Research Article - Onkologia i Radioterapia ( 2020) Volume 14, Issue 3
A clinical study on GIST in Babylon
Monem Makki Alshok*Monem Makki Alshok, Department of Medicine, College of Medicine, University of Babylon, Babylon, Iraq, Email: dr_monem_alshok@yahoo.com
Received: 08-Jun-2020 Accepted: 22-Jun-2020 Published: 30-Jun-2020
Abstract
GIST (Gastrointestinal Stromal Tumor) is a type of soft tissue sarcoma arising from interstitial cell of Cajal in GIT. It is important to note that all GISTs can be cancerous. Disease-free survival rates (refer only to the percentage of people who experience a complete remission after finishing treatment) can be fulfilled by early surgery and therapy using tyrosine kinase inhibitor.
Aim: To study the behaviour of this uncommon GIT tumour in our patients and the overall survival rate following medical treatment.
Method: 34 patients with GIST were studied in Merjan Teaching hospital, their diagnosis made on OGD and histopathology and Immunohistochemistry. The period of the study started during the year 2013 to end of January 2015, the clinical manifestation and presentation and the main methods of treatment are studied.
Results: From these 32 patients with GIST, their age range from 22 years to 88 years with mean of 41, 16 are male and 18 are female making slight female preponderance. The main method of diagnosis is by OGD and two patients present with abdominal mass and anaemia and the diagnosis of GIST was made after laparotomy and histopathology and immunohistochemistry using KIT and CD 34 tests. The most common clinical presentations include anaemia, upper GIT bleeding and dyspepsia. All the patients were treated by Imatinib (30) and Sunitinib (2). Two patients died during the course of treatment making mortality rate of 6.3%. The overall prognosis and survival were determined by site, size and spread of tumour and 26 patients continue to receive CT during the year 2014. In conclusion GIST is a kind of tumour that has a wide spectrum of malignant potential and there is no clear risk factor were identified.
Keywords
Clinical study, Gastrointestinal stromal tumours, BabylonIntroduction
GIST is one of the uncommon tumour of the alimentary system and account for 1%-3% of primary gastric neoplasms and represents less than 1% of all GIT tumours and could be malignant [1]. The incidence is 15-20 per million, more in male, it can occur at any age and it is rarely familial or genetic and is sporadic in most patients and the tumour rarely reach more than 10 cm in size. GIST is a type of soft tissue sarcoma, whose classification is controversial, surgery is the treatment of choice and its prognosis is determined by tumour size and involvement of neighbouring organs and the presence of metastasis and in selected cases minimally invasive surgery can be performed with excellent results [2,3]. These tumours originally start at the (ICCs) Interstitial Cells of Cajal and these cells of autonomic nervous system and sometime ICCS are called pacemaker cells of Gastrointestinal Tracts (GIT), because they signal the smooth muscles in the digestive system to contract to propel food and liquid through GIT. More than half of GISTs start in the stomach, but GISTs can start anywhere along the GIT. A small number originate outside the GIT in nearby areas such as the omentum or peritoneum [4].
Epidemiology
As mentioned [3,4] incidence is 15-20 per million and currently, there are very few known risk factors for GISTs [5]. These tumours can occur in people of any age, but they are rare in people younger than 40 and most common in people aged 50-80 year [6]. Most GISTs are sporadic (not inherited) and have no clear etiology. In a very rare occasions GISTs have been found in more than one members of the same family and it had been found that these family members inherited gene mutation that can lead to GIST, most often this syndrome is caused by an abnormal c-kit gene passed from parent to child [7]. The c-kit gene is the same gene that is mutated in most sporadic GISTs, and people, who inherited this abnormal gene from parent have it in all their cells, whereas people with sporadic GIST only have it in the tumour cells. In a few cases, a change in a different gene, the PDGFRA gene, causes this syndrome and the defective PDGFRA genes, are found in about 5% to 10% of sporadic GISTs There is also rare association of GISTs with neurofibromatosis, and people with this rare condition have an increased risk of GISTs (most often in the stomach) as well as nerve tumor called paragangliomas and GISTs developing in these people are in their teens (20s) and caused by a change in one of the Succinate Dehydrogenase (SDH) gene, which is passed from parent to child [8].
GIST were originally classified as another type of neoplasm (Leiomyoma, Leiomyoblastoma or Leiomyosarcoma) due to its histological similarity, however advances in molecular biology and immunohistochemistry have allowed them apart from other GIT malignancies and define them as distinct clinical and histopathological entity with immunohistochemistry commonly positive for proto-oncogen c-kit (CD117), a receptor Tyrosine Kinase Protein (RTK) which is not seen in other GIT neoplasms [9,10]. Chemotherapy targeting RTK such as Imatinib and Sunitinib, has shown efficacy in GISTs and despite the efficacy of RTK inhibitor, the definitive management of GIST is surgical intervension. Other types of chemotherapy and radiation therapy are used much less often. The targeted therapy like Imitinab (Gleevec) and Sunitinab (Sutent) often work, but they still are not sure exactly how and when to give them to make them more effective [10]. Should they be given to all patients, even to the patients with very small tumor? Other drugs that targeted the KIT or PDGFRA proteins, are also being studied for use against GISTs. Some of these such as Sorafenib (Nexavar), Nilotinib (Tasigna) and Dasatinab (Sprycel) have helped some patients in early studies, but it isn't clear which one, if any would be the best one to use after standard targeted drugs stopped working. Other drugs being studied like B11B021, are aimed at different targets involved with cancer, such as heat shock proteins [11,12]. Comprehensive Cancer Network recommends resection of masses ≥ 2 cm and close surveillance for small tumours without high risk features. Adjuvant chemotherapy with Imatinib for at least 3 years to decrease risk of recurrence, as all GISTs are considered malignant with potential to metastasize. Regorafenib and Ponatinib are used in refractory cases. A new study has shown an effectiveness of noval therapy including Ipilimumab and Nivolumab. On January 2020, the FDA approved Avapritinib, as first line therapy for unresectable or metastatic GIST exhibiting PDGFRA exon 18 mutation including D842V mutation. In GISTs location, size of tumour and mitotic rate determine, the higher risk of metastasis.
Patients and Methods
In this paper a clinical study is performed on a number of cases presented to GIT unit and oncology unit in Babylon.
34 patients with GIST were studied in Merjan Teaching hospital, GIT endoscopy unit and oncology unit, their diagnosis made on history and clinical presentation. Imaging studies including U/S, CT scan are modality of choice. CT-PET scan occasionally done and especially for assessment of therapeutic response. Upper and lower GIT endoscopy done and including endoscopic U/S for small tumours. MRI scan done in cases of rectal GISTs (Table 1).
| Type | Used for |
|---|---|
| CT-scan | Demonstration of site, location, any invasion and metastasis. |
| For CT-guided biopsy and sensitivity and response to therapy | |
| MRI scan | Rectal GISTs, any haemorrhage and liver secondary |
| PET-CT scan with 2 (F-18 ) Flouro-2-deoxy-D-glucose | Tumour staging |
| Differentiation between benign and malignant type and study effectiveness of adjuvant therapy | |
| Ultrasonography | Not the primary method used to visualize GISTs, but it can visualize tumour size more than 5 cm |
Table 1. Demonstrate type of imaging studies used for GISTs
Biopsy of the lesions endoscopic or resection biopsy studied for histopathology and tissue immunohistochemical staining for KIT (Receptor Tyrosine Kinase), CD 117 test transmembrane glycoprotein. We were unable to do Platelets derived growth factor receptor A (PDGFRA) mutation which can be demonstrated in 5% of GISTs cases and also another marker CD34, Desmin, Actin S10012.
The period of the study started during the end of the year 2013 to the end of the year 2015. Clinical presentation and the main methods of treatment will be mentioned.
Result
In this study 34 patients were studied, their age range from 22-80 years and a mean age of 41 years. 16 patients are male and 18 are female making slight female preponderance. The main method of diagnosis is by OGD, and in 2 patients whom they present with anemia and abdominal mass, the diagnosis was made after laparotomy, histopathology and immunohistochemistry using KIT and CD 34 tests.
The most common clinical presentation include: Anemia, Upper GIT bleeding and dyspepsia, and 30 patients were treated with Imitinib and the remaining patients received Sunitinib. One patient died during the course of treatment making a mortality rate of 3.1%.
The overall prognosis and survival were determined by site, size and presence or absence of spread or metastasis. We noticed that 26 patients continue to receive the treatment and follow up during the year of follow up 2014 and 2015.
The location and site of GISTs in the reported cases were as the following: Gastric 72%, Intestinal 20%, Extra intestinal 8%. The clinical presentation were nonspecific, mainly depends on the site of GIT involvement include GIT bleeding noticed in 6 cases (hematemesis), abdominal discomfort, distension and pain with mild anemia, 2 patients present with intestinal obstruction. Asymptomatic presentation or mild dyspepsia noticed in about 30% of patients.
During Upper GIT endoscopy we demonstrate the typical endoscopic appearance of well circumscribed tumour with some kind of mild ulceration on the top in over 50% of case particularly gastric GIST (Figures 1-3).
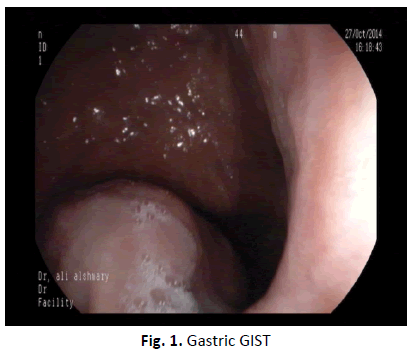
Figure 1. Gastric GIST
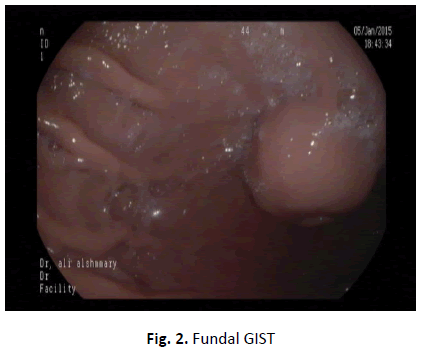
Figure 2. Fundal GIST
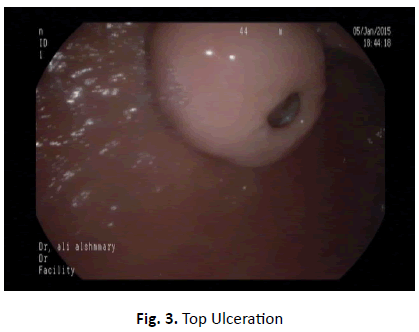
Figure 3. Top Ulceration
On histopathological examination, the lesion commonly involves the muscularis properia and with appearance of spindle cell type (Figure 4).
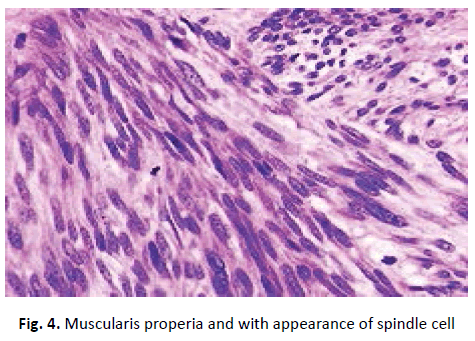
Figure 4. Muscularis properia and with appearance of spindle cell
The CT scan appearance was demonstrated in (Figure 5) as heterogenous mass with central necrosis.
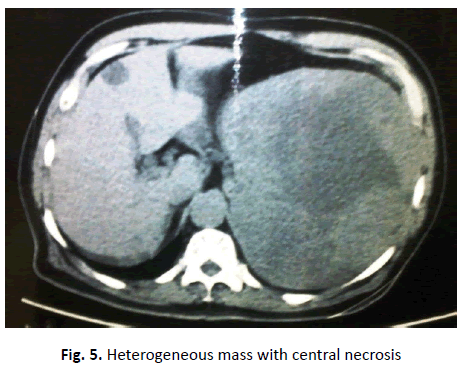
Figure 5. Heterogeneous mass with central necrosis
The typical EUS appearance of Gastric GIST demonstrated as one patient was sent to GIT hospital in Baghdad (Figure 6).
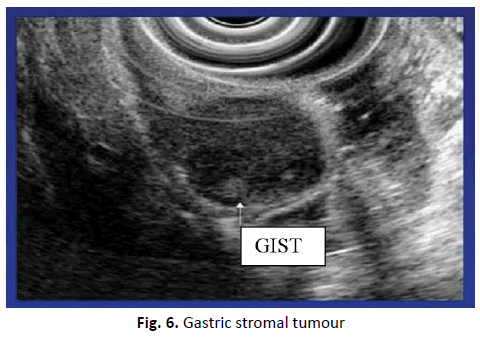
Figure 6. Gastric stromal tumour
Discussion
GIST is an uncommon tumour [13] of the most usual age at presentation is, above 40 years as had been demonstrated in the studied patients and most recorded cases were in age group ≥ 30 only one of them of 22 years of age, and in a study [13] demonstrated a male to female ratio of 2:1, but in our studied patients there is slight female preponderance and this could be explained on small number of recorded cases of GIST. Due to small size of the studied GIST cases series, we can't consider the stomach as a common site of involvement, and the tumor may range in its size and small size tumours usually benign [14,15]. Numerous studies have attempted to delineate criteria to distinguish benign GISTs from malignant GISTs, and it was found that tumor size and mitotic rate are the most consistently the strongest predictors [7,15]. In the present study correlation was found between the size of tumor and biologic behaviour with almost all small tumours showed benign nature except in one small intestinal Stromal tumor which on microscopy shows malignant features though the size of tumor less than 3 cm in size. Mostly GISTs are solid tumours, but larger size tumors may be cystic, because of local necrosis in malignant tumour. Difficulties may arise in pelvic GISTs or extra intestinal GISTs due to confusion with ovarian tumours [8,16]. In one of the studied cases, that was initially sonographically considered as ovarian tumour, but on histopathologic examination and immunohistochemical studies was considered as extra intestinal GIST. According to some of the scientist, benign behaviour is common in gastric GISTs [17], whereas the benign tumours outnumber malignant ones by a ratio of 3-5:1 and 20%-25% of gastric GISTs and 40%-50% of small intestinal GISTs are malignant [18]. In this study the ratio of benign to malignant GISTs was less than 1.25:1. In literatures [19] Gastric GISTs showed 75% benign nature and 25% malignant, means 3:1, small intestinal GISTs 40% benign cases and 60% malignant cases 2:3 as had been noticed in the present study. Extra intestinal GISTs (EGISTs) are rarely reported group of tumours that arise outside GIT, but histologically resemble their GIT counterpart [20]. Few scientist suggested that most, if not all, cases of EGISTs are likely to represent mural GISTs with extensive extramural growth with eventual loss of contact with muscle layer of the gut [21]. Approximately 80% are located in the omentum or mesentery and the remainder develop in the retro peritoneum and rarely in the gallbladder and urinary bladder. Gross appearance of EGIST varies from firm, fleshy gray-red masses to cystic ones. They lack the whorled appearance of the conventional smooth muscle tumours. Cystic change is seen in the majority, associated with extensive haemorrhage or necrosis [22]. A high index of suspicion of GIST should be kept in mind if we met a large cystic abdominal mass preoperatively when associated with intraoperative cystic fluid content [23]. In one study [24] surgical resection of GISTs cases showed 40% are mesenteric, 40% are retroperitoneal and occurrence of GISTs in the urinary bladder is very rare. In the present cases there is demonstration that tumor abutting the muscle coat of urinary bladder, but with no infiltration into it and no extension to the large bowel. In 3 out of 5 cases of EGISTs reported in women with mean age of 38 years and as we mentioned there is clinical and sonological suspicion of ovarian cyst in this case. The main differential histopathological diagnosises, which should be considered in the differential diagnosis of EGISTs include Fibromatosis, Smooth Muscle tumours, Neural tumours, Malignant Fibrous Histiocytomas (MFH). The distinction from Fibromatosis can be made on histopathlogical examination [25]. In fibromatosis cellularity is low and it is composed of spindle cell within collagenous stroma, necrosis and mitotic activity are not seen. Other conditions can be differentiated from EGISTs on basis of morphology and immunohistochemistry [26,27]. In the present cases no other tumors or masses were found and ruling out the possibility of metastasis and positive immunohistochemical staining for CD 117 is the defining feature of GISTs and helping us in establishing the diagnosis. The main types of treatment used for GISTs cases in general include Surgery, the use of targeted chemotherapy like imatinib (Gleevec) and Sunitinib (Sutent), other treatment like other chemotherapy and radiotherapy are used much less often and in the present study we were unable to use other kinds of treatment such as Sorafenib (Nexavar), Nilotinib (Tasigna) and Dasatinib (Sprycel) that have helped some patients in another studies [28-34].
Conclusion
GISTs have a wide spectrum of malignant potential, there is no clear risk factors for GIST were identified in the present study and surgical resection remains the gold standard in the treatment and a large studies establish the promising role of the adjuvant and neo-adjuvant therapy for better disease- free survivals.
References
- Friedman L, Brandt L. Sleisenger and Fordtran’s gastrointestinal and liver disease. Tenth edition 2016 by Saunders. An imprint of Elsevier. 2010.
- El-Menyar A, Mekkodathil A, Al-Thani H. Diagnosis and management of gastrointestinal stromal tumors: An up-to-date literature review. J Can Res Ther. 2017;13:889-900.
- Wang ZH, Liang XB, Wang Y, Ma GL, Qu YQ, et al. Epidemiology survey of gastrointestinal stromal tumor in Shanxi Province in 2011. Zhonghua Yi Xue Za Zhi. 2013;93:2541-2544.
- Bokhary RY, Al-Maghrabi JA. Gastrointestinal stromal tumors in Western Saudi Arabia. Saudi Med J. 2010;31:437-441.
- Wang L, Vargas H, French SW. Cellular origin of gastrointestinal stromal tumors: A study of 27 cases. Arch Pathol Lab Med. 2000;124:1471-1475.
- Vasundhara G, Parvathi A, Bhagya L. Our experience with gastrointestinal stromal tumors over a period of three years from a tertiary care centre. Intern J Res Med Sci. 2016;4:3709.
- Al-Shoha M. A case of synchronous gist and MALToma. World Congress of Gastroenterology. 2016.
- Zuluaga, Siljic I, Valencia J, Cardona U, Benitez J. Gastric gist tumors resolution of video laparoscopy: report of 22 cases. World Congress of Gastroenterology. 2017.
- Michael M, Simi S, Ilan W. A case of gist bleed: a challenging endoscopic diagnosis. World Congress of Gastroenterology. 2017.
- Michelle AM, Marino-Enriquez A, Fletcher J, Dorfman D, Raut C, et al. Thyroid hormone inactivation in gastrointestinal stromal tumors. N Eng J Med GISTs. 2014;370:1327-1334.
- Von Mehren M. Soft tissue sarcoma, NCCN clinical practice guidelines in oncology. J Natl Compr Canc Netw. 2016;14:758-786.
- King DM. The radiology of gastrointestinal stromal tumors (GIST). Cancer Imaging. 2005;5:150-156.
- Blay JY, Bonvalot S, Casali P. Consensus meeting for the management of gastrointestinal stromal tumors. Ann Oncol. 2005;16:566-578.
- Spierings J, Vries E, Timens W, Groen H, Boezen M, et al. Expression of TRAIL and TRAIL death receptors in stage iii non-small cell lung cancer tumors. Clin Cancer Res. 2003;9:1-6.
- Fletcher CD, Berman JJ, Corless C. Diagnosis of gastrointestinal stromal tumors: a consensus approach. Hum Pathol. 2002;33:459-465.
- Hirota S, isozaki K, Moriyama Y. Gain-of-function mutations of c-kit in human gastrointestinal stromal tumors. Science. 1998;279:577-580.
- Rubin BP. Gastrointestinal stromal tumors: an update. Histopathology. 2006;48:83-96.
- Eisenberg BL, Judson I. Surgery and imitanib in the management of GIST: emerging approaches to adjuvant and neoadjuvant therapy. Ann Surg Oncol. 2004;11:465-475.
- Papanikolaou IS, Triantafyllou K, Kourikou A, Rösch T. Endoscopic ultrasonography for gastric submucosal lesions. World J Gastrointest Endosc. 2011;3:86-94.
- Kim GH, Park do Y, Kim S, Kim DH, Kim DH, et al. Is it possible to differentiate gastric GISTs from gastric leiomyomas by EUS? World J Gastroenterol. 2009;15:3376-3381.
- Akahoshi K, Sumida Y, Matsui N, Oya M, Akinaga R, et al. Preoperative diagnosis of gastrointestinal stromal tumor by endoscopic ultrasound-guided fine needle aspiration. World J Gastroenterol. 2007;13:2077-2082.
- Lau S, Tam KF, Kam CK, Lui CY, Siu CW, et al. Imaging of gastrointestinal stromal tumour (GIST). Clin Radiol. 2004;59:487-498.
- Yip D, Zalcberg J, Ackland S, Barbour AP, Desai J, et al. Controversies in the management of gastrointestinal stromal tumors. Asia Pac J Clin Oncol. 2014;10:216-227.
- Al-Thani H, El-Menyar A, Rasul KI, Al-Sulaiti M, El-Mabrok J, et al. Clinical presentation, management and outcomes of gastrointestinal stromal tumors. Int J Surg. 2014;12:1127-1133.
- Tirumani SH, Baheti AD, Tirumani H, O'Neill A, Jagannathan JP. Update on gastrointestinal stromal tumors for radiologists. Korean J Radiol. 2017;18:84-93.
- Vlenterie M, Flucke U, Hofbauer LC. Pheochromocytoma and gastrointestinal stromal tumors in patients with neurofibromatosis type I. Am J Med. 2013;126:174-180.
- Miettinen M, Lasota J. Gastrointestinal stromal tumors: pathology and prognosis at different sites. Semin Diagn Pathol. 2006;23:70-83.
- Miettinen M, Fetsch JF, Sobin LH, Lasota J. Gastrointestinal stromal tumors in patients with neurofibromatosis 1: a clinicopathologic and molecular genetic study of 45 cases. Am J Surg Pathol. 2006;30:90-96.
- Demetri GD, von Mehren M, Blanke CD. Efficacy and safety of imatinib mesylate in advanced gastrointestinal stromal tumors. N Engl J Med. 2002;347:472-480.
- Casali PG. Gastrointestinal stromal tumours: ESMO-EURACAN clinical practice guidelines for diagnosis, treatment and follow-up. Annals Oncol. 2018;29:68-78.
- Saleh N. Avapritinib approved for GIST Subgroup. Cancer Discov. 2020;10:334-334.
- Atallah C. Survival and treatment trends for small bowel and colorectal gastrointestinal stromal tumors. Gastroenterol. 2019;156:1411.
- Peng Liu, Fengbo Tan, Heli Liu, Bin Li, Tianxiang Lei, et al. The use of molecular subtypes for precision therapy of recurrent and metastatic gastrointestinal stromal tumor. Onco Targets Ther. 2020;13: 2433-2447.
- Chiao-En W, Chin-Yuan T, Shang-Yu W, Chun-Nan Y. Clinical diagnosis of gastrointestinal stromal tumor (gist): from the molecular genetic point of view. Cancers (Basel). 2019;11:679.



