |
| 3 | Syringic acid | 3.57 | 8342.55 | 12.5 |
| 4 | Ferulic acid | 3.143 | 6239.002 | 74.5 |
| 5 | Cinnamic acid | 6.653 | 3920.33 | 36.9 |
Study of apoptosis using Acridine Orange and Ethidium Bromide AO/EB double staining method
By using fluorescence microscopy to identify contracted nuclei that were doubly stained with acridine orange and ethidium bromide, apoptosis was identified in this work. Acridine orange/ethidium bromide dye can be used to distinguish between apoptosis, necrosis, and living cells. Apoptotic cells now have a yellow-orange shape instead of the green colour of living, healthy cells. Red is the colour of necrotic cells [19]. While EB is exclusively taken up by dead cells and displays red fluorescence after DNA intercalating, AO penetrates both living and dead cells and emits green fluorescence [20]. It should be remembered that healthy cells contain intact cell membranes, evenly green nuclei, and chromatin. Additionally, necrotic cells have red nuclei with damaged cell membranes, while apoptotic cells have orange nuclei with condensed chromatin [21].
Through the use of the AO/EB stain and phenolic extract, the current investigation demonstrated the presence of apoptosis in cancer cells SK-GT-4. The cells were separated into four groups: necrotic cells (uniformly orange-stained cell nuclei), late apoptotic cells (bright green nuclei with condensed or fragmented chromatin), living cells (normal green nucleus), and early apoptotic cells (bright green nuclei with condensed or fragmented chromatin). Figure illustrates how untreated cancer cells appear green when stained with AO/EB and cannot see any of apoptosis processes (Figure 7). The majority of the phenolic extract-treated cells were stained with EB stain, which glows orange under a fluorescence microscope as seen in (Figure 8). The EB stain can now penetrate within the cells because of the disruption to the cells' membranes, which suggests that the cells have reached a late stage of apoptosis [22]. These findings confirmed that the phenolic extract treatment caused the esophageal cancer cell line SK-GT-4 to undergo apoptosis induction. According to some research, phenol extracts may encourage the death of cancer cells by triggering the release of cytochrome c from the mitochondria and the activation of caspases 3, 8, and 9, while also suppressing the expression of Bcl-2 and BclXL through altering NF-κB signalling [23]. On the other hand, by lowering the mitochondrial membrane potential, various phenolic acids found in the phenolic extract, including gallic acid, Ferulic acid, cinnamic acid, syringic acid, and vanillic acid, cause apoptosis and increase lactate dehydrogenase levels. Additionally, these substances cause DNA breakage in cell lines of breast, lung, liver, and prostate cancers [24].
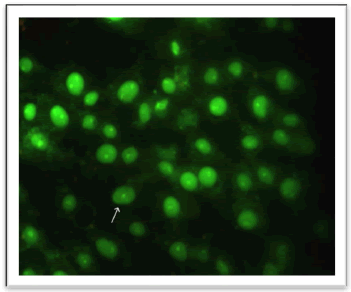
Figure 7: Fluorescent Microscope of SK-GT-4 Cell Line of untreated. The cell healthy (white arrow) (40X)
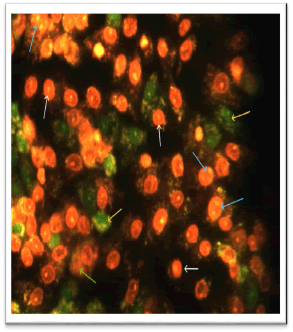
Figure 8: Fluorescent Microscope of SK-GT-4 Cell Line with IC50 of phenolic extract of Spirulina platensis for 72hr. The cell death (white arrow), the remains of dead cells (green arrow), the fragmentation of nucleus (blue arrow) and the late stage of apoptosis (yellow arrow) (40X)
Conclusion
The study's findings demonstrated that Spirulina platensis's phenolic extract increased the amount of intracellular free radicals, which resulted in DNA damage and caused the cancer cells SK-GT-4 to undergo apoptosis. Phenolic acids, which are present in the extract, are assumed to be the cause of this action. These findings demonstrate that, in the cell line of esophageal cancer, phenolic extract is a potent inhibitor of cell proliferation through apoptotic effects.
Acknowledgments
This work is supported by the University of Thi-Qar as a part of higher studies and research Projects.
References
- Mohseni N, Sarvestani FS, Ardestani MS, Kazemi-Lomedasht F, et al. Inhibitory effect of gold nanoparticles conjugated with interferon gamma and methionine on breast cancer cell line. Asian Pac J Trop Biomed. 2016;6:173-8.
- Hanahan D, Weinberg RA. Hallmarks of cancer: the next generation. cell. 2011;144:646-474.
- Fitzmaurice C, Allen C, Barber RM, Barregard L, Bhutta ZA, et al. Global, regional, and national cancer incidence, mortality, years of life lost, years lived with disability, and disability-adjusted life-years for 32 cancer groups, 1990 to 2015: a systematic analysis for the global burden of disease study. JAMA oncol. 2017;3:524-548.
- Inhibitory effect of gold nanoparticles conjugated with interferon gamma and methionine on breast cancer cell line
- Thrift AP, Whiteman DC. The incidence of esophageal adenocarcinoma continues to rise: analysis of period and birth cohort effects on recent trends. Ann oncol. 2012;23:3155-3162.
- Huang WY, Cai YZ, Zhang Y. Natural phenolic compounds from medicinal herbs and dietary plants: potential use for cancer prevention. Nutr cancer. 2009;62:1-20.
- Arts IC, Hollman PC. Polyphenols and disease risk in epidemiologic studies. Am j clin nutr. 2005;81:317-325.
- Lambert JD, Hong J, Yang GY, Liao J, Yang CS. Inhibition of carcinogenesis by polyphenols: evidence from laboratory investigations. Am J clin nutr. 2005;81:284-291.
- Akbarizare M, Ofoghi H, Hadizadeh M, Moazami N. In vitro assessment of the cytotoxic effects of secondary metabolites from Spirulina platensis on hepatocellular carcinoma. Egypt Liver J. 2020;10:1-8.
- Weber G, Shen F, Prajda N, Yeh YA, Yang H, et al. Increased signal transduction activity and down-regulation in human cancer cells. Anticancer res. 1996;16:3271-3282.
- Fotsis T, Pepper MS, Aktas E, Breit S, Rasku S, et al. Flavonoids, dietary-derived inhibitors of cell proliferation and in vitro angiogenesis. Cancer res. 1997;57:2916-2921.
- Azmanova M, Pittoâ?Barry A. Oxidative stress in cancer therapy: friend or enemy? Chembiochem. 2022;23:202100641.
- Mahmoud M, Mohamed EM, Aboul-Enein AM, Diab AA, Shalaby EA. Anticancer and antioxidant activities of ethanolic extract and semi-purified fractions from guava and mango seeds. Biomass Convers Biorefinery. 2023:1-7.
- Kredy HM, Braige RH, Khathi MT. Extraction of Poly Phenols from Cladium Mariscus Seeds and studying its Antifungal Activity with Isolation and Identification of one Compound of Flavone Glycosides.
- Radovanovic B, Mladenovic J, Radovanovic A, Pavlovic R, Nikolic V. Phenolic composition, antioxidant, antimicrobial and cytotoxic activites of Allium porrum L.(Serbia) extracts. J Food Nutr Res. 2015;3:564-569.
- Musa MA, Kolawole Q. 7, 8-Diacetoxy-3-(4-methylsulfonylphenyl)-4-phenylcoumarin Induces ROS-dependent Cell Death in the A549 Human Lung Cancer Cell Line. Anticancer Res. 2023;43:1001-1007.
- Shi L, Zhao W, Yang Z, Subbiah V, Suleria HA. Extraction and characterization of phenolic compounds and their potential antioxidant activities. Environ Sci Pollut Res. 2022:81112-81129.
- Marko-Varga G, Barceló D. Liquid chromatographic retention and separation of phenols and related aromatic compounds on reversed phase columns. Chromatographia. 1992;34:146-154.
- Zhang Q, Ruan F, Yang M, Wen Q. Natural compound chaetocin induced DNA damage and apoptosis through reactive oxygen speciesâ?dependent pathways in A549 lung cancer cells and in vitro evaluations. IET nanobiotechnology. 2023;17:465-475.
- Kari S, Subramanian K, Altomonte IA, Murugesan A, Yli-Harja O, et al. Programmed cell death detection methods: a systematic review and a categorical comparison. Apoptosis. 2022;27:482-508.
- Cohen C. Apoptosis.
- Abolhasani MH, Safavi M, Goodarzi MT, Kassaee SM, Azin M. Identification and anti-cancer activity in 2D and 3D cell culture evaluation of an Iranian isolated marine microalgae Picochlorum sp. RCC486. DARU J Pharm Sci. 2018;26:105-116.
- Li J, Yuan J. Caspases in apoptosis and beyond. Oncogene. 2008:6194-6206.
- Yin MC, Lin CC, Wu HC, Tsao SM, Hsu CK. Apoptotic effects of protocatechuic acid in human breast, lung, liver, cervix, and prostate cancer cells: potential mechanisms of action. J agric food chem. 2009;57:6468-6473.



