Case Report - Onkologia i Radioterapia ( 2020) Volume 14, Issue 6
Acute skin toxicity after contact brachytherapy for t lymphoma in the foot and ankle a case report and review of the literature
Fadwa Allouche1*, M El Baraka2, Fz Terrab1, H Kabbaj1, Ma Youssoufi2, F Farhane1, Z Alami1, T Bouhafa1 and K Hassouni12Medical Physics Unit, Fez Hassan Ii University Hospital Center, Morocco
Fadwa Allouche, Department of Radiation Oncology, Fez Hassan Ii University Hospital Center, Morocco, Email: dr.allouch.fadwa@gmail.com
Received: 29-Oct-2020 Accepted: 16-Nov-2020 Published: 30-Nov-2020
Abstract
Cutaneous T lymphoma is a relatively common type of Non-Hodgkin Lymphoma (NHL). It originates from mature T lymphocytes in the skin and takes on several anatomical and clinical forms which can be very aggressive. Contact brachytherapy is used to treat cancer of the skin in various places. In areas of poor vascularity, subject to constant trauma, such as extremities, radiotherapy can lead to prolonged scarring, chronic poorly treatable ulcerations, or even necrosis, requiring subsequent surgery. In this case study, we report on a young patient with cutaneous T-type mycosis fungoid lymphoma located on the right foot who was treated with contact brachytherapy. The cancer was cured, but the patient developed complex and acute skin toxicity which finally resolved after 8 weeks.
Keywords
T lymphoma, contact brachytherapy, toxicity, foot ulcer
Introduction
Cutaneous T cell lymphomas (CTCL) are a group of neoplastic T cell proliferations that should be distinguished from ganglion lymphomas. These are clonal growths which infiltrate mainly through the skin reach the lymph nodes only at an advanced stage and infiltrate the bone marrow only exceptionally. They are rare, representing only 2% of all lymphomas with an estimated annual incidence of 1-2/100,000. They differ from lymphoma lymph nodes by their biological and clinical behavior, as well as by their prognosis which is generally much better than that of lymph node lymphoma [1]. There is no treatment for CTCL which can be considered "strictu-sensu" as curative at present. The treatments available are aimed at achieving and prolonged remissions. They are numerous, but only a minority has been validated by controlled studies. This is due to the fact that the rarity of cutaneous lymphomas and their slow progression (years) require large studies with prolonged follow-up, which is difficult to achieve in practice. It has nevertheless been clearly demonstrated that the survival of patients suffering from CTCL is not prolonged by multidrug therapy [2]. “Softer” therapies, either local for cutaneous use, or systemic, modulation of the biological response is therefore preferred, and this in particular at the early stages where the anti-tumor immune response is always preserved. Among the treatments proposed for this pathology that we mention contact brachytherapy. Brachytherapy is the delivery of radiation through flexible catheters connected to a programmable source and has been used for the treatment of multiple skin malignancies, including advanced basal cell and squamous cell carcinomas and Merkel cell carcinoma [3, 4]. The use of fully customized molds in brachytherapy allows precise control of the depth of penetration of radiation to complex convex surfaces.
Case Report
This is a 29-year-old patient, treated for hepatitis B in 2016, presenting for 2 years granulomatous erythematous lesions progressively increasing in size, involving the right foot and ankle with on examination a well-limited subcutaneous tumor, of firm consistency next to the right Achilles heel 14x3 cm and ictyosiform scales next to the tumors of the 2 feet, with cutaneous T lymphoma type mycosis fungo biopsy. When examining the lymph node areas was not lymphadenopathy. The patient underwent a biological workup, including complete blood count, electrolytes and basal metabolic profile were normal. The patient underwent a thoraco-abdominal-pelvic CT scan of the, with no sign of distant metastasis. Patient classified T3N0M0, stage IIb. He benefited from 34 sessions of Puvatherapy - 10 sessions of phototherapy in the dermatology department, May without improvement. Then he began systemic treatment with methotrexate 25 mg/week, he had not responded to methotrexate hence the indication for radiotherapy retained in him. The patient received High-Rate Contact Brachytherapy (HDR), with a total dose of 30Gy in 5 fractions, 6Gy/fraction, 2 fractions per day at 6-hour intervals. The application was made with a Catheter Flap-Set type applicator (Figure 1) by the Gamma Medplus machine from VARIAN.
Figure 1: Application by a catheter flap set
After a detailed discussion with the patient regarding possible treatment options, he agreed to High-Flow Contact Brachytherapy (HDR) and was referred to the Brachytherapy Department at HASSAN II University Hospital, FEZ. The patient consented in writing to take a medical illustration and agreed to publish his case report, including photographs. The patient specifically agreed to the case being used for educational purposes. The rationale and logistics for cutaneous BT, as well as possible acute and chronic side effects of treatment were discussed in detail and the patient provided written authorization for cutaneous BT. Due to the location of the foot upon consent, the patient was particularly advised of an increased risk of longterm toxicity with possible non-healing skin ulceration in the BT site, requiring long-term dressings and/or surgery, he was also advised on skin care during and after BT, and received written information on skin radiation therapy, In skin care advice, patients are encouraged to use any moisturizer according to the patient's wish, provided that it does not cause any contact allergy. A moisturizer for the skin should be applied during and after radiation therapy.
Brachytherapy technique
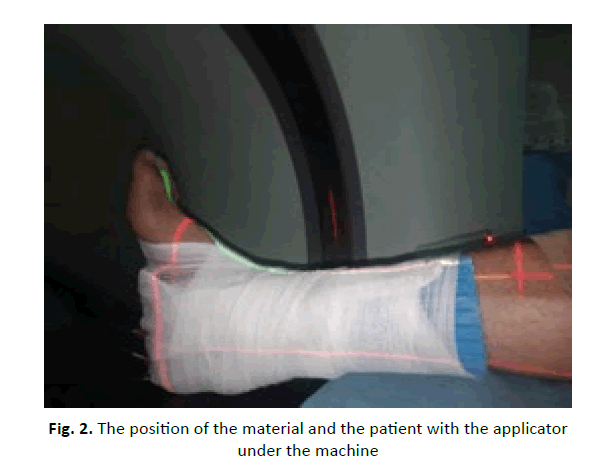
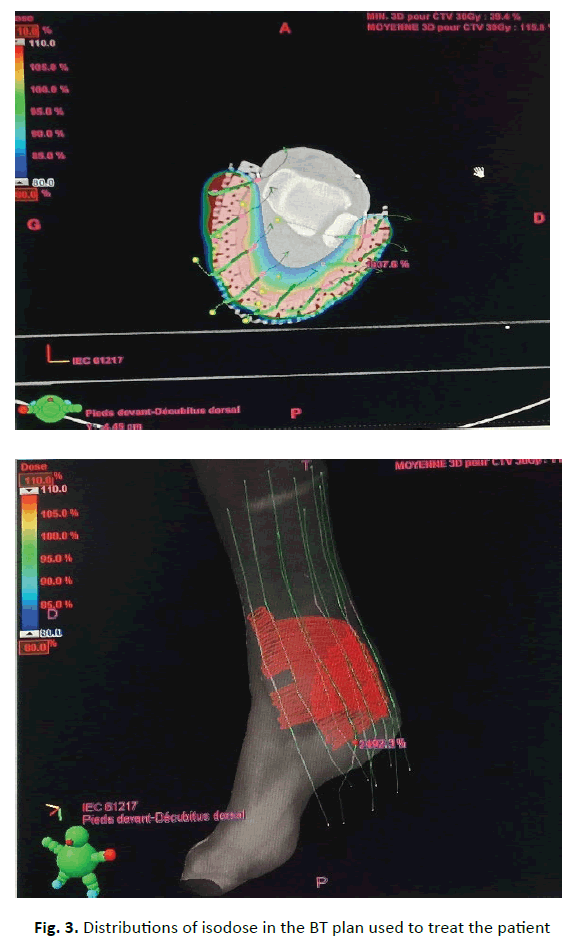
The catheter flap set is used to treat cancer. It is designed to create a defined space between source and tissue, and between source channels. The highly flexible material easily adapts to complex anatomical shapes. The catheter channels have a diameter of 2 mm and are positioned on the median axis of the flap with a bolus of 4 mm. The catheter flap is often affixed to a Thermoplastic Mesh (TM), commonly used in radiation therapy, to maintain a reproducible orientation relative to the patient's anatomy. The TM material was heated and formed around the patient's ankle and foot. The catheter was attached to the TM. This TM design and the position of the Flap catheter allowed the Ir-192 HDR source to travel in close proximity to the skin tissue to be treated. To ensure good visualization, metal markers (lead wires) were placed at the edge of the tumour area and in the tubes. During the planning scanner, the correct positioning of the material with the applicator was checked in order to avoid misplacement, Planning based on three-dimensional Computed Tomography (CT) was performed using 3mm CT sections of thickness, Images were imported into Eclipse (Varian Medical Systems, Palo Alto, CA)(Figure 2). The Planning Treatment Volume (PTV) was described or "Contoured" on the CT images (Figure 2). The patient received treatment according to plan and tolerance was good. Treatment Planning (TPS) was performed on the VARIAN Gamma Medplus machine. The plan was evaluated using the slice-by-slice visualization. The technique at the time of this treatment was to prescribe at the skin surface and cover the treatment volume with the 80% isodose line. Currently. The dose was prescribed at a depth of 3 mm. The patient received HDR-BT. Due to the location of the lesion on the foot, the prescribed standard dose of 30Gy in 6Gy/fraction (equivalent of 32 Gy at 100% isodose) was reduced in this case to 37.50 Gy at 80% (equivalent of 30 Gy at 100 (Table 1). The dose was delivered in five fractions, twice daily, with a minimum break of six hours between fractions over two weeks. The validation of the dosimetry was passed without any problems and the patient was fully compliant the parameters of the HDR-BT plan are shown in Table 1.
Figure 2: The position of the material and the patient with the applicator under the machine
Figure 3: Distributions of isodose in the BT plan used to treat the patient
| Processing details | HDR contact brachytherapy to the skin on the right foot + ankle |
| The prescribed dose | 30Gy at the surface of the skin 05fractions of 06Gy per fraction* The technique at the time of this treatment was to prescribe at the skin surface and cover the treatment * The dose expected to cover the treatment volume is 80% 30 Gy volume with the 80% isodose line |
| Target volume | CTV = GTV PTV = CTV + 0.5 (volume 56.69cc) The dose was prescribed at 3mm depth |
| Number of active catheters used | 9 |
| Treatment plan parameters | D90=5.46Gy V100=47cm3 D95=5.69 Gy V95=50cm3 |
| Maximum dermal dose | The maximum cutaneous dose is 110% and is located at the centre of the lesion and not at the level of healthy skin. The maximum dermal dose in a small area of â??â??healthy skin around the lesion does not exceed 95% |
Tab. 1. Treatment plan parameters for HDR skin brachytherapy
Discussion
Cutaneous T-cell lymphomas (CTCL) are a group of extra-nodal non-Hodgkin lymphomas characterized by skin infiltration of malignant monoclonal T cells. They typically afflict adults with a median age of 55 to 60 years and the annual incidence is approximately 0.5 per 100,000. Mycosis fungoides (MF), Sézary syndrome (SS) and primary cutaneous peripheral T lymphocytes non specified elsewhere are the most important subtypes of CTCL. CTCL is a complex concept in terms of etiopathogenesis, diagnosis, therapy and prognosis [5]. A lymphoma is considered to be mainly cutaneous if it meets certain well-defined criteria of diagnostic clinical pathology and does not present a localization during the initial evaluation of the extension. The main types of CTCL are: mycosis fungoides (44% of cutaneous lymphomas, PCL), Sézary syndrome (2% of PCL), skin T cells with large CD30 + cells (9% of PCL), and lymphomas Skin cells with large CD30- cells (6% of the LCP). Mycosis Fungoides (MF) is the first described of the lymphomas. It is characterized by a slow evolution three clinical phases: erythema in non-infiltrated plaques, infiltrated plaques, nodules and tumors. Its five-year survival rate is high (87%). Sézary syndrome is defined by the association of erythroderma due to diffuse infiltration of the skin by neoplastic T lymphocytes associated with a large number (1000/mm3) of circulating neoplastic T lymphocytes and polyadenopathy. The prognosis for Sézary syndrome is more severe than that of MF, its fiveyear survival rate being 11% CD30 + large cells Cutaneous T lymphoma often presents with a red skin tumor (s) and immediately infiltrates with a cytological aspect of high-grade lymphoma (large anaplastic, immunoblastic or pleomorphic cells). These disturbing clinical and pathological aspects contrast with its favorable prognosis (>90% at five years survival), hence the importance of recognizing and distinguishing it from secondary skin involvement of T lymphoma of the lymph nodes and other CD30 cutaneous T lymphomas. - Cutaneous large cell T lymphomas are rapidly growing tumors clinically similar to cutaneous CD30 + large T cell lymphomas, but made up of large CD30-T T cells. The absence of expression of the CD30 lymphocyte marker and a markedly worse prognosis (15% five-year survival) distinguish this cutaneous lymphoma from large CD30 + T lymphoma. It also demonstrates that CD30 is an immunophenotypic marker with high prognostic value in cutaneous T lymphomas. Since 1997, a classification intended exclusively for cutaneous lymphomas have been developed by experts in cutaneous lymphomas within the framework of the European Organization for the Research and Treatment of Cancers (EORTC) [6]. This EORTC classification, which is very close to the most recent WHO classification, has the advantage of distinguishing the different types of cutaneous lymphomas according to their histopathology, clinical characteristics and their prognosis (indolent, intermediate or aggressive)[7]. This clinical pathological classification allows in most cases to quickly establish a prognosis, and therefore facilitates the choice of the optimal treatment [8]. As with lymph node lymphoma, clinical staging is performed according to the TMN system. It includes a skin and systemic examination, laboratory examinations and radiology evaluation. The diagnosis of CTCL is difficult at an early stage due to the presence of multiple clinical presentations and the absence of definitive diagnostic criteria [9-12]. Therefore, in most cases it takes an average of 6 years from the onset of the disease until the diagnosis is confirmed [9-11]. Recently they have made progress in the accurate diagnosis of CTL. To diagnose CTCL, guidelines prepared by the National Comprehensive Cancer Network recommends biopsy of suspicious skin sites and subsequent evaluation in terms of dermatopathology, immunohistochemistry, and molecular analysis (TCR gene rearrangement) [13]. Observation and palpation of the skin is an essential part of suspecting CTL. Palpation of the lymph nodes remains the traditional approach for staging these disorders [12-14]. Frequently, many biopsies are necessary to make the definitive diagnosis, as the morphological and phenotypic manifestations of CTL are variable and information derived from a single biopsy can lead to a misdiagnosis [12-16]. Identifying malignant cells in the peripheral blood of patients with CTL is invaluable for detecting SS at an early stage and determining prognosis [17, 18]. However, the blood test is of limited value because there is no precise marker in this test to detect CTCLs sensitively. Lactate Dehydrogenase (LDH) is a nonspecific marker of tumor burden and is linked to a prognosis of CTCL [13, 19]. These studies provide a robust technique for the assessment of gene aberration in CTCLs [20, 21]. The detection of a malignant T cell clone is a critical marker for a definitive diagnosis of CTCL [5]. Soluble T cell specific IL-2 receptor (sIL-2r) is not specific for the diagnosis of CTCL but is a potential marker of the activity, severity and prognosis of this disorder. The association between an increase in sIL-2r and either adnexal disease or advanced FM has been reported. This factor has better specificity as a prognostic factor than LDH [19]. Magnetic Resonance Imaging (MRI) or Computed Tomography (CT) is used to investigate lymph node and systemic involvement [14-19]. Fluorodeoxyglucose positron emission Fluoride-18 computed tomography-CT (18F-FDG PET-CT) can determine the and extra-cutaneous lesions in CTL, response to treatment, and disease recurrence. In comparison with computed tomography, this modality is more sensitive and specific for detecting both cutaneous and extra cutaneous involvement, in particular for determining lymph node involvement [14]. Among the therapeutic arsenals currently available, a distinction is made between local treatments for cutaneous use, systemic treatments which modulate the biological response, and systemic cytotoxic treatments. These are used alone or sometimes in combination, with the aim of maximizing the chances of complete remission while preserving immunity, since the anti-tumor immune response is a factor naturally limiting the progression of CTCL. The choice of the type of therapy mainly takes into account the histopathological type and the stage of lymphoma, the latter integrating both cutaneous and extra cutaneous extension. However, you should be aware that the choice between treatments of comparable effectiveness is often based on availability and local habits. This is particularly the case with treatments intended strictly for the skin in the early stages of mycosis fungoides. Several treatments are available and can be used alone or in combination:
a) Local skin treatments: topical corticosteroids, carmustine, mechlorethamine, radiotherapy, electron baths, phototherapy (PUVA)
b) Modulators of the biological response: extracorporeal photopheresis, interferon alpha, interferon gamma, retinoids (neotigason® acitretin, bexarotene, Targretin®)
c) Cytostatics
Radiation therapy is an effective skin therapy for the treatment of CTCL [5,22,23]. Lymphocytes are sensitive to radiation therapy. In more advanced cases, radiation therapy to local lesions or to the entire skin can control the disease. For cases with a single lesion, this modality can be curative [5,22]. Radiation therapy to the limbs in particular has been avoided, because there is a risk of osteonecrosis [24,25]. Brachytherapy provides better dose delivery than external beam radiation therapy due to rapid dose escalation and the ability to spare fragile normal tissue. Areas of poor vascularity, such as the lower limbs, are subjected to constant trauma. In such places, radiation therapy can lead to prolonged scarring. External beam radiation therapy, especially X-ray treatment, some authors also raise concerns about the risk of osteonecrosis or chondronecrosis. Such risks have been of particular concern in injuries to the anterior tibia or hand, or in locations of the ear, where radiation damage could result in injury to tendons, joints and bones, with impaired hand function. [26]. Electron Beam Radiation Therapy (EBRT) is effective treatment of CTCL [5,24-27] in stages I to III [5,25]. Whole-body total electron beam from the skin is a suitable modality for more advanced cases [5,9,25]. The complete response rate is lower in tumor stage disease compared to plaque stage cases (36% vs. 98.3%) [24]. X-ray radiotherapy (30-40 Gy administered in fractions of 2 to 4 Gy, 3 to 4 x / week) is an effective therapeutic adjunct in the tumor stage (IIB), and is particularly suitable for localized tumor lesions [28]. Neelis KJ et al. [29] used low-dose external beam radiation therapy to treat mycosis fungoid lesions refractory to PUVA and topical steroids. They reported that 65 lesions in 24 patients treated with 8 Gy in two fractions had a response rate of 92% with no dermal toxicity noted [29]. Thomas et al. found a complete response rate of approximately 94.4% among 58 patients with CTL treated with a fraction of radiation therapy, the majority between 7 and 8 Gy, using either photons or electrons. The mean follow-up time was 41.3 months, and no significant long-term side effects were observed [30]. Low-dose total skin electron beam therapy has also shown satisfactory results with a good side effect profile for patients with more diffuse skin disease [31, 32]. Brachytherapy is a form of radiation therapy, whereby a radioactive source is placed near the tumor, either directly next to or inside the tumor itself. This procedure delivers a high dose of radiation to the target with only a minimal dose affecting surrounding tissue. One of these applicators is the catheter flap, which is designed to allow the HDR Ir-192 source to travel 5mm from the skin surface. This method is non-invasive and ideal for delivering tumoricidal doses of radiotherapy to superficial lesions while limiting the unfavorable delivery of radiation to healthy tissue due to the rapid drop in dose at the periphery of the lesions [33]. This is particularly desirable when treating anatomical sites close to tissues vulnerable to irradiation or which present significant cosmetic challenges to surgical excision such as the scalp, face and hands [33,34]. There are few reports using brachytherapy for CTCL [33]. DeSimone et al. reported on 10 patients with facial fungal mycosis lesions successfully treated with HDR brachytherapy doses of 8 Gy in 2 fractions of 4 Gy. There was no recurrence during the median 6-month follow-up period [35]. Goddard AL et al. [23] presented a case series using HDR brachytherapy for the treatment of acral skin lesions of CTCL in six patients with eight lesions also treated with 8 Gy in 2 fractions.
They reported an 88% control rate with a single locally recurrent lesion in an average follow-up period of 15.8 months [23]. Tao J et al. [33] reported a patient with multiple CTCL on bilateral feet. His lesions on both feet were successfully treated with a total of 8 Gy in two fractions by high dose rate surface brachytherapy Freiburg Flap Applicator, followed by 20 Gy in 10 fractions. Electronic treatments using an external 6 MeV beam on the dorsal large lesions. Both feet were still in remission at his last follow-up 21 months and 19 months after completing his left and right foot treatments, respectively [33]. Regarding the prognosis, CTCL are lifelong disorders that recur after stopping treatment, even in cases that do not progress [36]. Despite the introduction of several treatment options for CTCLs, as they progress and become refractory to treatment, malignant cells have the propensity to infiltrate lymph nodes and peripheral blood vessels, resulting in debilitating conditions. Progression to the stage of the tumor where neoplastic cells spread to lymph nodes and internal organs have been reported in less than 5% of cases with CTCL [9]. Patients with MF have a course lasting from several years to several decades; many of them die of unrelated disorders, while about 25% of them die of lymphoma [19]. Immunosuppression and opportunistic infections are the most common causes of death disease [37]. Therefore, it is almost impossible to draw a conclusion about the efficacy and toxicity of contact brachytherapy in the extremities. One of the largest reported series of contact brachytherapy (radon mold technique) in non-melanoma skin cancer comes from the Peter McCallum Cancer Center [38]. It included 642 patients with lesions located in the upper limbs in 49% of cases and lower limbs in 17%. Gauden et al. Reported from a series of 200 patients with 236 lesions treated, of which 26 were located at the extremities [39]. The treatment was delivered by HDR-BT, with a Leipzig type applicator. Total dose of 36Gy, in 3 Gy per fraction, administered daily 2 weeks were prescribed between 3- and 4-mm. Local control was obtained in 98% of cases. Grade 1 acute dermal toxicity was reported in 168 treated lesions (71%) and grade 2 in 81 (34%). The results were rated as good or excellent in 208 cases (88%). Late cutaneous hypopigmentation was noted in 13 cases (5.5%). Four patients relapsed locally and underwent subsequent surgery. Svoboda et al. published a series of 137 skin lesions of various pathologies (primary skin cancers and skin metastases of other origins) treated in 87 HDR-BT patients. The total dose ranged from 12 to 50 Gy, administered in 1 to 15 prescribed fractions to the surface of the applicator. Out of 137 lesions treated, 16 lesions located on the hands, 5 on the arms and 13 on the lower limbs. A complete response was obtained in all but four Basal Cell Carcinomas (BCC). The authors reported no late toxicity [40]. Joslin and his colleagues developed on 20 cases treated with 45 to 47.5 Gy in 10 or 11 fractions [41]. Poor healing affected 3 cases, with superficial necrosis involving 3 other cases (one after injury).
The authors concluded that careful consideration of dosesplitting regimens is needed in the lower extremities. Many articles published in the field are relatively old, some presenting older techniques in radiotherapy and pre-computer BT planning. This also happens in the references section below. Unfortunately, information on significant radiation toxicity due to BT persists among our non-BT colleagues. It's ultimately up to us to increase our BT presence and show a wider audience not only what we are doing well, but also what we have learned from past experience, and how BT has improved in terms of business. Efficiency and safety for our patients and staff. There is, however, an increased number of recently observed publications on the benefits of cutaneous BT, including the location of the legs [42, 43, 44, 45]. Some of these publications focus specifically on the benefit of BT in the elderly population [46, 47]. We sincerely hope that our readers will contribute more to this trend. The prognosis for the SS is poor. Its median survival rate is 2 to 4 years 1, and its 5-year survival rate is approximately 18% to 20% [48].
Conclusion
Cutaneous T Cell Lymphoma (CTCL) is a debilitating disease that has a serious impact on the quality of life. This case report demonstrates that HDR brachytherapy provides excellent results for the local control of CTCL lesions, offering a consistent and controlled dosage for topographic sites with little or no toxic skin effects.
References
- Bekkenk MW, Geelen FA, Vader PC, Heule F, Geerts ML, et al. Primary and secondary cutaneous CD30 (+) lymphoproliferative disorders: A report from the dutch cutaneous lymphoma group on the long-term follow-up data of 219 patients and guidelines for diagnosis and treatment. Blood. 2000;95:3653-3661.
- Kaye FJ, Bunn PA Jr, Steinberg SM, Stocker JL, Ihde DC, et al. A randomized trial comparing combination electron-beam radiation and chemotherapy with topical therapy in the initial treatment of mycosis fungoides. N. Engl J Med. 1989;321:1784-1790.
- Salah HB, Bahri M, Turki H, Abdelmoula M, Frikha M, et al. Radiotherapy for cutaneous cancers with xeroderma pigmentosum. Cancer Radiother. 2011;15:400-403.
- Cotter SE, Devlin PM, Sahni D, Hansen JL, O’Farrell DA, et al. Treatment of cutaneous metastases of merkel cell carcinoma with surface-mold computer-optimized high-dose-rate brachytherapy. J Clin Oncol. 2010;28:464- 466.
- Bagherani N, Smoller BR. An overview of cutaneous T cell lymphomas. F1000Res. 2016; 5:1882.
- Willemze R, Kerl H, Sterry W, Berti E, Cerroni L, et al. EORTC classification for primary cutaneous lymphomas: A proposal from the cutaneous lymphoma study group of the European Organisation for Reasearch and Treatment of Cancer. Blood. 1997;90:354-371.
- Jaffe ES, Harris NL, Diebold J, Muller-Hermelink HK. World health organization classification of neoplastic diseases of the hematopoietic and lymphoid tissues. A progress report. Am J Clin Pathol. 1999;111:8-12.
- Willemze R, Meijer CJ. EORTC classification for primary cutaneous lymphomas: the best guide to good clinical management. european organization for research and treatment of cancer. Am Dermatopathol. 1999;21:265-273.
- Rodd AL, Ververis K, Karagiannis TC. Current and emerging therapeutics for cutaneous t-cell lymphoma: histone deacetylase inhibitors. Lymphoma, 2012:1-10.
- Sidiropoulos KG, Martinez-Escala ME, Yelamos O, Guitart J, Sidiropoulos M. Primary cutaneous T-cell lymphomas: a review. J Clin Pathol. 2015;68:1003-1010.
- Kirsch IR, Watanabe R, O’Malley JT, Williamson DW, Scott LL, et al. TCR sequencing facilitates diagnosis and identifies mature T cells as the cell of origin in CTCL. Sci Transl Med. 2015;7:308ra158.
- Hughes CF, Newland K, McCormack C, Lade S, Prince HM. Mycosis fungoides and Sézary syndrome: Current challenges in assessment, management and prognostic markers. Australas J Dermatol. 2015;57:182-191.
- Benjamin Chase A, Markel K, Tawa MC. Optimizing care and compliance for the treatment of mycosis fungoides cutaneous t-cell lymphoma with mechlorethamine gel. Clin J Oncol Nurs. 2015;19:131-139.
- Alanteri E, Usmani S, Marafi F, Esmail A, Ali A, et al. The role of fluorine-18 fluorodeoxyglucose positron emission tomography in patients with mycosis fungoides. Indian J Nucl Med. 2015;30:199-203.
- Smoller BR, Bishop K, Glusac E, Kim YH, Hendrickson M. Reassessment of histologic parameters in the diagnosis of mycosis fungoides. Am J Surg Pathol. 1995;19:1423-1430.
- Kash N, Massone C, Fink-Puches R, Cerroni L. Phenotypic variation in different lesions of mycosis fungoides biopsied within a short period of time from the same patient. Am J Dermatopathol. 2016;38:541-545.
- Gibson JF, Huang J, Liu KJ, Carlson KR, Foss F, et al. Cutaneous T-cell lymphoma (CTCL): current practices in blood assessment and the utility of T-cell receptor (TCR)-Vβ chain restriction. J Am Acad Dermatol. 2016;74:870-877.
- Aggarwal S, Topaloglu H, Kumar S. Systematic review of burden of cutaneous t-cell lymphoma. Value Health. 2015;18:38.
- Eklund Y, Aronsson A, Schmidtchen A, Relander T. Mycosis Fungoides: A Retrospective Study of 44 Swedish Cases. Acta Derm Venereol. 2016;96:669-673.
- Lauenborg B, Christensen L, Ralfkiaer U, Kopp KL, Jønson L, et al. Malignant T cells express lymphotoxin α and drive endothelial activation in cutaneous T cell lymphoma. Oncotarget. 2015;6:15235-15249.
- Katona TM, O’Malley DP, Cheng L, Hiatt KM, Wang M, et al. Loss of heterozygosity analysis identifies genetic abnormalities in mycosis fungoides and specific loci associated with disease progression. Am J Surg Pathol. 2007;31:1552-1556.
- Tandberg DJ, Craciunescu O, Kelsey CR. Radiation therapy for cutaneous t-cell lymphomas. Dermato Clin. 2015;33:703-713.
- Goddard AL, Vleugels RA, LeBoeuf NR, O’Farrell DA, Cormack RA, et al. Palliative therapy for recalcitrant cutaneous t-cell lymphoma of the hands and feet with low-dose, high dose-rate brachytherapy. JAMA Dermatol. 2015;151:1354-1357.
- London Cancer. Skin Cancer Radiotherapy Guidelines. 2013.
- Royal College of Radiologists UK. Radiotherapy dose fractionation 3rd edition. 2019.
- Allan E, Stanton A, Pye, D. Fractionated high dose rate brachytherapy moulds-a precise treatment for carcinoma of the pinna. Radiother Oncol. 1998;48:277-281.
- Ahmed SK, Grams MP, Locher SE, McLemore LB, Sio TT, et al. Adaptation of the stanford technique for treatment of bulky cutaneous t-cell lymphoma of the head. Pract Radiat Oncol. 2016;6:183-186.
- Chmielowska E, Studzinski M, Giebel S, Krause A, Olejniczak M, et al. Follow-up of patients with mycosis fungoides after interferon α2b treatment failure. Postepy Dermatol Alergol 2015;32:67-72.
- Elsayad K, Kriz J, Moustakis C, Scobioala S, Reinartz G, et al. Total skin electron beam for primary cutaneous t-cell lymphoma. Int J Radiat Oncol Biol Phy. 2015;93:1077-1086.
- Gamsiz H, Beyzadeoglu M, Sager O, Dincoglan F, Uysal B, et al. Evaluation of mycosis fungoides management by total skin electron beam therapy with “translational technique. J Buon. 2015;20:1124-1131.
- Micaily B, Miyamoto C, Kantor G, Lessin S, Rook A, et al. Radiotherapy for unilesional mycosis fungoides. Int J Radiat Oncol Biol Phys. 1998;42:361-364.
- Neelis KJ, Schimmel EC, Vermeer MH, Senff NJ, Willemze R, et al. Low-dose palliative radiotherapy for cutatneous B- and T-cell lymphomas. Int J Radiat Oncol Biol Phys. 2009;74: 154-158.
- Thomas TO, Agrawal P, Guitart J, Rosen ST, Rademaker AW, et al. Outcome of patients treated with a single-fraction dose of palliative radiation for cutaneous T-cell lymphoma. Int J Radiat Oncol Biol Phys. 2013;85:747-753.
- Kamstrup MR, Lindahl LM, Gniadecki R, Iversen L, Skov L, et al. Low-dose total skin electron beam therapy as a debulking agent for cutaneous T-cell lymphoma: an open-label prospective phase ii study. Br J Dermatol. 2012;166:399-404.
- Kroeger K, Elsayad K, Moustakis C, Haverkamp U, Eich HT. Low-dose total skin electron beam therapy for cutaneous lymphoma: Minimal risk of acute toxicities. Strahlenther Onkol. 2017; 193:1024-1030.
- Tao J, Hentz C, Mysz M, Rashed I, Eilers D, et al. Extensive cutaneous t-cell lymphoma of the feet treated with high-dose-rate brachytherapy and external beam radiation. Case Rep Dermatol Med. 2018;5610925:1-6.
- Alam M, Nanda S, Mittal BB, Kim NA, Yoo S. The use of brachytherapy in the treatment of nonmelanoma skin cancer: a review. J Am Acad Dermatol. 2011;65:377- 388.
- DeSimone JA, Guenova E, Carter JB, Chaney KS, Aldridge JR, et al. Low-dose high-dose-rate brachytherapy in the treatment of facial lesions of cutaneous T-cell lymphoma. J Am Acad Dermatol. 2013;69:61-65.
- Rook AH, Gelfand JM, Wysocka M, Troxel AB, Benoit B, et al. Topical resiquimod can induce disease regression and enhance T-cell effector functions in cutaneous T-cell lymphoma. Blood 2015;126:1452-1461.
- Moyal L, Feldbaum N, Goldfeiz N, Rephaeli A, Nudelman A, et al. The therapeutic potential of an-7, a novel histone deacetylase inhibitor, for treatment of mycosis fungoides/sezary syndrome alone or with doxorubicin. PLoS One. 2016;11:0146115.
- Ainslie J, Ashby MA, Pacella JA. Use of radon mould technique for skin cancer: results from the Peter MacCallum Cancer Institute (1975-1984). Br J Radiol. 1989;62:608-612.
- Gauden R, Pracy M, Avery AM. HDR brachytherapy for superficial non-melanoma skin cancers. J Med Imaging Radiat Oncol. 2013;57:212-217.
- Svoboda VH, Kovarik J, Morris F. High dose-rate microselectron molds in the treatment of skin tumors. Int J Radiat Oncol Biol Phys. 1995;31:967-972.
- Joslin CA, Flynn A, Hall EJ. Principles and practice of brachytherapy using after loading systems. Arnold Publishers. 2001;389.
- Guinot JL, Rembielak A, Perez-Calatayud J. GECESTRO ACROP recommendations in skin brachytherapy. Radiother Oncol. 2018;126:377-385.
- High dose rate brachytherapy in elderly patients with non-melanoma skin cancer-clinical and cosmetic outcome. RSNA press. 2019.
- Keen CE. HDR brachytherapy effective for elderly patients with nonmelanoma skin cancers. Applied Rad Oncol. 2020.
- Likhacheva A, Awan M, Barker CA. Definitive and postoperative radiation therapy for basal and squamous cell cancers of the skin: executive summary of an American society for radiation oncology clinical practice guideline. Pract Radiat Oncol. 2020;10:8-20.
- Lancellotta V, Kovács G, Tagliaferri L. The role of personalized interventional radiotherapy (brachytherapy) in the management of older patients with non-melanoma skin cancer. J Geriatr Oncol. 2019;10:514-517.
- Lancellotta V, Kovács G, Tagliaferri L. Age is not a limiting factor in interventional radiotherapy (brachytherapy) for patients with localized cancer. Biomed Res Int. 2018;2178469;1-10.
- Väkevä L, Niittyvuopio R, Leppä S, Heiskanen J, Lindström V, et al. Allogeneic haematopoietic stem cell transplantation for patients with cutaneous t-cell lymphoma. Acta Derm Venereol. 2016;96:816-817.